Chemistry Table of Contents 1 Matter Measurement and

Chemistry Table of Contents 1. Matter, Measurement, and Problem Solving 2. Atoms and Elements 3. Molecules, Compounds, and Chemical Equations 4. Chemical Quantities and Aqueous Reactions 5. Gases 6. Thermochemistry 7. The Quantum Mechanical Model of the Atom Return to TOC

Chapter 1 Table of Contents 1. 1 Atoms and Molecules 1. 2 The Scientific Approach to Knowledge 1. 3 The Classification of Matter 1. 4 Physical and Chemical Changes and Physical and Chemical Properties 1. 5 Energy: A Fundamental Part of Physical and Chemical Change 1. 6 The Units of Measurement 1. 7 The Reliability of Measurement 1. 8 Solving Chemical Problems Return to TOC 2

Section 1. 1 Atoms and Molecules The Properties of Matter • • • The properties of matter are determined by the properties of atoms and molecules What does this mean? Water behaves like water because molecules of water have the properties of water – Sounds kind of like a circular definition Return to TOC 3

Section 1. 1 Atoms and Molecules The Properties of Matter • • • Think about it this way Lets say you had a glass of water And a glass of hydrogen peroxide Return to TOC 4
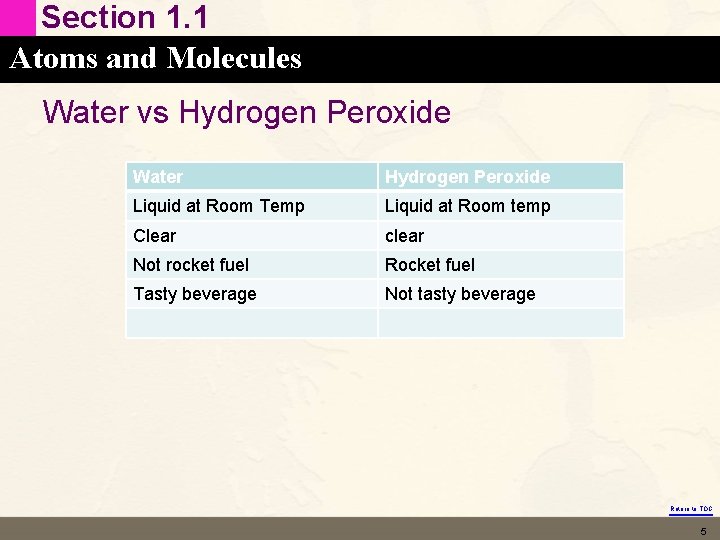
Section 1. 1 Atoms and Molecules Water vs Hydrogen Peroxide Water Hydrogen Peroxide Liquid at Room Temp Liquid at Room temp Clear clear Not rocket fuel Rocket fuel Tasty beverage Not tasty beverage Return to TOC 5

Section 1. 1 Atoms and Molecules Properties of Matter • • The question is WHY these properties are different. Why does a single oxygen make hydrogen peroxide so different from water? Return to TOC 6

Section 1. 1 Atoms and Molecules Chemistry • The Science that seeks to understand the behavior of matter (macroscopic) by studying the behavior of atoms and molecules (microscopic). • Why the extra oxygen in hydrogen peroxide makes its properties very different from water. Return to TOC 7

Section 1. 2 The Scientific Approach to Knowledge Scientific Laws • Often the same observations apply to many different systems. • This type of generalized behavior is formulated into a statement called a Scientific Law • A brief statements that summarizes past observations and predicts future ones • Describes what happens • Does not explain why. Return to TOC 8

Section 1. 2 The Scientific Approach to Knowledge Scientific Theory/Model • Laws and observations can combine to construct a Scientific Theory or Model • Tries to explain why something happens not just what happens. Return to TOC 9
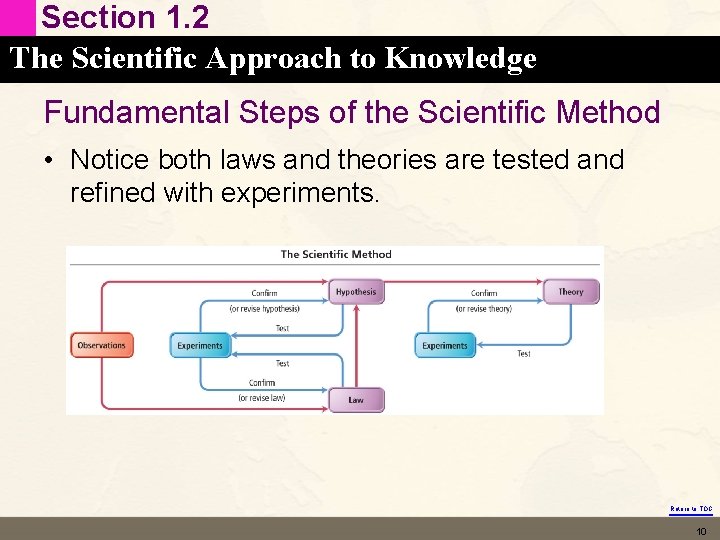
Section 1. 2 The Scientific Approach to Knowledge Fundamental Steps of the Scientific Method • Notice both laws and theories are tested and refined with experiments. Return to TOC 10

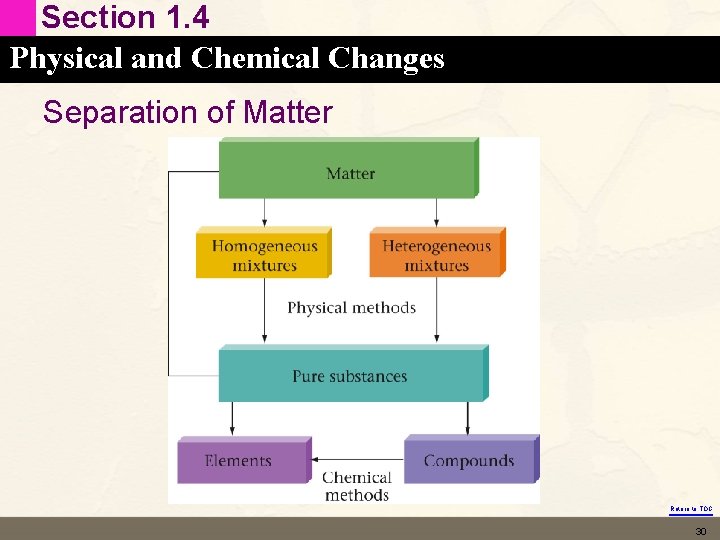
Section 1. 3 The Classification of Matter • Anything occupying space and having mass. • Two familiar ways to classify matter • Based on its “state” – solid vs liquid vs gas • Based on its composition – mixtures vs pure substances Return to TOC 11

Section 1. 3 The Classification of Matter States of Matter • Solid – Particles tightly packed – Rigid, Has fixed volume and shape. • Liquid – Particles loosely packed – Definite volume – No specific shape (takes on shape of container) • Gas – Lots of space between the particles – No fixed volume or shape. – Takes on the shape and volume of container. Return to TOC 12

Section 1. 3 The Classification of Matter States of Matter: Types of Solids • • Fig 1. 3 Crystalline (patterns with repeating order) • Diamond, salt, Amorphous (no shape) • Glass, plastic sodium silicate glass http: //www. tech. plym. ac. uk/sme/mats 324 c 1%20 fibres. htm Return to TOC 13

Section 1. 3 The Classification of Matter States of Matter: Compressibility • • • Fig 1. 4 Solids • Not at all Liquids • Somewhat Gases • Easy to compress Return to TOC 14

Section 1. 3 The Classification of Matter Return to TOC 15

Section 1. 3 The Classification of Matter Classifying Matter according to its Composition • Pure Substance vs Mixture – – Pure substance has constant composition Mixtures have variable composition – Na. Cl and H 2 O are two pure substances that combine to make a mixture (salt water) Return to TOC 16

Section 1. 3 The Classification of Matter Pure Substances • • Constant composition. Elements – – – • Hydrogen (H 2) Helium (He) Gold (Au) Compounds – – – Pure water (H 2 O) Carbon dioxide (CO 2) Sodium chloride (Na. Cl) Return to TOC 17

Section 1. 3 The Classification of Matter Element • Pure substance that cannot be broken down into other substances by chemical methods. • One type of atom • Examples: § Iron (Fe), aluminum (Al), oxygen* (O 2), and hydrogen* (H 2) * 7 elements in the periodic table always exist naturally in a diatomic state in their pure form Return to TOC 18

Section 1. 3 The Classification of Matter Compound • • • Pure substance that can be broken down into those elements by chemical methods. Two different types of atoms Examples: § Water (H 2 O), carbon dioxide (CO 2), table sugar (C 12 H 22 O 11). Return to TOC 19

Section 1. 3 The Classification of Matter Concept Check Categorize the following as elements vs compounds? H 2 O 2, NO 2, Mg, Na. OH, N 2, Cs Return to TOC 20

Section 1. 3 The Classification of Matter Mixtures • • Variable composition. Examples § Salt water, wine, coffee, sweetened tea • Can be separated into pure substances: – – • By physical method Elements and/or compounds. We can easily separate the water and the salt back out of a glass of salt water. How? Return to TOC 21

Section 1. 3 The Classification of Matter Homogeneous Mixture • • • Same throughout. Does not vary in composition from one region to another. Examples – – – Air Brass (solid solution of copper and zinc) Salt stirred into water Return to TOC 22

Section 1. 3 The Classification of Matter Heterogeneous Mixture • • • Having visibly distinguishable parts. Contains regions that have different properties from those of other regions. Examples – – Oil and vinegar dressing Sand stirred into water Return to TOC 23

Section 1. 3 The Classification of Matter Concept Check For each substance indicate if it is a pure substance or a mixture. For each pure substance indicate whether element or compound. For each mixture indicate homogeneous or heterogeneous. § Distilled Water § Filtered Sea water § Oxygen gas Return to TOC 24

Section 1. 3 The Classification of Matter Compound vs. Mixture Water H 2 O is a compound made up of hydrogen and oxygen constant composition Air is a mixture of gases including hydrogen (H 2) and oxygen (O 2) variable composition Return to TOC 25

Section 1. 3 The Classification of Matter Separating Mixtures • Mixtures can be separated into individual components because the different components have different physical and chemical properties • For example sand water can be separated by decanting – carefully pouring off the water into another container Return to TOC 26

Section 1. 3 The Classification of Matter Separating Mixtures • Distillation – Method used to separate components of a homogeneous mixture Return to TOC 27
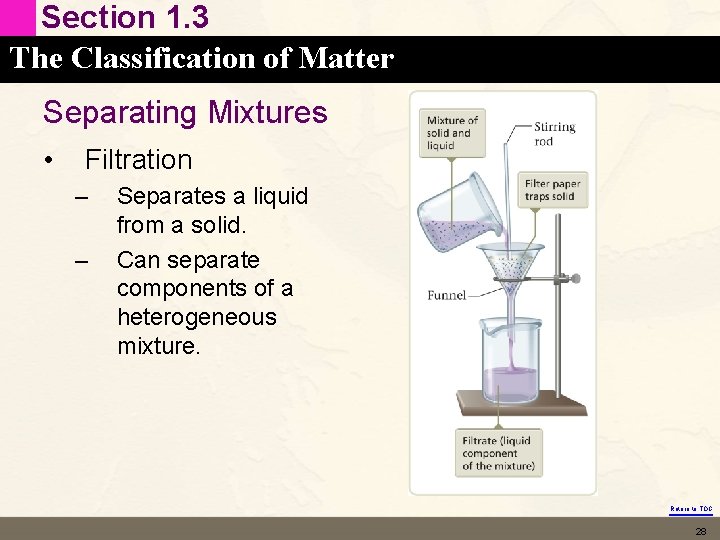
Section 1. 3 The Classification of Matter Separating Mixtures • Filtration – – Separates a liquid from a solid. Can separate components of a heterogeneous mixture. Return to TOC 28

Section 1. 3 The Classification of Matter Return to TOC 29
Section 1. 4 Physical and Chemical Changes Separation of Matter Return to TOC 30
Section 1. 4 Physical and Chemical Changes Physical Changes • Change in the form of a substance, not in its chemical composition. • The atoms or molecules do not change their identity only their state • These changes are reversible – Usually with temperature • Example: § Boiling or freezing water Return to TOC 31

Section 1. 4 Physical and Chemical Changes Boiling Water is a physical change • • • In both states, water molecules are still intact. Motions of molecules and the distances between them change. Reversible with temperature Fig 1. 7 Return to TOC 32

Section 1. 4 Physical and Chemical Changes Chemical Change • A given substance becomes a new substance or substances with different properties and different composition. – There is chemistry that made this change happen – Only reversible with more chemistry • Example: § Rusting of Iron Return to TOC 33

Section 1. 4 Physical and Chemical Changes Rusting Iron is a Chemical Change • • When iron rusts, the iron atoms combine with oxygen atoms to form a different chemical substance Not reversible except with more chemistry Fig 1. 8 Return to TOC 34

Section 1. 4 Physical and Chemical Changes Physical vs Chemical Changes • Dry Ice Sublimating – Physical • Sugar Dissolving – Physical • Propane Burning – Chemical Return to TOC 35

Section 1. 4 Physical and Chemical Changes Physical vs Chemical Change • Boiling Water • H 2 O(l) H 2 O(g) • Water molecules still intact • Electrolysis of Water • 2 H 2 O(l) 2 H 2(g) + O 2(g) • Water molecules split apart into hydrogen and oxygen molecules Return to TOC 36

Section 1. 4 Physical and Chemical Changes Concept Check The box on the left represents liquid water. Which diagram a, b or c represents water molecules after they have been vaporized by boiling? Return to TOC 37

Section 1. 4 Physical and Chemical Changes Concept Check What do the remaining boxes represent? Return to TOC 38
Section 1. 4 Physical and Chemical Changes Concept Check Classify each of the following as a physical or chemical change. § § Dissolving sugar in water Digesting sugar Chopping wood Burning wood Return to TOC 39

Section 1. 4 Physical and Chemical Properties • Physical and Chemical Changes are reflections of physical and chemical properties • Physical Property – Property a substance displays without changing its composition • Chemical Property – Property a substance displays only by changing its composition via a chemical change Return to TOC 40

Section 1. 4 Physical and Chemical Changes Physical vs Chemical Properties • What is an example • Lets use gasoline and hydrochloric acid • Physical Properties – Smell of gasoline – Boiling point of hydrochloric acid • Chemical Property – Flammability of gasoline – Corrosiveness of hydrochloric acid Return to TOC 41

Section 1. 4 Physical and Chemical Changes Physical vs Chemical Properties • Physical Properties – Odor, taste, color, appearance, melting point, boiling point, density • Chemical Properties – Corrosiveness, flammability, acidity, toxicity, reactivity Return to TOC 42

Section 1. 5 Energy: A Fundamental Part of Physical and Chemical Change Energy Changes • Changes in energy are a part of physical and chemical changes. • Water boiling (physical change) – Requires the input of energy (as heat) • Iron rusting (chemical change) – Releases energy (as heat) Return to TOC 43

Section 1. 5 Energy: A Fundamental Part of Physical and Chemical Change Definition of Energy • Scientific Definition of Energy is: • The capacity to do work – Energy from food does work in our bodies to keep us alive and move us around – Energy from fuel does work when it moves a car – Some forms of work are more subtle and we will see them in later chapters Return to TOC 44

Section 1. 5 Energy: A Fundamental Part of Physical and Chemical Change Total Energy of an Object • Potential Energy (Ep) – Energy associated with the objects position or composition • Kinetic Energy (Ek) – Energy associated with the objects motion • At first we are going to talk about Energy and Energy conversion from the point of view of physics but it will connect back to chemistry Return to TOC 45

Section 1. 5 Energy: A Fundamental Part of Physical and Chemical Change Energy Interconversion • Weight on a high shelf – Hi potential energy due to its position • Push the weight off the shelf – Weight falls and as it accelerates Ep of position is converted to Ek of motion • Weight hits the ground – Ek of motion is converted to thermal energy – Thermal energy is a type of Ek because the energy of motion of the object is converted to energy of motion in the atoms and molecules of the ground, Return to TOC 46

Section 1. 5 Energy: A Fundamental Part of Physical and Chemical Change Law of Conservation of Energy • Energy is neither Created Nor Destroyed – Converted from one form to another – Energy can change from Ep to Ek – Total amount of energy remains constant Return to TOC 47

Section 1. 5 Energy: A Fundamental Part of Physical and Chemical Change Nature does not like Instability Fig 1. 10 • Systems with high potential energy tend to change in a way that lowers their potential energy • From unstable to stable Return to TOC 48

Section 1. 5 Energy: A Fundamental Part of Physical and Chemical Change Energy in Chemistry • Some chemical substances have high Ep – – Energy is concentrated in these substances These substances react in order to lower their Ep To become more stable Gasoline is a good example Return to TOC 49

Section 1. 5 Energy: A Fundamental Part of Physical and Chemical Change Energy in Chemistry • Chemical potential energy • Reactions tend to proceed in a direction that decreases Ep and increases stability. Molecules in gasoline Hi Ep Unstable Molecules in exhaust Lo Ep Stable Return to TOC 50

Section 1. 5 Energy: A Fundamental Part of Physical and Chemical Change Energy in Chemistry • What is chemical potential energy – Potential energy from position of a weight is fairly easy to imagine – Chemical potential energy arises from the forces between the charged particles that make up atoms and molecules – It is their positions relative to each other that give some substances more potential energy than others – So chemical potential energy is energy of position as well. Return to TOC 51

Section 1. 6 The Units of Measurement The Fundamental SI Units Return to TOC 52
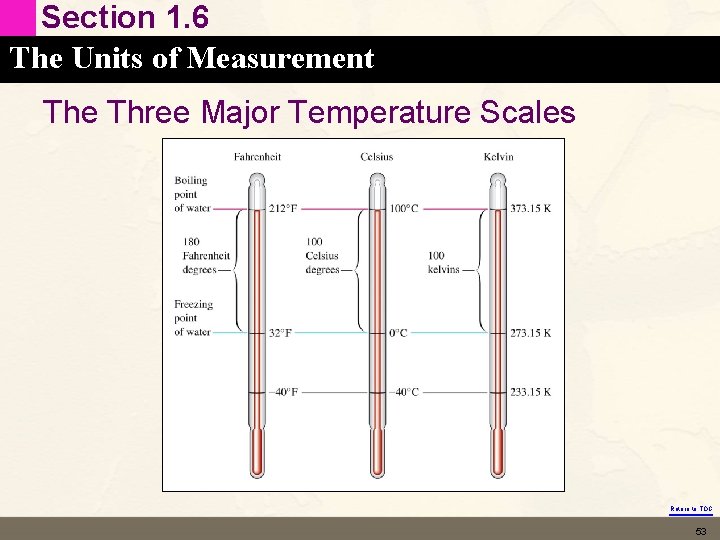
Section 1. 6 The Units of Measurement The Three Major Temperature Scales Return to TOC 53

Section 1. 6 The Units of Measurement The Kelvin: A Measure of Temperature • Called the absolute temperature scale – Lowest possible temperature is 0 K (where all molecular motion stops) – We will see where this definition comes from in Chapter 5 Return to TOC 54

Section 1. 6 The Units of Measurement Kelvin to Celsius Conversion • Kelvin scale has 100 units between the freezing and • boiling points of water (just like Celsius scale) 100 K = 100 C or 1 K = 1 C • But they have a different zero point. K is obtained by adding 273. 15 (measured value) to the Celsius temperature (correction for the zero point) • K = °C + 273. 15 Return to TOC 55

Section 1. 6 The Units of Measurement Learning Check • What is normal body temperature of 37. 0 C in Kelvin? Return to TOC 56

Section 1. 6 The Units of Measurement Fahrenheit – Celsius Conversions • On the Fahrenheit scale, there are 180 F between the freezing and boiling points; on the Celsius scale there are 100 C. So we need a correction factor for the size of the degree on the two different scales. Return to TOC 57

Section 1. 6 The Units of Measurement Fahrenheit – Celsius Conversions • • In the formula for calculating the Fahrenheit temperature, adding 32 adjusts the zero point of water from 0 C to 32 F. In the formula for calculating the Celsius temperature, we subtract 32 to adjust the zero point of water from 32 to 0 C then use the correction factor to the size of the degree Return to TOC 58
Section 1. 6 The Units of Measurement Learning Check The normal body temperature for a dog is 102 o F. What is this equivalent to on the Kelvin temperature scale? Return to TOC 59

Section 1. 6 The Units of Measurement Converting Between Temperature Scales K = °C + 273. 15 °C = K – 273. 15 Return to TOC 60

Section 1. 6 The Units of Measurement Prefix Multipliers • Unit sizes are often inconveniently large or small, so they can be modified using prefixes to refer to smaller or larger quantities. Return to TOC 61

Section 1. 6 The Units of Measurement Know the prefix and symbol You are only responsible for the metric prefixes from mega to nano. Return to TOC 62

Section 1. 6 The Units of Measurement Derived Units • Volume Fig 1. 13 – Any unit of length cubed becomes a unit of volume – m 3, cm 3 etc – More common metric unit is the liter (L) = 10 cm 3 – 1 L = 1000 cm 3 – or milliliter (m. L) – 1 ml = 1 cm 3 (know this) Return to TOC 63
Section 1. 6 The Units of Measurement Derived Units • Density – ratio of mass to volume • Typically g/m. L or g/cm 3 – The SI unit is kg/m 3 but you will rarely see this Return to TOC 64

Section 1. 6 The Units of Measurement Derived Units • Intensive vs Extensive • Properties of matter that are independent of amount are referred to as intensive – Density – Concentration • Properties of matter that depend on amount are referred to as extensive – Mass – Volume Return to TOC 65

Section 1. 6 The Units of Measurement Calculations with Density A certain mineral has a mass of 17. 8 g and a volume of 2. 35 cm 3. What is the density of this mineral? Return to TOC 66
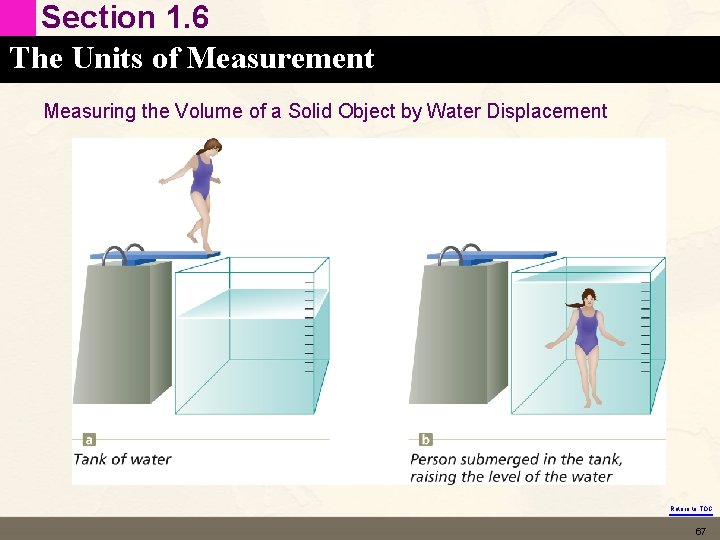
Section 1. 6 The Units of Measurement Measuring the Volume of a Solid Object by Water Displacement Return to TOC 67

Section 1. 6 The Units of Measurement Learning Check If 75. 0 g of copper is added to 50. 0 m. L of water in a graduated cylinder, the water level rises to 58. 4 m. L. What is the density of copper in g/cm 3? (1 m. L = 1 cm 3) Return to TOC 68

Section 1. 6 The Units of Measurement Density as a conversion factor Ethanol has a density of 0. 789 g/cm 3. What is the mass of a 25. 0 m. L sample of ethanol? Return to TOC 69

Section 1. 6 The Units of Measurement Learning Check What is the volume (in m. L) of a sample of ethanol that has a mass of 125 g? Return to TOC 70

Section 1. 7 The Reliability of a Measurement Certain and Uncertain Digits • The volume is read at the bottom of the liquid curve (meniscus). • Meniscus of the liquid occurs at about 20. 15 m. L. § Certain digits: 20. 15 § Uncertain digit: 20. 15 Return to TOC 71

Section 1. 7 The Reliability of a Measurement Certain and Uncertain Digits • Below are two measurements of the mass of the same object. The same quantity is being described at two different levels of precision or certainty Return to TOC 72

Section 1. 7 The Reliability of a Measurement Significant Figures • To record a measured number – observe the numerical values of marked lines – estimate the value of number between marks • The recorded value is all the numbers you observe plus the value you estimate • All the numbers recorded in these readings are called the significant figures in a measurement • They are significant because they give you information about the measurement. Return to TOC 73

Section 1. 7 The Reliability of a Measurement Concept Check What is the length of the wooden dowel with each measuring device? Don’t forget the Estimated value! Return to TOC 74

Section 1. 7 The Reliability of a Measurement Significant Figures • All of the digits we record in a measured number (including the estimated digit) are called significant digits. • Significant means that the numbers are meaningful – that they give information about the measurement – Even the uncertain or estimated value – No further (insignificant digits) should be recorded Return to TOC 75

Section 1. 7 The Reliability of a Measurement Exact Numbers • • In contrast to recorded measurements there are defined or exact values (1 foot = 12 inches) or counted items (15 puppies). Significant figures do not apply here because these numbers are not measured Return to TOC 76

Section 1. 7 The Reliability of a Measurement Rules for Counting Significant Figures 1. All non zero digits are significant § 3456 has 4 sig figs (significant figures). 2. There are three classes of zeros. a. Captive zeros are significant § 16. 07 has 4 sig figs. b. Leading zeros are not significant. § 0. 048 has 2 sig figs. c. Trailing zeros are significant only if the number contains a decimal point. § 9. 300 has 4 sig figs. 150 has 2 sig figs. Return to TOC 77

Section 1. 7 The Reliability of a Measurement Practice Worksheet 1 Return to TOC 78

Section 1. 7 The Reliability of a Measurement Rounding • • • Digit to be removed is less than 5 – drop all digits Digit to be removed is greater than 5 – round up Digit to be removed is equal to 5 – – § § Digit before 5 is even - drop the 5 and all digits after Digit before 5 is odd round that digit up Round 5. 68 to 2 SF rounds to 5. 7 Round 3226 to 2 SF 3200 NOT 32 Round 4355 to 2 SF 4400 (digit before 5 is odd) Round 2250 to 2 SF 2200 (digit before 5 is even) Return to TOC 79

Section 1. 7 The Reliability of a Measurement Exponential Notation • Example § § • 300. written as 3. 00 × 102 Contains three significant figures. Two Advantages § § Number of significant figures can be easily indicated. Fewer zeros are needed to write a very large or very small number. Return to TOC 80

Section 1. 7 The Reliability of a Measurement Concept Check Round each of the following values to 2 SF and write the answer in standard and scientific notation. 1. 13505 g 1. 0. 000460 g 2. 1501 g Return to TOC 81
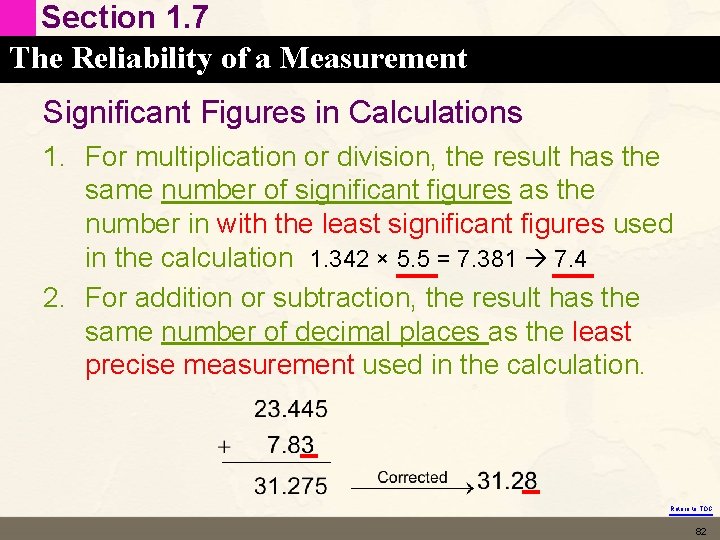
Section 1. 7 The Reliability of a Measurement Significant Figures in Calculations 1. For multiplication or division, the result has the same number of significant figures as the number in with the least significant figures used in the calculation 1. 342 × 5. 5 = 7. 381 7. 4 2. For addition or subtraction, the result has the same number of decimal places as the least precise measurement used in the calculation. Return to TOC 82

Section 1. 7 The Reliability of a Measurement Practice Worksheet 1 Return to TOC 83

Section 1. 7 The Reliability of a Measurement Precision vs Accuracy • These are two ways to characterize the reliability of a measurement • Accuracy -Agreement of a particular value with the true value • Precision - Degree of agreement among several measurements of the same quantity. − Reflects the reproducibility of a measurement • Differences in precision and accuracy reflect different types of error that can occur when taking measurements Return to TOC 84
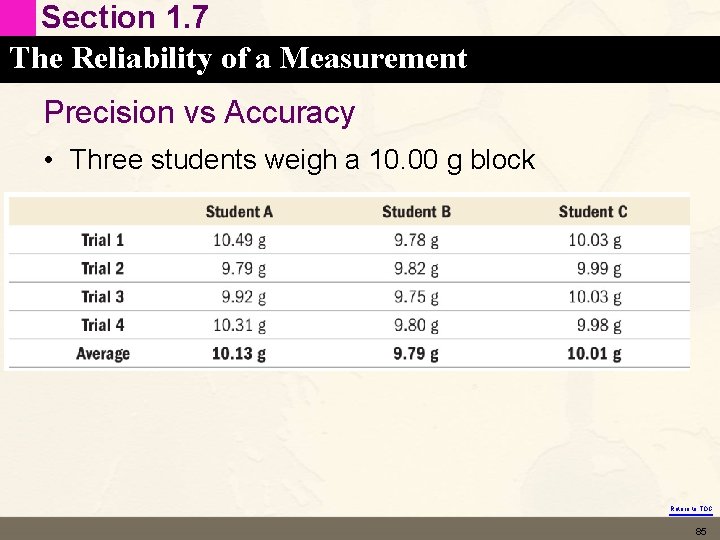
Section 1. 7 The Reliability of a Measurement Precision vs Accuracy • Three students weigh a 10. 00 g block Return to TOC 85

Section 1. 7 The Reliability of a Measurement Precision vs Accuracy • Three students weigh a 10. 00 g block • Student A is not accurate (far from true value) and not precise (readings are all over the place) – This is due to random error – Notice random error averages out because it has an equal probability of being to high or too low Return to TOC 86

Section 1. 7 The Reliability of a Measurement Precision vs Accuracy • Student B is precise (reproducible) but not accurate (far from true value) – This is probably due to a systematic error – Notice systematic error tends to be all in one direction Return to TOC 87

Section 1. 7 The Reliability of a Measurement Random vs Systematic Error • Random error averages out because it has an equal probability of being to high or too low – Random error usually has to due with faulty technique – Scientist is sloppy or careless or untrained • Systematic error tends to be all in one direction – Systematic error usually arises from faulty equipment – Broken or faulty balance for example Return to TOC 88
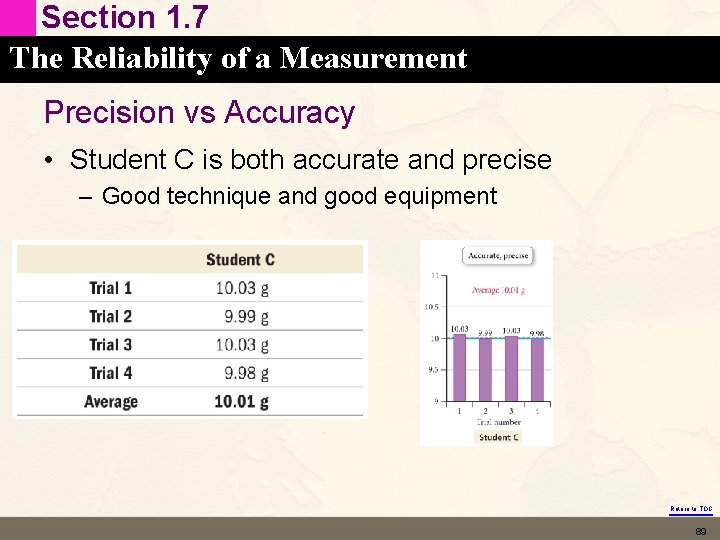
Section 1. 7 The Reliability of a Measurement Precision vs Accuracy • Student C is both accurate and precise – Good technique and good equipment Return to TOC 89

Section 1. 7 The Reliability of a Measurement Precision • • Precision (actually used 2 ways) Agreement between several measurements (when talking about sources of error) Also the number of digits used to express a single value • The more digits you use the more precise the value is • The balance is more precise than the bathroom scale Bathroom Scale Balance Grapefruit 1 1. 5 lb 1. 476 lb Grapefruit 2 1. 5 lb 1. 518 lb Return to TOC 90

Section 1. 7 The Reliability of a Measurement Precision • • • The word precision can be misleading Weighing Result 1 2. 486 g 2 2. 487 g 3 2. 485 g 4 2. 484 g 5 2. 488 g These measurements have both types of precision. They have multiple decimal places and they agree well with each other Is this measurement necessarily accurate though? Return to TOC 91

Section 1. 8 Solving Chemical Problems Return to TOC 92

Section 1. 8 Solving Chemical Problems Types of Problems • Unit conversion problems – Dimensional Analysis is your friend! • Equation problems – Plug and Chug – We actually already did some of these when doing density calculations • Combinations of the above Return to TOC 93

Section 1. 8 Solving Chemical Problems Is there a trick to solving problems? • Yes • You have to learn the method or strategy • You have to go slow – don’t panic • You have to identify where you are starting and where you are going – Just like a map app! Return to TOC 94

Section 1. 8 Solving Chemical Problems Steps in Dimensional Analysis • • Write down what you have (left) Write down what you want (right) Use conversion factors Cancel your units Write in your numbers Do your math done Return to TOC 95

Section 1. 8 Solving Chemical Problems Example #1 An iron sample has a mass of 4. 50 lb. What is the mass of this sample in grams? Have 4. 50 lbs Want grams Return to TOC 96

Section 1. 8 Solving Chemical Problems Example #2 An sample of ethanol has a volume of 0. 0500 L. What is the mass of this sample in grams? The density of ethanol is 0. 789 g/cm 3. Have 0. 0500 L Want grams Return to TOC 97
Section 1. 8 Solving Chemical Problems Learning Check Perform the following metric conversions using dimensional analysis and conversion factors. 1. How many grams are in 2. 55 kg? 2. How many cm are in 25 km? 3. How many kg are in 35. 0 L of ethanol? Return to TOC 98

Section 1. 8 Solving Chemical Problems Homework and Review • Links Chapter 1 Module • Chapter 1 Review Questions 4, 11 -14, 16, 19, 25 -28, 30, 31 • Odd numbered problems 33 -89 (skip 35, 53, 59, 63, 67 and 87) • Sapling HW – Try do do the Sapling HW without your notes to determine what topics you still need to study. Return to TOC 99
- Slides: 99