chemistry Slide 1 of 37 11 1 Describing

chemistry Slide 1 of 37

11. 1 Describing Chemical Reactions On May 6, 1937, the huge airship Hindenburg erupted into a fireball. Within a short time, 210, 000 cubic meters of hydrogen had burned and the airship was destroyed. The chemical reaction that occurred is “hydrogen combines with oxygen to produce water. ” You will learn to represent this chemical reaction by a chemical equation. Slide 2 of 37 © Copyright Pearson Prentice Hall

11. 1 Describing Chemical Reactions > Writing Chemical Equations How do you write a word equation? Slide 3 of 37 © Copyright Pearson Prentice Hall

11. 1 Describing Chemical Reactions > Writing Chemical Equations Word Equations To write a word equation, write the names of the reactants to the left of the arrow separated by plus signs; write the names of the products to the right of the arrow, also separated by plus signs. Reactant + Reactant Product + Product Slide 4 of 37 © Copyright Pearson Prentice Hall

11. 1 Describing Chemical Reactions > Writing Chemical Equations Methane + Oxygen Carbon dioxide + Water Slide 5 of 37 © Copyright Pearson Prentice Hall

11. 1 Describing Chemical Reactions > Writing Chemical Equations iron + oxygen iron(III) oxide Slide 6 of 37 © Copyright Pearson Prentice Hall

11. 1 Describing Chemical Reactions > Writing Chemical Equations Hydrogen Peroxide Water and Oxygen Slide 7 of 37 © Copyright Pearson Prentice Hall

11. 1 Describing Chemical Reactions > Writing Chemical Equations A chemical equation is a representation of a chemical reaction; the formulas of the reactants (on the left) are connected by an arrow with the formulas of the products (on the right). Slide 8 of 37 © Copyright Pearson Prentice Hall

11. 1 Describing Chemical Reactions > Writing Chemical Equations How do you write a skeleton equation? Slide 9 of 37 © Copyright Pearson Prentice Hall

11. 1 Describing Chemical Reactions > Writing Chemical Equations A skeleton equation is a chemical equation that does not indicate the relative amounts of the reactants and products. Here is the equation for rusting: Fe + O 2 Fe 2 O 3 Slide 10 of 37 © Copyright Pearson Prentice Hall

11. 1 Describing Chemical Reactions > Writing Chemical Equations Write the formulas of the reactants to the left of the yields sign (arrow) and the formulas of the products to the right. Slide 11 of 37 © Copyright Pearson Prentice Hall

11. 1 Describing Chemical Reactions > Writing Chemical Equations A catalyst is a substance that speeds up the reaction but is not used up in the reaction. Without Catalyst With Catalyst Slide 12 of 37 © Copyright Pearson Prentice Hall

11. 1 Describing Chemical Reactions > Writing Chemical Equations Slide 13 of 37 © Copyright Pearson Prentice Hall

Describing Chemical Reactions > Writing Chemical Equations Animation 12 Relate chemical symbols and formulas to the information they communicate. Slide 14 of 37 © Copyright Pearson Prentice Hall

Slide 15 of 37 © Copyright Pearson Prentice Hall

Slide 16 of 37 © Copyright Pearson Prentice Hall

Slide 17 of 37 © Copyright Pearson Prentice Hall

Practice Problems for Conceptual Problem 11. 1 Problem Solving 11. 2 Solve Problem 2 with the help of an interactive guided tutorial. Slide 18 of 37 © Copyright Pearson Prentice Hall

11. 1 Describing Chemical Reactions > Balancing Chemical Equations What are the steps in writing a balanced chemical equation? Slide 19 of 37 © Copyright Pearson Prentice Hall

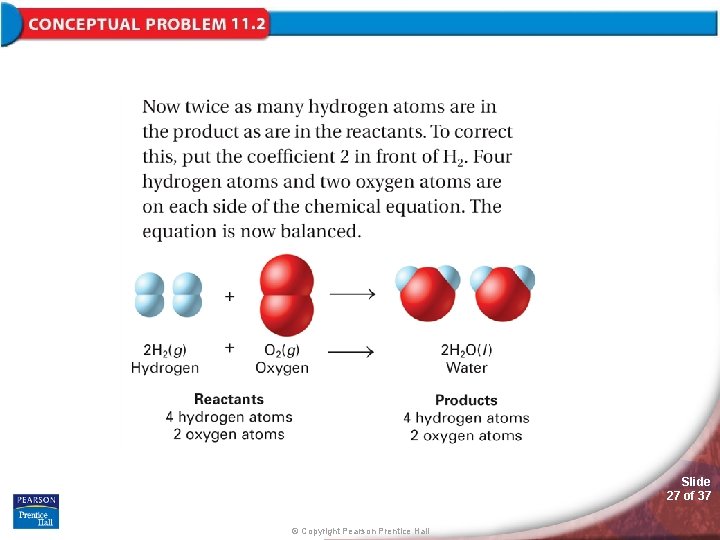
11. 1 Describing Chemical Reactions > Balancing Chemical Equations To write a balanced chemical equation, first write the skeleton equation. Then use coefficients to balance the equation so that it obeys the law of conservation of mass. Slide 20 of 37 © Copyright Pearson Prentice Hall

11. 1 Describing Chemical Reactions > Balancing Chemical Equations This is a balanced equation for making a bicycle. The numbers are called coefficients—small whole numbers that are placed in front of the formulas in an equation in order to balance it. Slide 21 of 37 © Copyright Pearson Prentice Hall

11. 1 Describing Chemical Reactions > Balancing Chemical Equations A chemical reaction is also described by a balanced equation in which each side of the equation has the same number of atoms of each element and mass is conserved. Slide 22 of 37 © Copyright Pearson Prentice Hall

Describing Chemical Reactions > Balancing Chemical Equations Simulation 11 Sharpen your skills by balancing chemical equations. Slide 23 of 37 © Copyright Pearson Prentice Hall

Slide 24 of 37 © Copyright Pearson Prentice Hall

Slide 25 of 37 © Copyright Pearson Prentice Hall

Slide 26 of 37 © Copyright Pearson Prentice Hall
Slide 27 of 37 © Copyright Pearson Prentice Hall

Practice Problems for Conceptual Problem 11. 2 Problem Solving 11. 4 Solve Problem 4 with the help of an interactive guided tutorial. Slide 28 of 37 © Copyright Pearson Prentice Hall

Slide 29 of 37 © Copyright Pearson Prentice Hall

Slide 30 of 37 © Copyright Pearson Prentice Hall

Slide 31 of 37 © Copyright Pearson Prentice Hall

Slide 32 of 37 © Copyright Pearson Prentice Hall

11. 1 Section Quiz. 1. Propane gas reacts with oxygen to produce water vapor and carbon dioxide. Choose the correct word equation for this reaction. a. propane + carbon dioxide water + oxygen b. propane + oxygen + water carbon dioxide c. propane + oxygen + water + carbon dioxide d. propane + oxygen water + carbon dioxide Slide 33 of 37 © Copyright Pearson Prentice Hall

11. 1 Section Quiz. 2. Which of the following is a skeleton equation? a. H 2 + CO CH 3 OH b. 2 H 2 + CO CH 3 OH c. 2 H 2 + CO 2 CH 3 OH d. hydrogen + carbon monoxide methanol Slide 34 of 37 © Copyright Pearson Prentice Hall

11. 1 Section Quiz. 3. What coefficient for H 2 SO 4 is required to balance the following equation? Ca 3(PO 4)2 + ____ H 2 SO 4 3 Ca. SO 4 + 2 H 3 PO 4 a. 1 b. 2 c. 3 d. 4 Slide 35 of 37 © Copyright Pearson Prentice Hall

END OF SHOW
- Slides: 36