CHEMISTRY Safety in the Chemistry Laboratory and Creating

CHEMISTRY Safety in the Chemistry Laboratory and Creating the Science Notebook

Marie Curie (1867 -1934) � First famous woman scientist in the modern world � Received Nobel Prizes in both Chemistry and Physics � Coined the term radioactivity � Her notebooks are still so radioactive that they cannot be handled without special PPE.

Edwin Howard Armstrong (18901954) � American electrical engineer � Invented many of the circuits used in radio, radar, and television � Inventor of modern frequency modulation (FM) radio

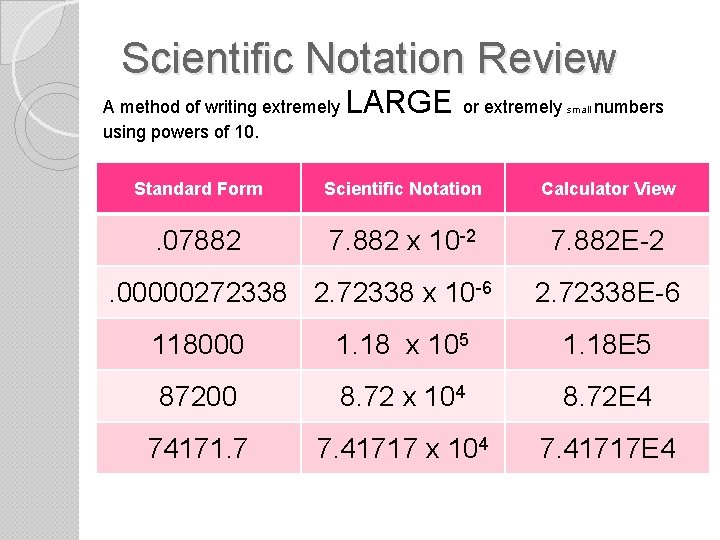
Scientific Notation Review A method of writing extremely using powers of 10. LARGE or extremely small numbers Standard Form Scientific Notation Calculator View . 07882 7. 882 x 10 -2 7. 882 E-2 . 00000272338 2. 72338 x 10 -6 2. 72338 E-6 118000 1. 18 x 105 1. 18 E 5 87200 8. 72 x 104 8. 72 E 4 74171. 7 7. 41717 x 104 7. 41717 E 4

Significant Figures � Precision-a measurement of the closeness of a set of measurements to each other � Accuracy-a measurement of the closeness of a measurement to the accepted or true value � Sig Fig Rules ◦ Non-zero digits and zeros between non-zero digits are always significant ◦ Leading zeros are never significant ◦ Zeros to the right of all non-zero digits are only significant if a decimal point is shown ◦ For values written in scientific notation, the digits in the coefficient are significant (everything before the “x”) ◦ In a regular math equation, there as many places behind the decimal point in the answer as there are sig figs in the original number

Significant Figures � 1234 � 0. 00030 � 0. 023 � 102010 � 890 � 780. � 91010 � 1000 � 1090. 0010 � 918. 010 � 0. 00120 � 0. 0001 � 3. 4 � 8120 x 104 � 9. 0 x 10 -3 � 9. 010 x 10 -2 � 72 The underlined numbers are significant.

Safety Goals �On an index card (in the bucket) answer the following: ◦ What is an appropriate safety goal for our class? Example: 78% safe ◦ Explain your answer.

Safety Goals �Let’s say that I teach 6 classes of Chemistry with 25 students in each class. �That’s a total of 150 students. �Let’s pretend ONE student sustains a major injury during a Chemistry lab. �That would mean my lab is 99. 99% safe. ◦ Which is pretty good, unless that one student is YOU.

Safety Goals �Our goal for this year is to be 100% safe �That means NO injuries or deaths in the Chemistry lab. �So, how are we going to make this happen?
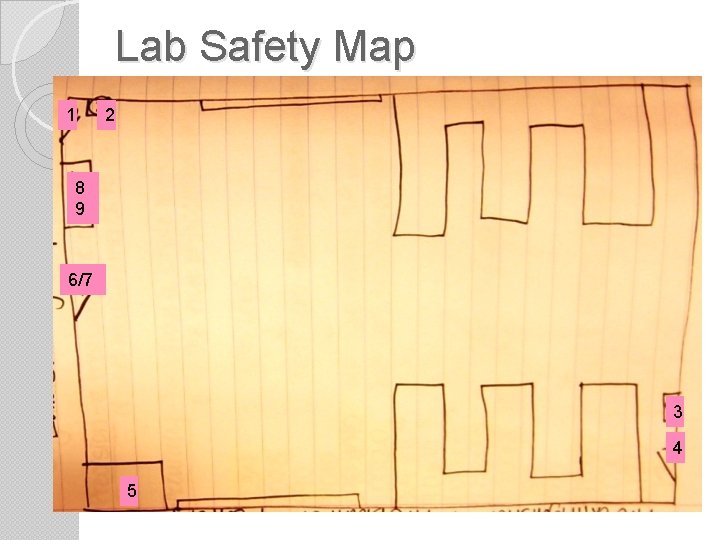
Lab Safety Map 1 2 8 9 6/7 3 4 5

MSDS Guiding Questions 1. 2. 3. 4. 5. 6. What section of information is most important? Why? What potential hazards does your substance pose? What type(s) of personal protective equipment (PPE) is/are recommended when handling your substance? What basic first aid do you need to be aware of before using your substance in the lab? How should your substance be disposed? Note any words or acronyms used in the MSDS with which you are not familiar.
- Slides: 12