Chemistry The Basics MR CONKEY PHYSICAL SCIENCE CHAPTER

Chemistry: The Basics MR. CONKEY PHYSICAL SCIENCE CHAPTER 2

A few things first… Precision V. S. Accuracy Precision – the gauge of how exact a measurement is. Significant Figures – all the digits that are known in a measurement, and the last digit that is estimated. Accuracy – the closeness of a measurement to the actual value that is being measured.

Organizing Scientific Data: Types of Graphs Tables: simplest way to organize data. Line graphs – useful for showing changes occurring in related variables. (e. g. change in temperature over time). Line graphs use slope = Rise/Run.
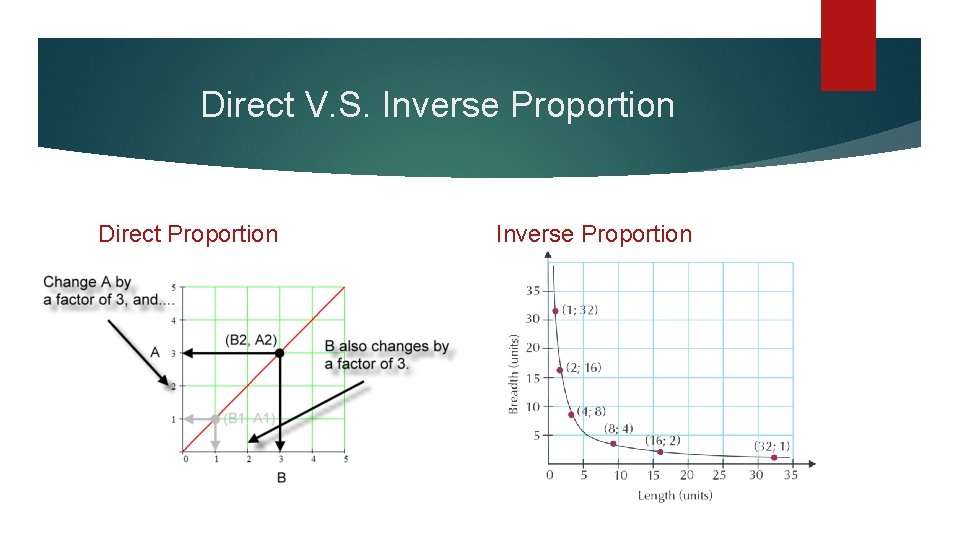
Direct V. S. Inverse Proportion Direct Proportion Inverse Proportion
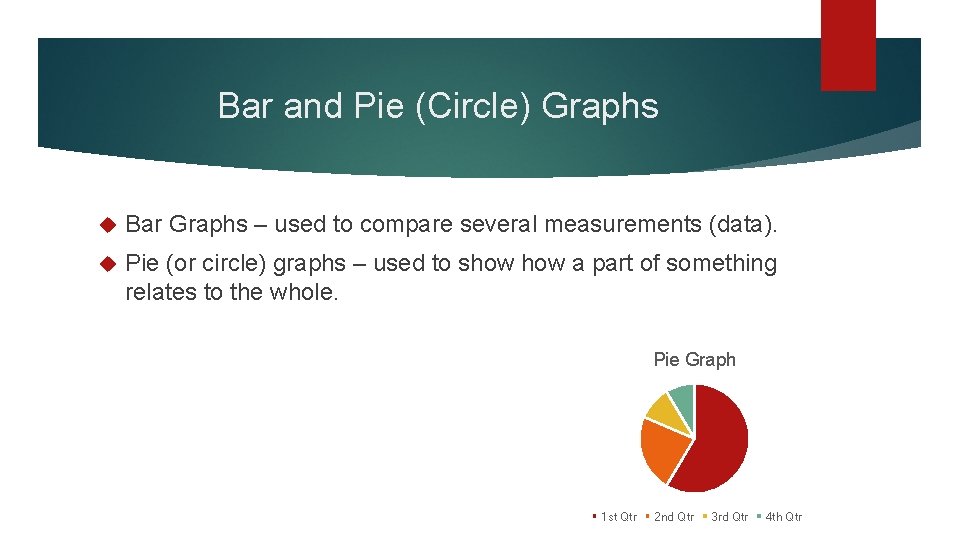
Bar and Pie (Circle) Graphs Bar Graphs – used to compare several measurements (data). Pie (or circle) graphs – used to show a part of something relates to the whole. Pie Graph 1 st Qtr 2 nd Qtr 3 rd Qtr 4 th Qtr

Classification of Matter – a small amount of the Universe that has both volume and mass and takes the form of a solid liquid or gas (the three states of matter on Earth). Pure Substances – matter that always has exactly the same composition. What are some examples? Elements – a substance that cannot be broken down into simpler substances. Atoms – the smallest particle of an element.

Compounds Compound – a substance that is made from two or more simpler substances and can be broken down into those simpler substances. (examples? ) They differ from the substances that make them up. Properties change when elements join together to make compounds (example: silicon dioxide). Compounds always contain two or more elements joined in a fixed proportion.

Mixtures They tend to retain some properties of their individual substances that make them up, but their properties are less constant than the properties of a substance. Properties of mixtures can vary because the composition of a mixture is not fixed. Mixtures are classified by how well the parts (substances) are distributed throughout the mixture.

Heterogeneous V. S. Homogeneous Mixtures Heterogeneous Mixture In a Heterogeneous mixture the parts of the mixture are noticeably different from one another. Comes from the Greek words hetero and genus meaning “different” and “kind” Homogeneous Mixture In a Homogeneous the substances are so evenly distributed that it is difficult to distinguish one substance in the mixture from another. It appears to contain only one substance.

Mixture Classification A mixture can be classified as a solution, a suspension, or a colloid based on the size of its largest particles. Solution – When substances dissolve and form a homogeneous mixture. (examples? ) Suspension – a heterogeneous mixture that separates into layers over time. Colloid – contains some particles that are intermediate in size between the small particles in a solution and the larger particles in a suspension. They do not separate into layers.

Physical Properties A physical property is any characteristic of a material that can be observed or measured without changing the composition of the substances in the material. Viscosity, conductivity, malleability, hardness, melting point, boiling point, and density are examples. Viscosity – the tendency of a liquid to keep from flowing. The greater the viscosity the slower the liquid moves.

Physical Properties (cont. ) Viscosity can be influenced by heat. (what do you think will happen to the honey? ) Discuss the importance of viscosity for a few minutes with your shoulder partners. Conductivity – A material’s ability to allow heat to flow through it. What is an example of a material with high conductivity? How about low conductivity?

Physical Properties (the third installment!) Malleability – the ability for a solid to be hammered without shattering. What is the opposite of Malleable? What are some examples of both? Hardness – when trying to determine if a material is harder than another you could compare the two by seeing which one scratches the other. (which is harder than the other in order from least to greatest: stainless steel knife, copper sheet, quartz, diamond, knife sharpener, mercury).
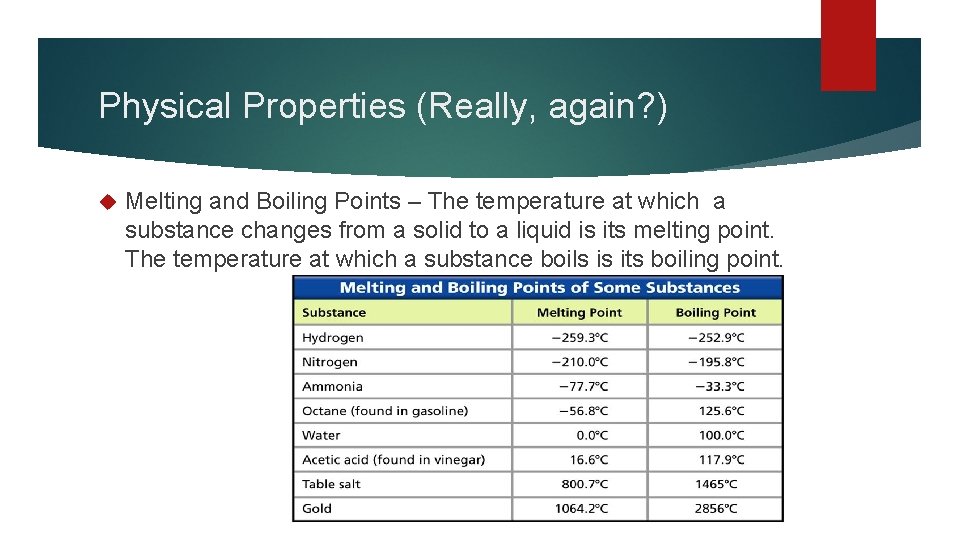
Physical Properties (Really, again? ) Melting and Boiling Points – The temperature at which a substance changes from a solid to a liquid is its melting point. The temperature at which a substance boils is its boiling point.

Density can be used to test the purity of a substance. Physical Properties are used to identify materials, to choose a material for a specific purpose, or to separate substances in a mixture.

Using Properties to Separate Mixtures Filtration – a process that separates materials based on the size of their particles (e. g. coffee). Distillation – a process that separates the substances in a solution based on their boiling points (e. g. seawater to fresh water).

Recognizing Physical Changes Physical Change – occurs when some of the properties of a material change, but the substances in the material remain the same. (what are some examples of this? )

Observing Chemical Properties Chemical Property – any ability to produce a change in the composition of matter. Two examples of chemical properties are flammability and reactivity. Flammability – a material’s ability to burn in the presence of oxygen. Reactivity – how readily a substance combines chemically with other substances.

Recognizing Chemical Changes Chemical Change – occurs when a substance reacts and forms one or more new substances. Three common types of evidence for a chemical change include: the production of gas, the formation of a precipitate, and a color change.

Recognizing Chemical Changes (Cont. ) Changes in color – a clue that a chemical change has produced at least one new substance. Production of gas – a release of gas is an indicator that a chemical change has occurred. Formation of a precipitate – Any solid that forms and separates from a liquid is a precipitate. How do you know a chemical change has occurred? Can the products be changed back to their original form?
- Slides: 20