CHEMISTRY STRUCTURE OF MATTER THE STRUCTURE OF MATTER
























- Slides: 31

CHEMISTRY: STRUCTURE OF MATTER

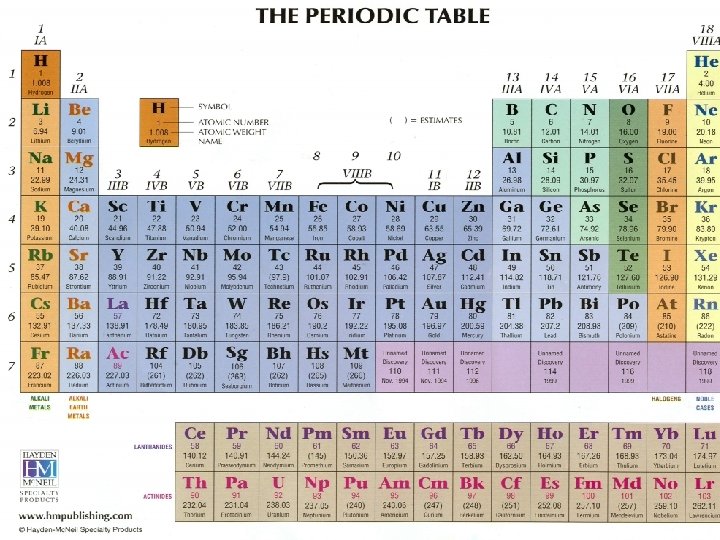
THE STRUCTURE OF MATTER • What is matter? –Matter is anything that takes up space and has mass • All matter is made up of atoms • Matter made up of only one kind of atom is called an element • Elements can be found on the periodic table

ELEMENT • What is an element? • A substance that is made up of only one type of atom • Cannot be broken down into other substances chemically • EX: gold, silver, iron

EX: GOLD

COMPOUND • Made up of two or more different types of atoms that are combined. • EX: water • Water is made of 2 atom of hydrogen 1 atoms of oxygen

SUBSTANCES • A substance is made up of two or more atoms. • Can be an element • Can be a compound.


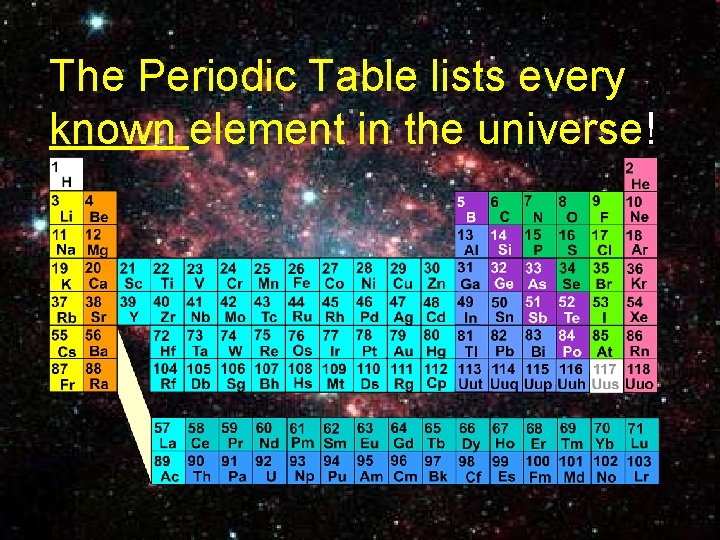
The Periodic Table lists every known element in the universe! 8


Is this substance an element? No, because it is made of more than 1 kind of atom. 10

Is it an element now? No, because it is made of more than 1 kind of atom. 11

Is it an element now? Yes! Because it is made of only one kind of atom. 12

Gold is made of only gold atoms. Is gold an element? YES! http: //thesimplefrontporch. files. wordpress. com/2009/04/gold-nugget. jpg 13

Water is made up of two kinds of atoms bonded together. Is water an element? NO! 14

Water is NOT an element! It is made of more than one kind of atom. Water is a molecule of hydrogen and oxygen atoms bonded together. 15

ATOMS VS. COMPOUNDS • What is the smallest particle an element could be divided into and still retain the properties of that element? an ATOM • Atoms make up molecules, which is defined as two or more atoms bonded together. • for example: CO 2 and H 2 O are compounds.

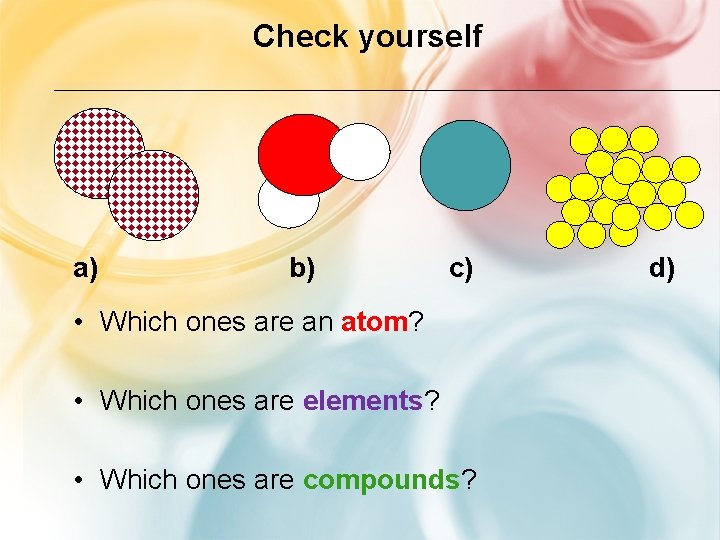
Check yourself a) b) c) • Which ones are an atom? • Which ones are elements? • Which ones are compounds? d)

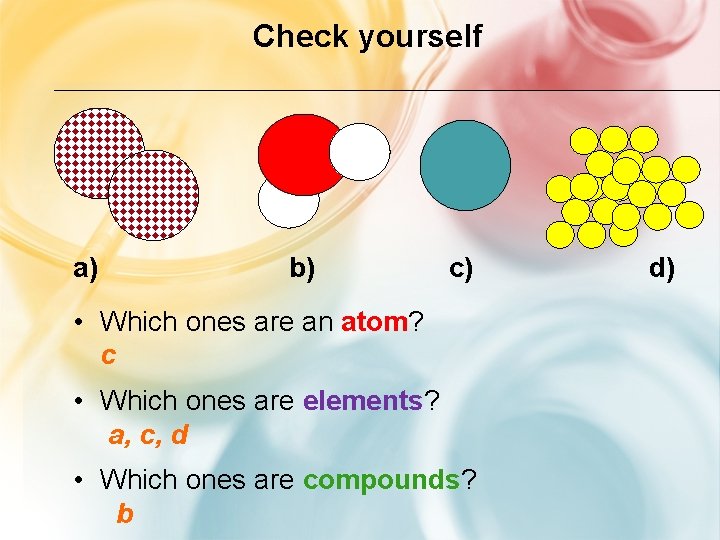
Check yourself a) b) c) • Which ones are an atom? c • Which ones are elements? a, c, d • Which ones are compounds? b d)

MASS AND WEIGHT? • Mass is a measure of how much matter an object has • Weight is a downward force due to gravity • On the surface of Earth they are directly related: Weight (N) = Mass (kg) x 10

MIXTURES • Two or more substances are called a mixture • Homogeneous - all the same • Heterogeneous – different substances • Brain. Pop: Compounds and mixtures • https: //www. brainpop. com/science/matter andchemistry/compoundsandmixtures/

MIXTURES • Homogeneous : - Materials that make up the mixture is uniformly spread out. • Particles will never settle , not easily seen. Ex, salty water mixture. • Also called a solution

MIXTURES • Heterogeneous : - Materials that make up the mixture is easily distinguished. • Parts can easily be seen with or without a microscope. Ex: pizza

TRICKY MIXTURES • Solution: very small particles; light can pass through • Colloid: larger particles, light gets scattered – they appear murky or opaque. Examples: milk and fog • Suspension: Even larger particles, light cannot get through and they appear very murky. They will eventually separate like oil & water, and can be filtered to separate compounds. Examples: salad dressings, blood, and aerosol sprays

NOTES SUMMARY: • What is the difference between a compound a mixture?

BASIC CHEMISTRY Everything is made up of these 2 things: Matter and Energy • Matter – has mass and occupies space • Energy – ability to do work

MATTER • Can change forms • It can be a liquid, solid, or gas

BASIC CHEMISTRY Energy can change forms: Solar cells change light to electrical energy, Steam Engine Heat to Motion (Kinetic Energy) UP Steam Engine

ENERGY • Cells convert (food) chemical energy to cellular energy.

PRINCIPLE OF CONSERVATION OF MASS • Mass cannot be created nor destroyed. It just changes forms. • Example: burning a log • After it has burned, the log is lighter. What happened? • Some of the mass was converted from solid to gas.

PRINCIPLE OF CONSERVATION OF ENERGY • Energy cannot be created or destroyed – Applies only to normal chemical reactions, NOT nuclear reactions. • In nuclear reactions, energy and mass can be destroyed or created. E=MC 2 E is energy, M is mass, C is the speed of light MASS IS CHANGED INTO ENERGY in nuclear reactions

SIDE NOTE: THE NATURE OF SCIENCE • These last 2 principles used to be referred to as “Laws” – meaning it was thought to be an absolute fact. • Nuclear theory blew that idea out of the water – literally. • This is why it is important for scientists to be cautious about their conclusions. They say “This is our current belief on a specific principle, based upon the evidence collect so far”. • It’s why we say “our hypothesis was supported” instead of “right”