Chemistry Second Edition Julia Burdge Lecture Powerpoints Jason

Chemistry Second Edition Julia Burdge Lecture Powerpoints Jason A. Kautz University of Nebraska-Lincoln 12 Intermolecular Forces and the Physical Properties of Liquids and Solids Copyright (c) The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display.

12 Intermolecular Forces and the Physical properties of Liquids and Solids 12. 1 Intermolecular Forces Dipole-Dipole Interactions Hydrogen Bonding Dispersion Forces Ion-Dipole Interactions 12. 2 Properties of Liquids Surface Tension Viscosity Vapor Pressure 12. 3 Crystal Structure Unit Cells Packing Spheres Closest Packing 12. 4 Types of Crystals Ionic Crystals Covalent Crystals Molecular Crystals Metallic Crystals 12. 5 Amorphous Solids 12. 6 Phase Changes Liquid-Vapor Phase Transition Solid-Liquid Phase Transition Solid-Vapor Phase Transition 12. 7 Phase Diagrams

12. 1 Intermolecular Forces Intermolecular forces are attractive forces that hold particles together in the condensed phases. The magnitude (and type) of intermolecular forces is what determines whether the particles that make up a substance are a gas, liquid, or solid. Gas Liquid Solid
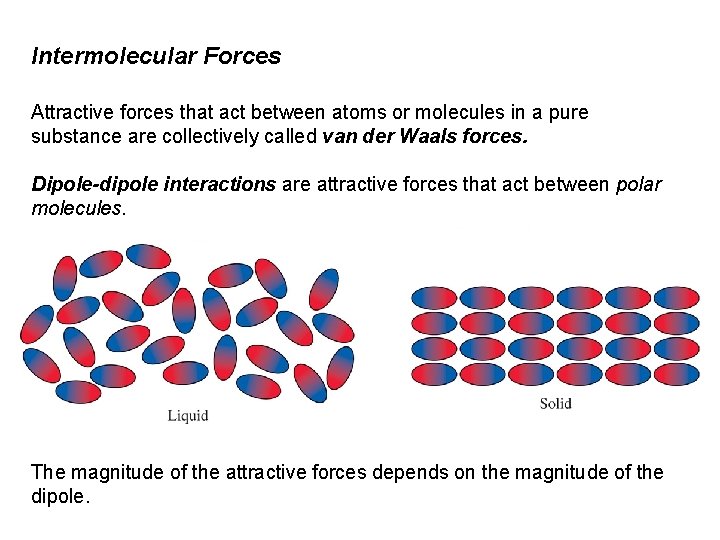
Intermolecular Forces Attractive forces that act between atoms or molecules in a pure substance are collectively called van der Waals forces. Dipole-dipole interactions are attractive forces that act between polar molecules. The magnitude of the attractive forces depends on the magnitude of the dipole.
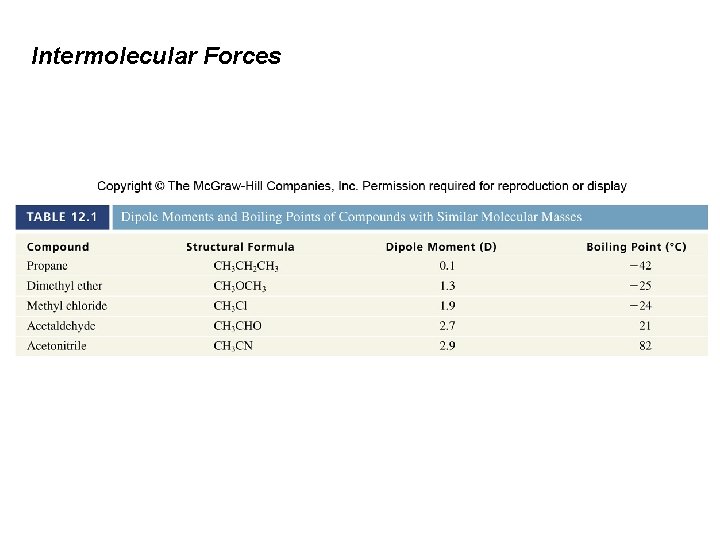
Intermolecular Forces
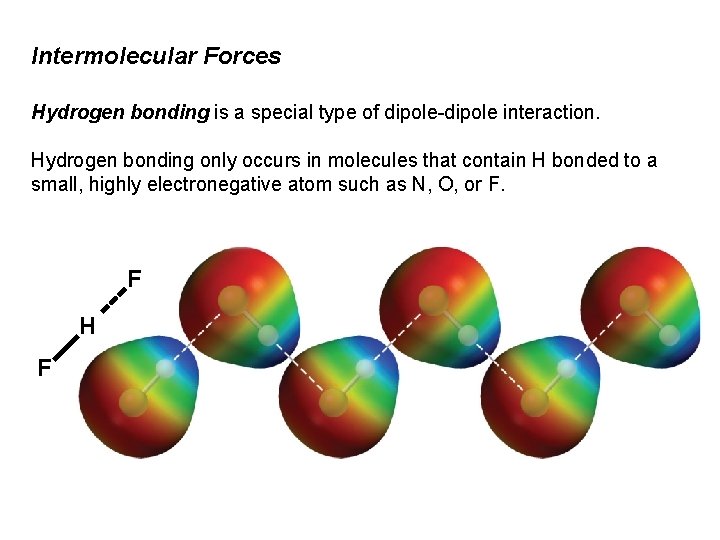
Intermolecular Forces Hydrogen bonding is a special type of dipole-dipole interaction. Hydrogen bonding only occurs in molecules that contain H bonded to a small, highly electronegative atom such as N, O, or F. F H F
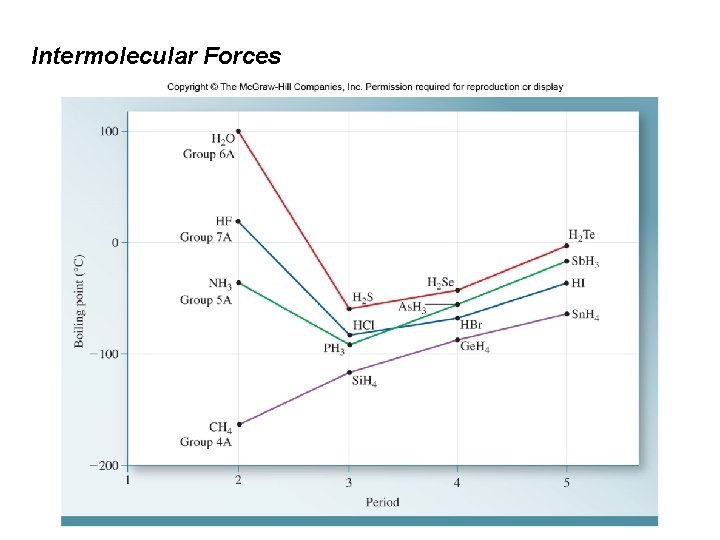
Intermolecular Forces
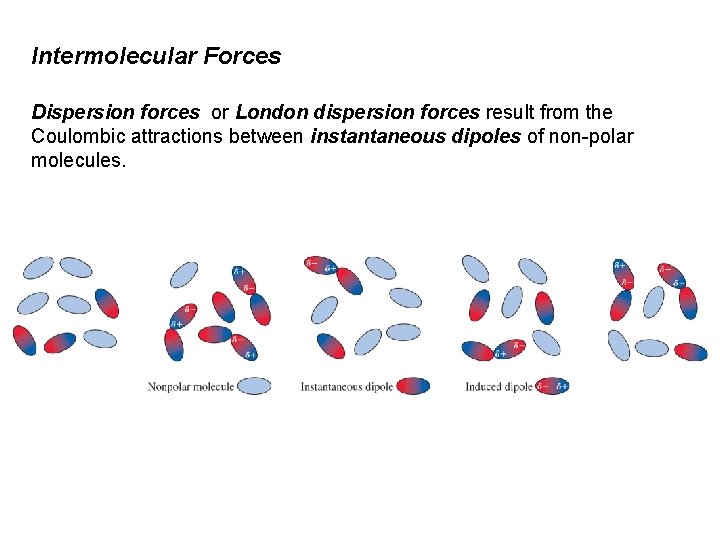
Intermolecular Forces Dispersion forces or London dispersion forces result from the Coulombic attractions between instantaneous dipoles of non-polar molecules.
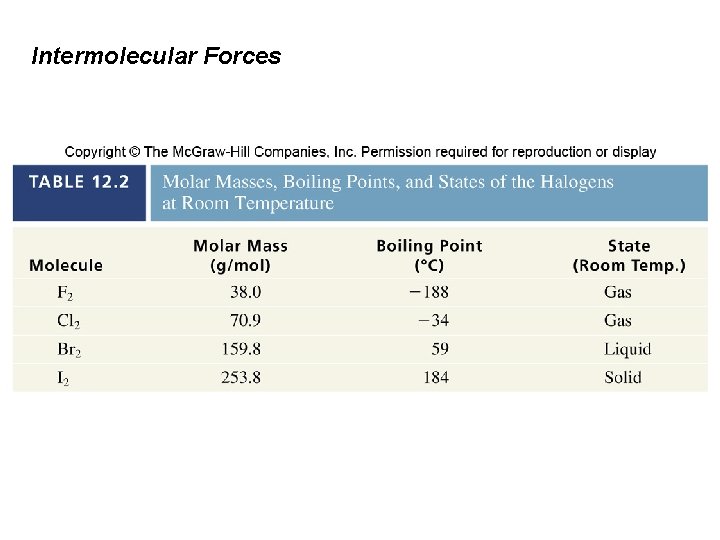
Intermolecular Forces

Intermolecular Forces What kinds of intermolecular forces exist in the following molecules: CH 3 CH 2 CH 2 CH 3 This compound is nonpolar; only dispersion forces CH 3 CH 2 OH This compound is polar and contains an O‒H bond. Dispersion, dipole-dipole, and hydrogen bonding
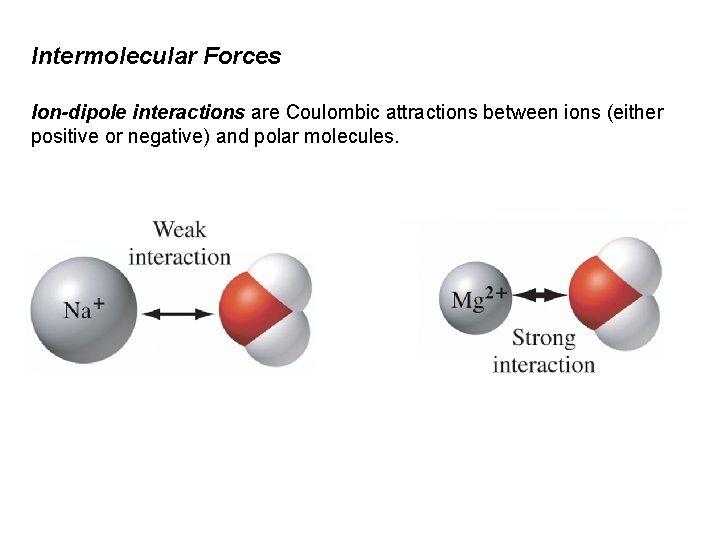
Intermolecular Forces Ion-dipole interactions are Coulombic attractions between ions (either positive or negative) and polar molecules.

12. 2 Properties of Liquids Surface tension is the amount of energy required to stretch or increase the surface of a liquid by a unit area. The stronger the intermolecular forces, the higher the surface tension.

Properties of Liquids Capillary action is the movement of a liquid up a narrow tube. Two types of forces bring about capillary action: cohesion is the attraction between like molecules adhesion is the attraction between unlike molecules Adhesive forces are greater than cohesive forces Cohesive forces are greater than adhesive forces

Properties of Liquids Viscosity is a measure of a fluid’s resistance to flow. The higher the viscosity the more slowly a liquid flows. Liquids that have higher intermolecular forces have higher viscosities.
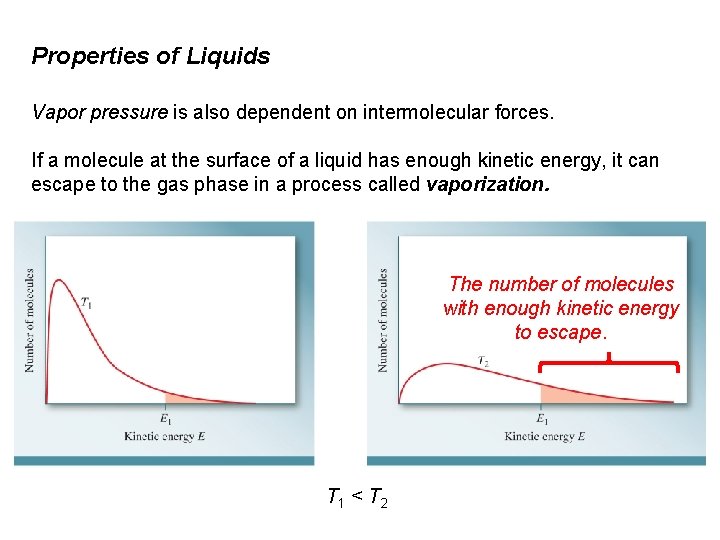
Properties of Liquids Vapor pressure is also dependent on intermolecular forces. If a molecule at the surface of a liquid has enough kinetic energy, it can escape to the gas phase in a process called vaporization. The number of molecules with enough kinetic energy to escape. T 1 < T 2

Properties of Liquids The vapor pressure increases until the rate of evaporation equals the rate of condensation. H 2 O(l) ⇌ H 2 O(g) Evaporation: H 2 O(l) → H 2 O(g) Condensation: H 2 O(l) ← H 2 O(g) When the forward process and reverse process are occurring at the same rate, the system is in dynamic equilibrium.
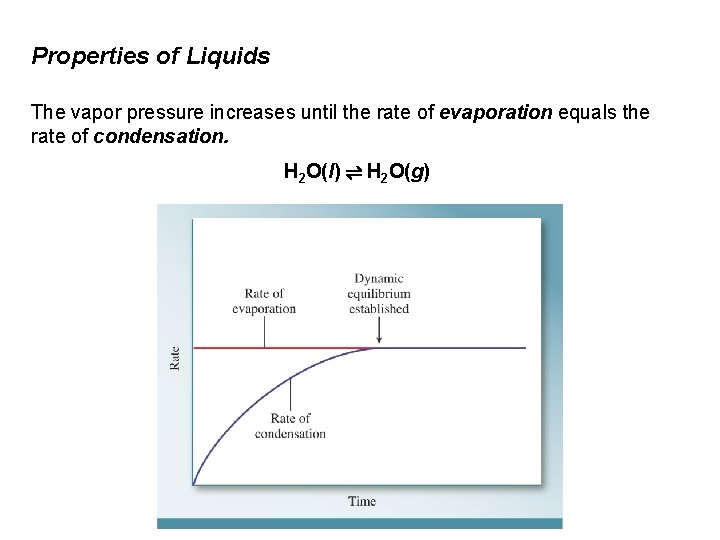
Properties of Liquids The vapor pressure increases until the rate of evaporation equals the rate of condensation. H 2 O(l) ⇌ H 2 O(g)
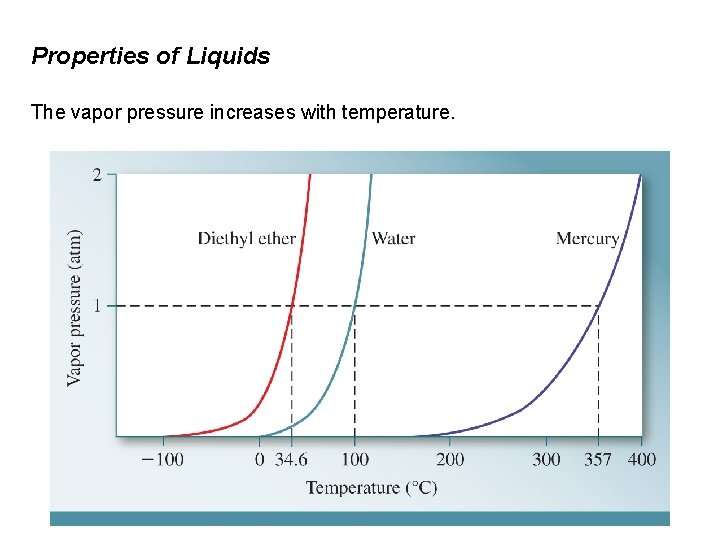
Properties of Liquids The vapor pressure increases with temperature.

Properties of Liquids The Clausius-Clapeyron equation relates the natural log of vapor pressure and the reciprocal of absolute temperature. ln P = natural log of vapor pressure ΔHvap = the molar heat of vaporization R = the gas constant (8. 314 J/K • mol) T = the kelvin temperature C is an experimentally determined constant
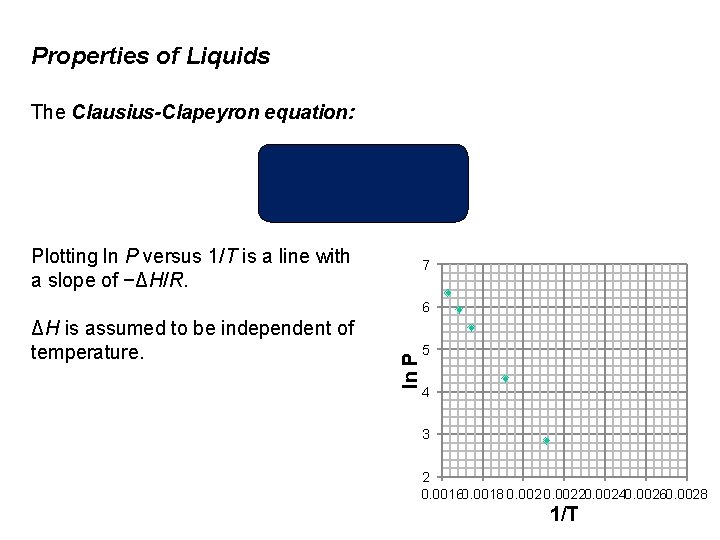
Properties of Liquids The Clausius-Clapeyron equation: Plotting ln P versus 1/T is a line with a slope of −ΔH/R. 7 ΔH is assumed to be independent of temperature. ln P 6 5 4 3 2 0. 00160. 0018 0. 00220. 00240. 00260. 0028 1/T

Properties of Liquids The Clausius-Clapeyron equation can be rearranged into a two point form:

Properties of Liquids The vapor pressure of ethanol is 1. 00 x 102 mm. Hg at 34. 9°C. What is its vapor pressure at 55. 8°C? (ΔHvap for ethanol is 39. 3 k. J/mol) Solution: Step 1: Given the vapor pressure at one temperature, P 1 use the equation below to calculate the vapor pressure at a second temperature. P 2 = 265 mm. Hg
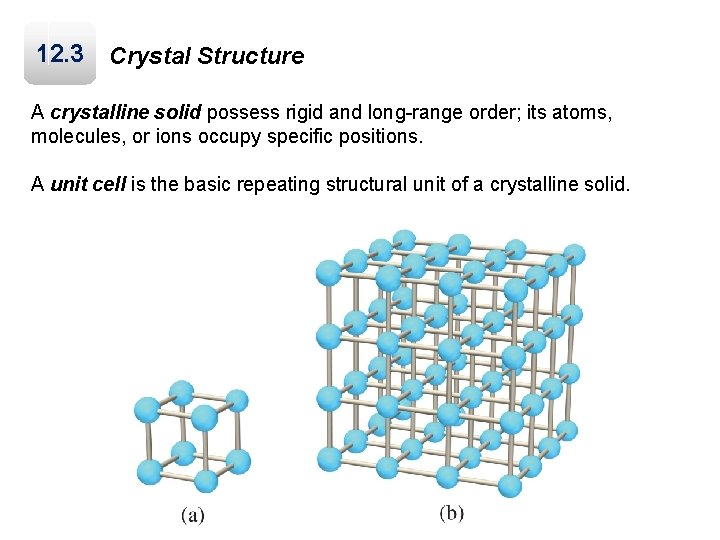
12. 3 Crystal Structure A crystalline solid possess rigid and long-range order; its atoms, molecules, or ions occupy specific positions. A unit cell is the basic repeating structural unit of a crystalline solid.
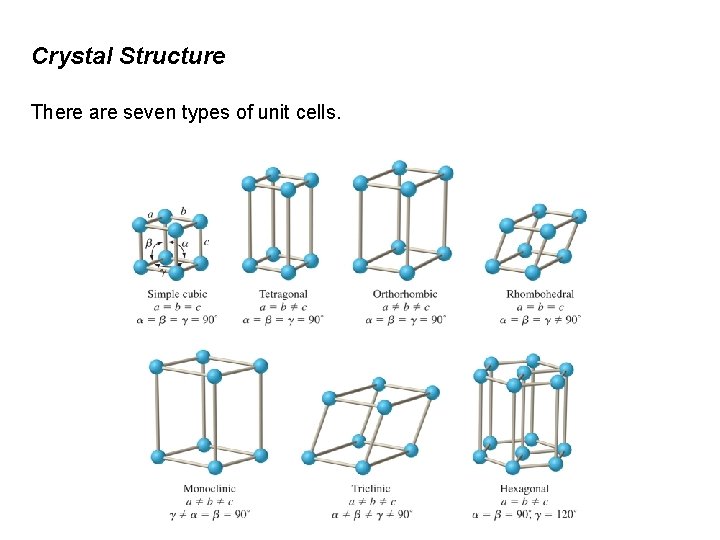
Crystal Structure There are seven types of unit cells.
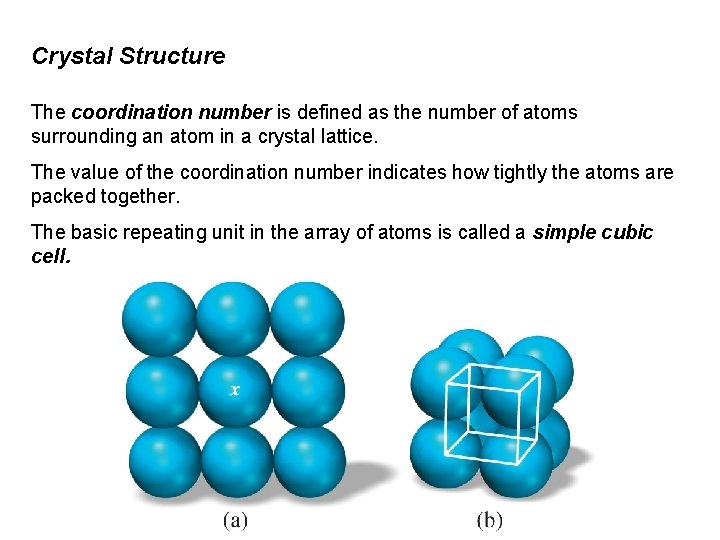
Crystal Structure The coordination number is defined as the number of atoms surrounding an atom in a crystal lattice. The value of the coordination number indicates how tightly the atoms are packed together. The basic repeating unit in the array of atoms is called a simple cubic cell.
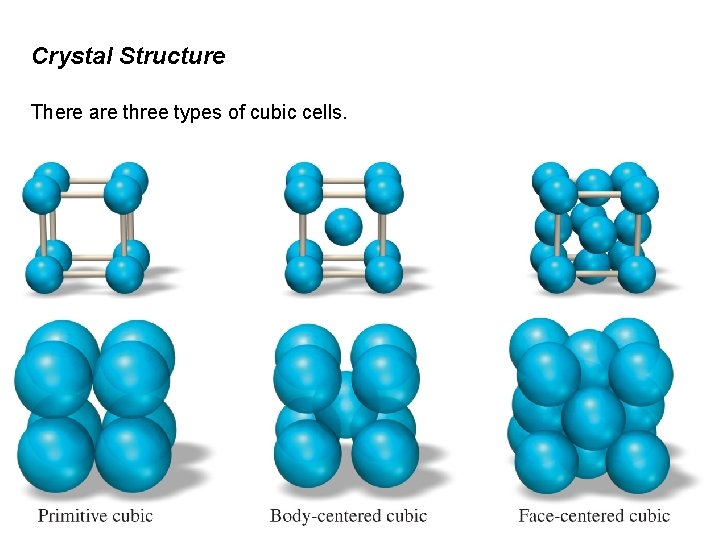
Crystal Structure There are three types of cubic cells.
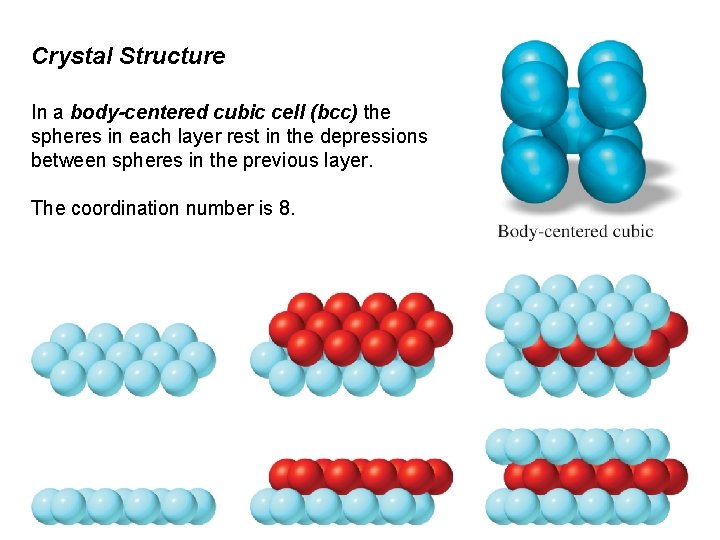
Crystal Structure In a body-centered cubic cell (bcc) the spheres in each layer rest in the depressions between spheres in the previous layer. The coordination number is 8.
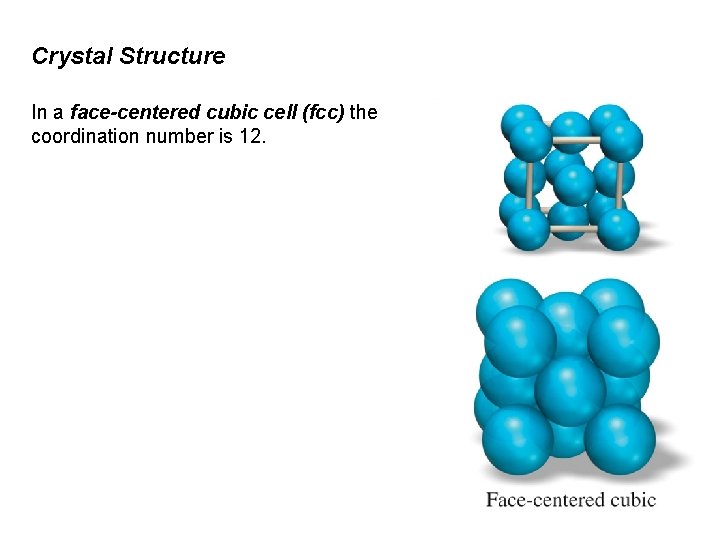
Crystal Structure In a face-centered cubic cell (fcc) the coordination number is 12.
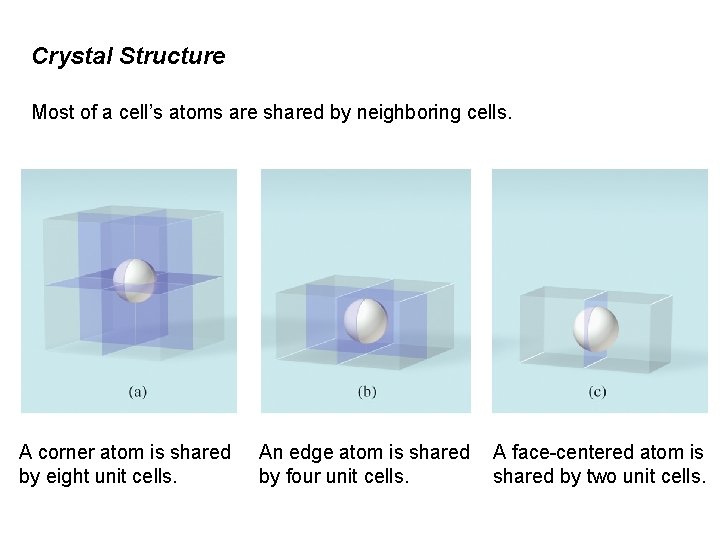
Crystal Structure Most of a cell’s atoms are shared by neighboring cells. A corner atom is shared by eight unit cells. An edge atom is shared by four unit cells. A face-centered atom is shared by two unit cells.
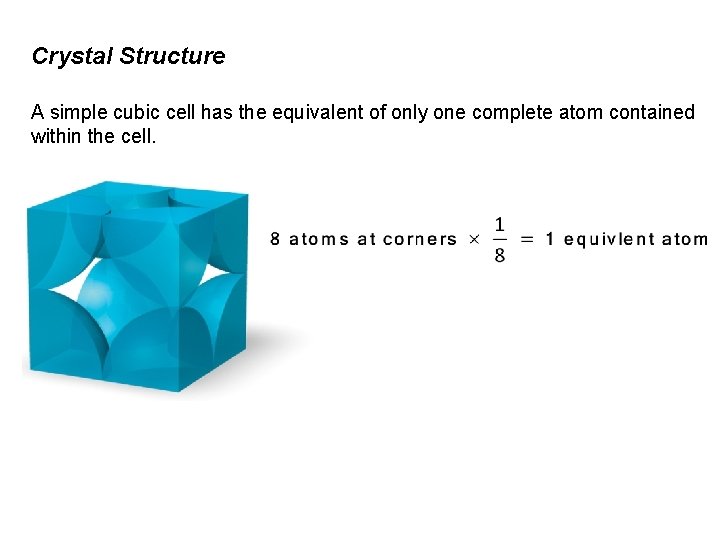
Crystal Structure A simple cubic cell has the equivalent of only one complete atom contained within the cell.
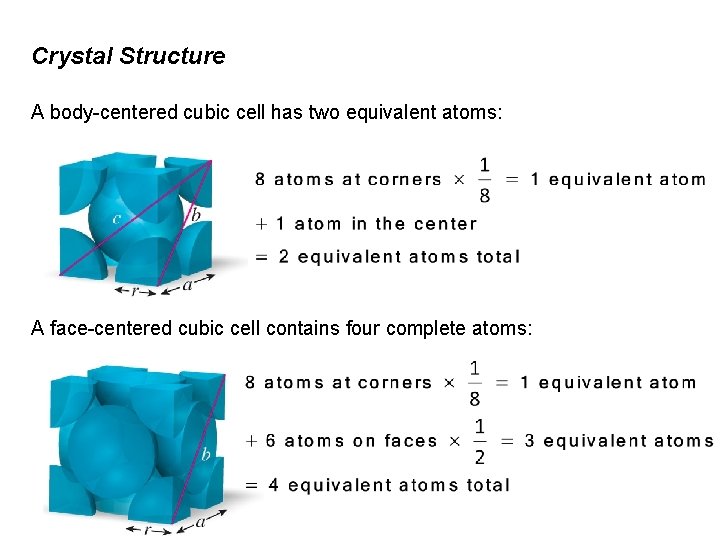
Crystal Structure A body-centered cubic cell has two equivalent atoms: A face-centered cubic cell contains four complete atoms:
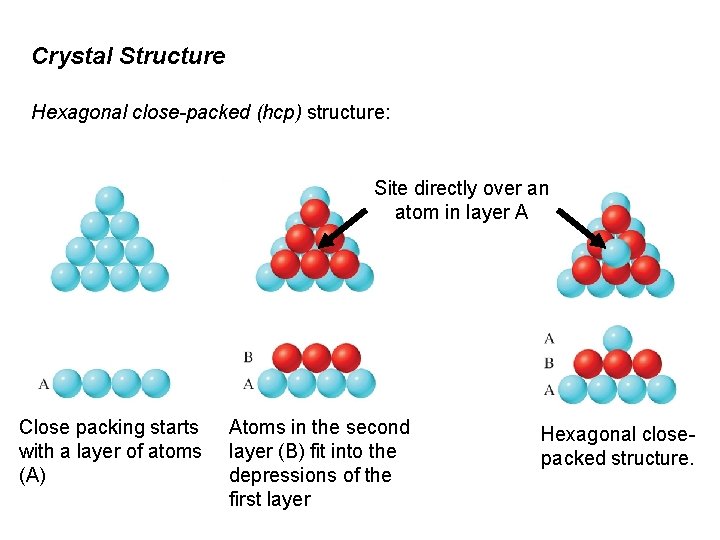
Crystal Structure Hexagonal close-packed (hcp) structure: Site directly over an atom in layer A Close packing starts with a layer of atoms (A) Atoms in the second layer (B) fit into the depressions of the first layer Hexagonal closepacked structure.
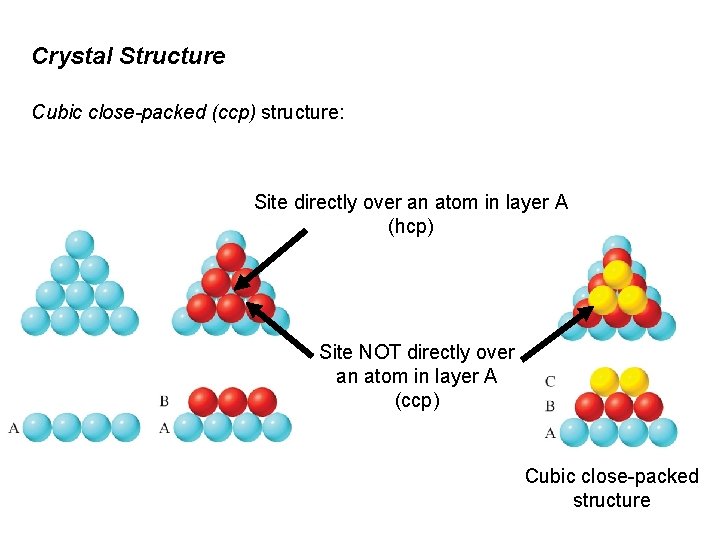
Crystal Structure Cubic close-packed (ccp) structure: Site directly over an atom in layer A (hcp) Site NOT directly over an atom in layer A (ccp) Cubic close-packed structure
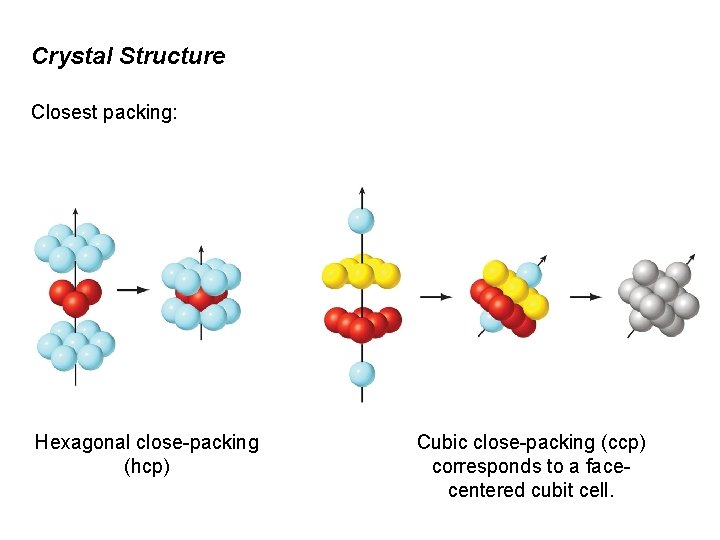
Crystal Structure Closest packing: Hexagonal close-packing (hcp) Cubic close-packing (ccp) corresponds to a facecentered cubit cell.
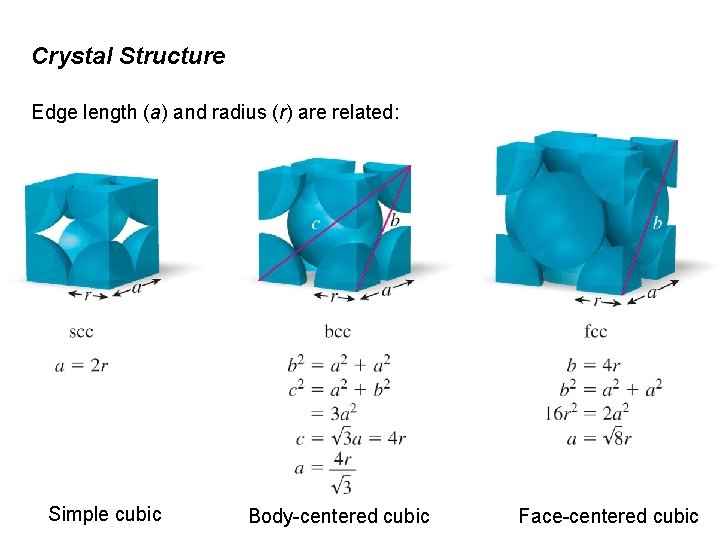
Crystal Structure Edge length (a) and radius (r) are related: Simple cubic Body-centered cubic Face-centered cubic

Crystal Structure When silver crystallizes, it forms face-centered cubic cells. The unit cell edge length is 4. 087 Å. Calculate the density of silver. Solution: Step 1: Density is calculated as follows: Step 2: Determine the mass of the unit cell.

Crystal Structure When silver crystallizes, it forms face-centered cubic cells. The unit cell edge length is 4. 087 Å. Calculate the density of silver. Solution: Step 3: Calculate the volume of the unit cell: Step 2: Determine the denisty.
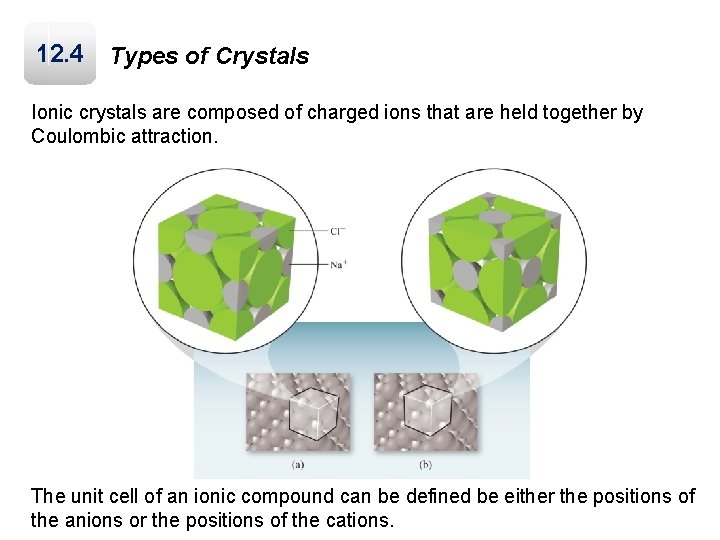
12. 4 Types of Crystals Ionic crystals are composed of charged ions that are held together by Coulombic attraction. The unit cell of an ionic compound can be defined be either the positions of the anions or the positions of the cations.
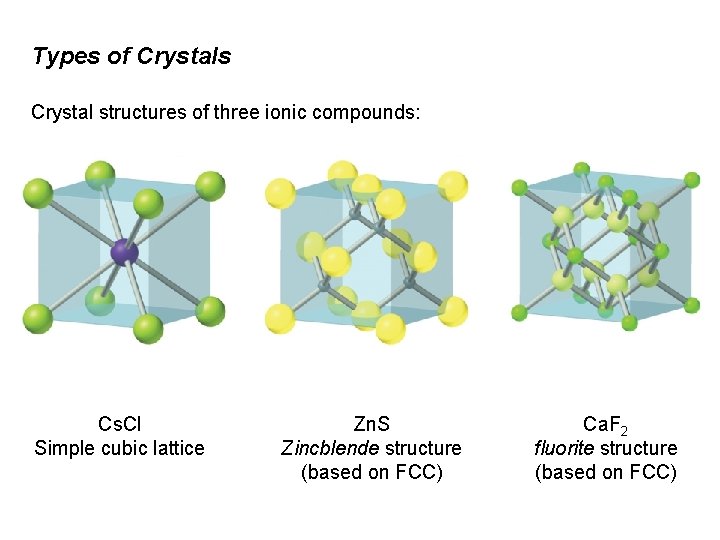
Types of Crystals Crystal structures of three ionic compounds: Cs. Cl Simple cubic lattice Zn. S Zincblende structure (based on FCC) Ca. F 2 fluorite structure (based on FCC)
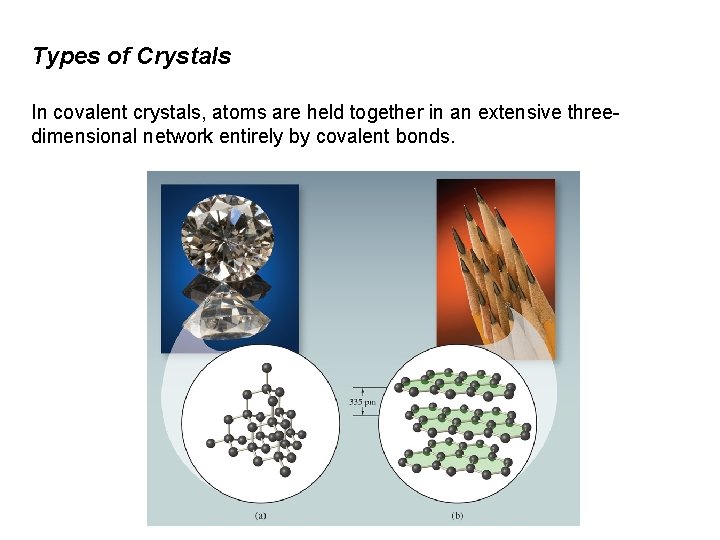
Types of Crystals In covalent crystals, atoms are held together in an extensive threedimensional network entirely by covalent bonds.

Types of Crystals In molecular crystals, the lattice points are occupied by molecules; the attractive forces between them are van der Waals forces and/or hydrogen bonding.
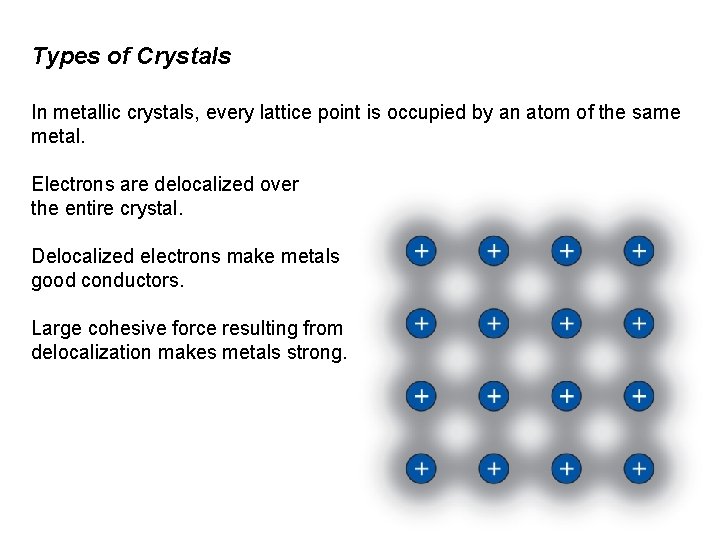
Types of Crystals In metallic crystals, every lattice point is occupied by an atom of the same metal. Electrons are delocalized over the entire crystal. Delocalized electrons make metals good conductors. Large cohesive force resulting from delocalization makes metals strong.
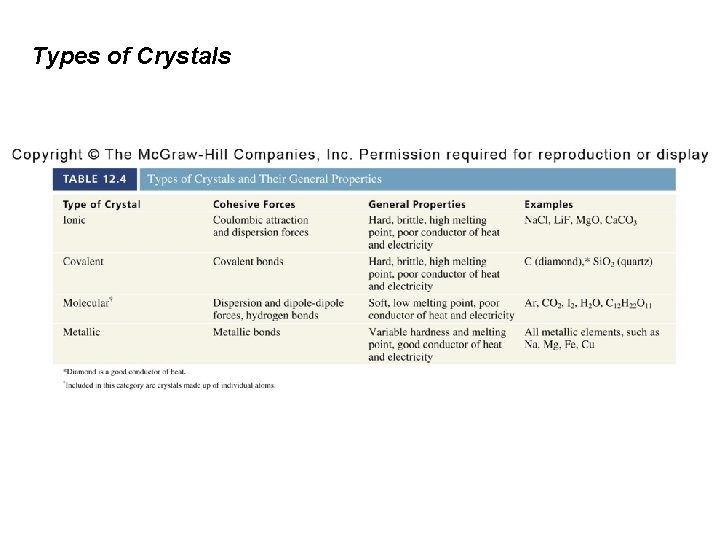
Types of Crystals

12. 5 Amorphous Solids Amorphous solids lack a regular three-dimensional arrangement of atoms. Glass is an amorphous solid. Glass is a fusion product. Si. O 2 is the chief component. Na 2 O and B 2 O 3 are typically fused with molten Si. O 2 and allowed to cool without crystallizing.

Amorphous Solids
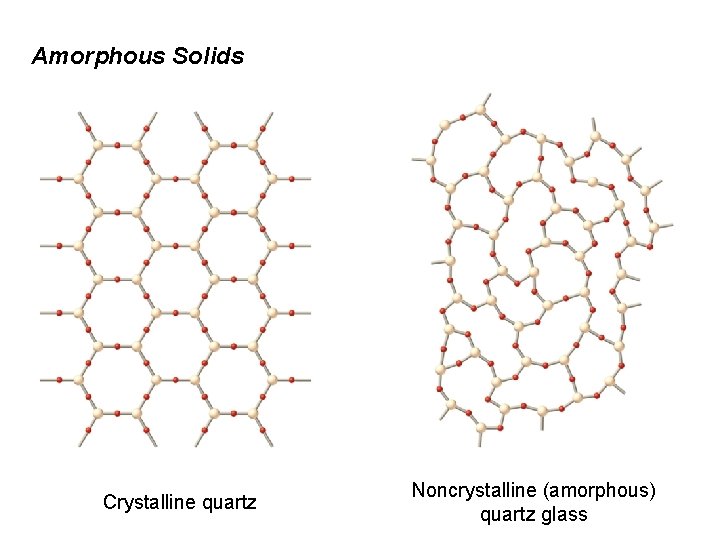
Amorphous Solids Crystalline quartz Noncrystalline (amorphous) quartz glass

12. 6 Phase Changes A phase is a homogeneous part of a system that is separated from the rest of a the system by a well defined boundary. When a substance goes from one phase to another phase, it has undergone a phase change. Example Phase Change Freezing of water H 2 O(l) → H 2 O(s) Evaporation (or vaporization) of water H 2 O(l) → H 2 O(g) Melting (fusion) of ice H 2 O(s) → H 2 O(l) Condensation of water vapor H 2 O(g) → H 2 O(l) Sublimation of dry ice CO 2(s) → CO 2(g) Deposition of iodine I 2(g) → I 2(s)
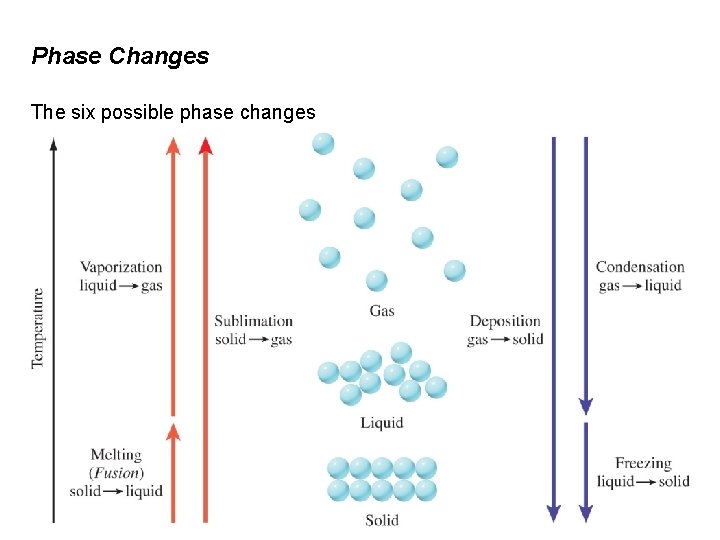
Phase Changes The six possible phase changes
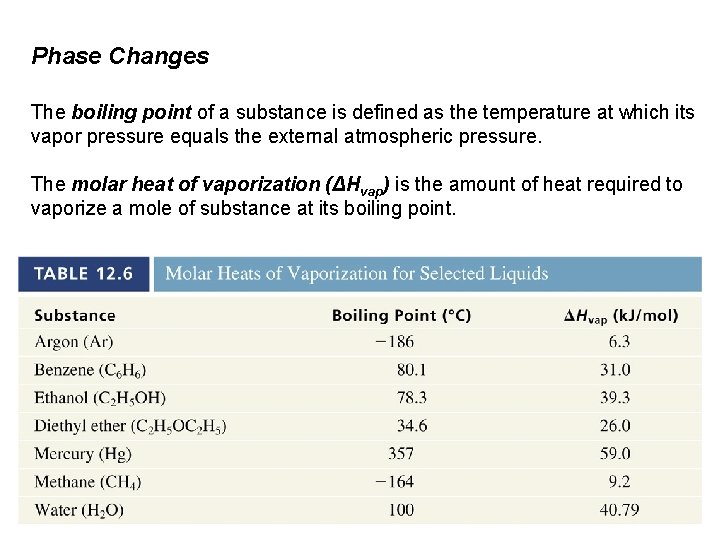
Phase Changes The boiling point of a substance is defined as the temperature at which its vapor pressure equals the external atmospheric pressure. The molar heat of vaporization (ΔHvap) is the amount of heat required to vaporize a mole of substance at its boiling point.

Phase Changes The transformation of a liquid to a solid is called freezing. The reverse process is called melting, or fusion. The melting point (freezing point) of a solid (or liquid) is the temperature at which the solid and liquid phases coexist in equilibrium. Ice ⇌ water H 2 O(s) ⇌ H 2 O(l) In dynamic equilibrium, the forward and reverse process are occurring at the same rate.

Phase Changes The molar heat of fusion (ΔHfus) is the energy required to melt 1 mol of a solid.
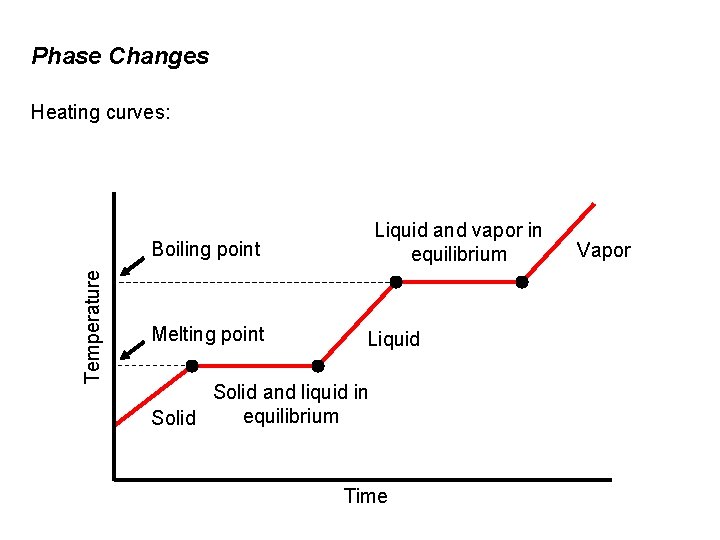
Phase Changes Heating curves: Liquid and vapor in equilibrium Temperature Boiling point Melting point Liquid Solid and liquid in equilibrium Solid Time Vapor

Phase Changes Sublimation is the process by which molecules go directly from the solid phase to the vapor phase. Deposition is reverse process of sublimation. The molar enthalpy of sublimation (ΔHsub) of a substance is the energy required to sublime 1 mole of a solid. ΔHsub = ΔHfus + ΔHvap Solid I 2 in equilibrium with its vapor
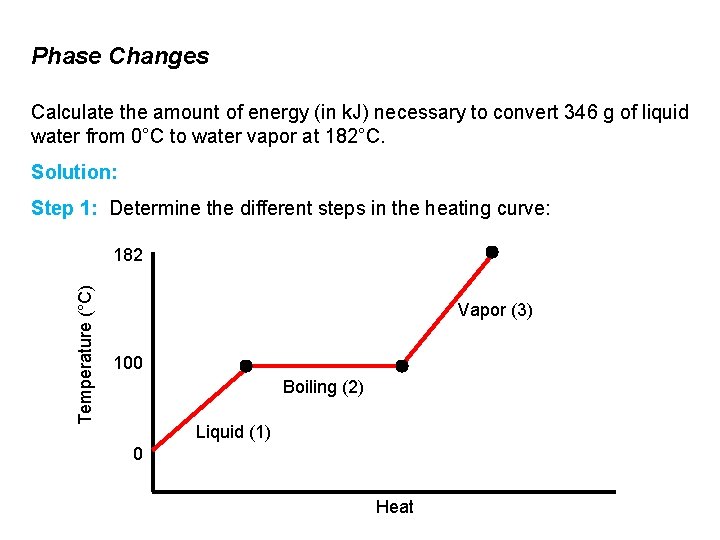
Phase Changes Calculate the amount of energy (in k. J) necessary to convert 346 g of liquid water from 0°C to water vapor at 182°C. Solution: Step 1: Determine the different steps in the heating curve: Temperature (°C) 182 Vapor (3) 100 Boiling (2) Liquid (1) 0 Heat
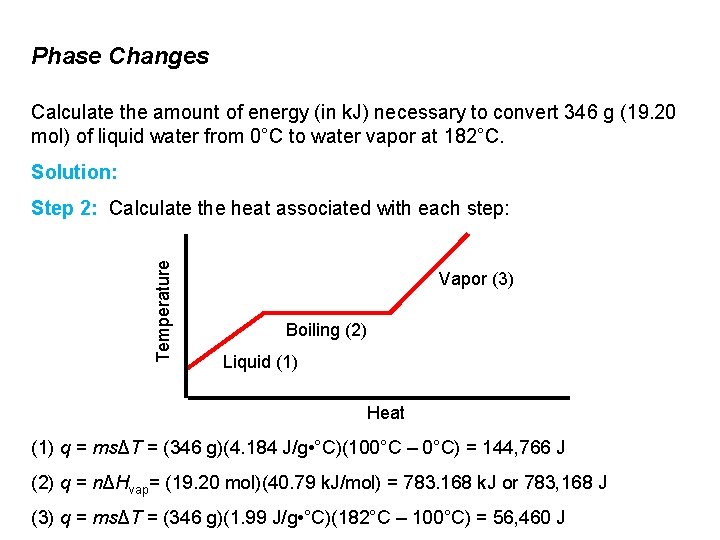
Phase Changes Calculate the amount of energy (in k. J) necessary to convert 346 g (19. 20 mol) of liquid water from 0°C to water vapor at 182°C. Solution: Temperature Step 2: Calculate the heat associated with each step: Vapor (3) Boiling (2) Liquid (1) Heat (1) q = msΔT = (346 g)(4. 184 J/g • °C)(100°C – 0°C) = 144, 766 J (2) q = nΔHvap= (19. 20 mol)(40. 79 k. J/mol) = 783. 168 k. J or 783, 168 J (3) q = msΔT = (346 g)(1. 99 J/g • °C)(182°C – 100°C) = 56, 460 J
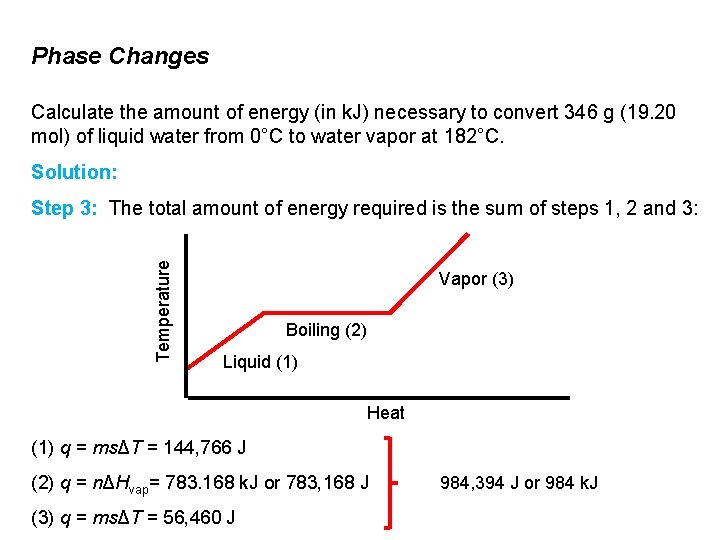
Phase Changes Calculate the amount of energy (in k. J) necessary to convert 346 g (19. 20 mol) of liquid water from 0°C to water vapor at 182°C. Solution: Temperature Step 3: The total amount of energy required is the sum of steps 1, 2 and 3: Vapor (3) Boiling (2) Liquid (1) Heat (1) q = msΔT = 144, 766 J (2) q = nΔHvap= 783. 168 k. J or 783, 168 J (3) q = msΔT = 56, 460 J 984, 394 J or 984 k. J
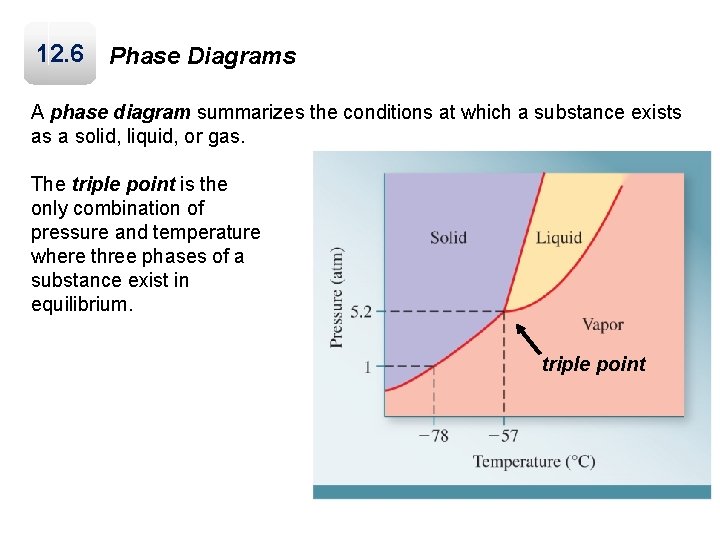
12. 6 Phase Diagrams A phase diagram summarizes the conditions at which a substance exists as a solid, liquid, or gas. The triple point is the only combination of pressure and temperature where three phases of a substance exist in equilibrium. triple point
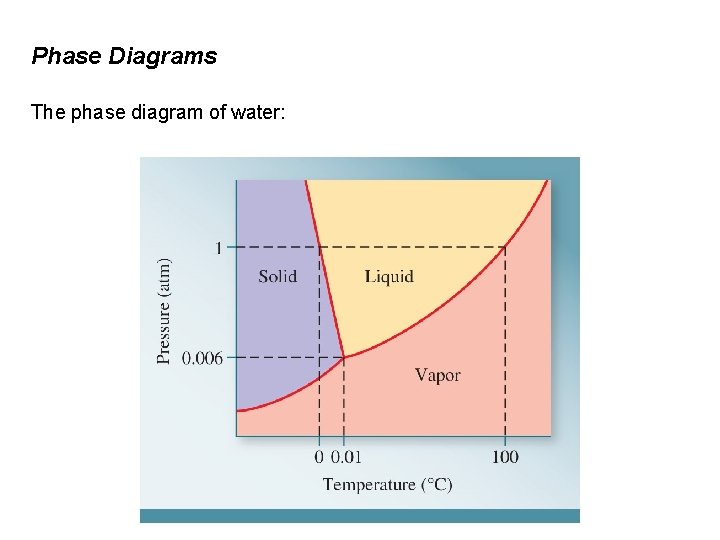
Phase Diagrams The phase diagram of water:

12 Key Concepts Intermolecular Forces Dipole-Dipole Interactions Hydrogen Bonding Dispersion Forces Ion-Dipole Interactions Properties of Liquids Surface Tension Viscosity Vapor Pressure Crystal Structure Unit Cells Packing Spheres Closest Packing Types of Crystals Ionic Crystals Covalent Crystals Molecular Crystals Metallic Crystals Amorphous Solids Phase Changes Liquid-Vapor Phase Transition Solid-Liquid Phase Transition Solid-Vapor Phase Transition Phase Diagrams
- Slides: 59