CHEMISTRY ORGANIC CHEMISTRY STUDY GUIDE Define Chemistry science

















- Slides: 18

CHEMISTRY & ORGANIC CHEMISTRY STUDY GUIDE

Define “Chemistry” science that deals with the composition & properties of matter & the way that matter reacts with each other 2. Define “element” matter consisting of only 1 kind of atom 3. Define “chemical symbol” abbreviation for the element name Matter= mass- quantity or amount of an object volume-space an object takes up 1.

4. Write the chemical symbol for the following elements: *Carbon____ *Hydrogen _____ *Oxygen_____ *Nitrogen_____ *Phosphorous_____ *Potassium_____ *Sodium_____ *Chlorine_____ *Calcium_____ *Magnesium_____

4. Write the chemical symbol for the following elements: *Carbon C *Hydrogen H *Oxygen O *Nitrogen N *Phosphorous P *Potassium K *Sodium Na *Chlorine Cl *Calcium Ca *Magnesium Mg

5. 6. Define “chemical reaction”. A combination of atoms that occurs so greater stability occurs. Atoms will gain/lose/or share electrons for the purpose of achieving stability. Explain the following chemical reactions: H 2 + O 2 H 2 O the reactants are ______ the product is ________

EXPLAIN l 6 CO 2 + 6 H 20 →C 6 H 12 O 6 + 6 O 2 light The reactants are ________ The products are________ 7. 8. 9. Define “compound”. Matter composed of 2 or more types of elements—ionic bond Define “molecule”. Matter composed of 2 or more atoms of elements—covalent bond. The smallest part of a compound where atoms are bonded covalently Salt has a chemical formula that is Na. Cl; is salt an element? Explain. An element is 1 kind of atom Na & Cl are 2 different elements

10. Write the chemical formula for the following compounds (abbreviation for a compound): 1. 2. 3. 11. 12. 13. Water __________ Glucose_________ Carbon dioxide_______ Chemical reactions that RELEASE energy are called exothermic. Chemical reactions that REQUIRE energy are called endothermic. The Chemistry of LIVING things is called organic chemistry.

14. § § Types of Chemistry Organic— compounds contain the element Carbon except for the compound CO 2. Inorganic—compounds that DO NOT contain the element Carbon.

15. 16. 17. The 4 most abundant ELEMENTS in living things are C, H, O, and N. The most abundant ELEMENT in living things is Carbon. The most abundant COMPOUND in living things is H 2 O.

18. 19. Types of MIXTURES—define a. Solution: homogeneous-best mixeduniform in color; transparent b. Suspension: heterogeneous- most particles settle out upon standing; not evenly mixed c. Colloid: homogeneous mixture-always cloudy; particles never settle out; can’t see light through it Draw the particle size for: Solution Colloid Suspension

Determine the type of MIXTURE for each of the following: 20. § § § § § Dirt in water_____ Sugar dissolved in hot tea______ Egg white______ Fog________ Salad dressing______ Orange juice with pulp_____ Milk (homogenized)_____ Blood______&_______ explain Guntersville lake______&______explain

Determine the type of MIXTURE for each of the following: 20. § § § § § Dirt in water-suspension Sugar dissolved in hot tea-solution Egg white-colloid Fog-colloid Salad dressing-suspension Orange juice with pulp-suspension Milk (homogenized)-colloid Blood-solution&-suspension explain: food dissolved in blood & plasma Guntersville lake-solution & suspension explain: oxygen dissolved dirt suspended

21. 22. Parts of a solution *solute: part that is dissolved (usually a solid) *solvent: part that does the dissolving (usually a liquid) A solution may become SUPERSATURATED. Explain. Sponge- ground after heavy rain-there is too much solute for the solvent-it is impossible for the solution to remain homogeneous-some solute will settle out.

For the following SOLUTIONS, indicate the part that is the solute and the part that is the solvent: 23. § § § Coffee granules in water: coffee is the solute; Water is the solvent Fingernail polish remover dissolves fingernail polish: Fingernail polish remover is the solvent; fingernail polish is the solute. Gojo hand cleaner dissolves grease on our hands: Gojo is the solvent; grease is the solute. Ammonium hydroxide in water: Ammonium hydroxide is the solute; water is the solvent 30 ml of 95% alcohol dissolves 0. 3 g of methylene blue dye: 95% alcohol is the solvent; methylene blue dye is the solute.

Water is called the UNIVERSAL SOLVENT. Types of SOLUTIONS: *Acid-define: has more H+ than OH*Base-define: has more OH- than H+ *Neutral-define: has = OH- and H+ H+ is called hydrogen ion; OH- is called hydroxide ion Determine whether the following solutions are ACID/BASE/NEUTRAL 24. 25. 26. 27. • • • HCL H+ + Cl. Na. OH Na+ + OHH 2 O H+ + OHKOH K+ + OHNH 3 H+ + NO 3 Determine whether the following solutions are ACID/BASE/NEUTRAL based on observable characteristics: (Acids=sour; base=bitter/slick) 28. • • Aspirin acid Soap base Lemon juice acid Milk neutral

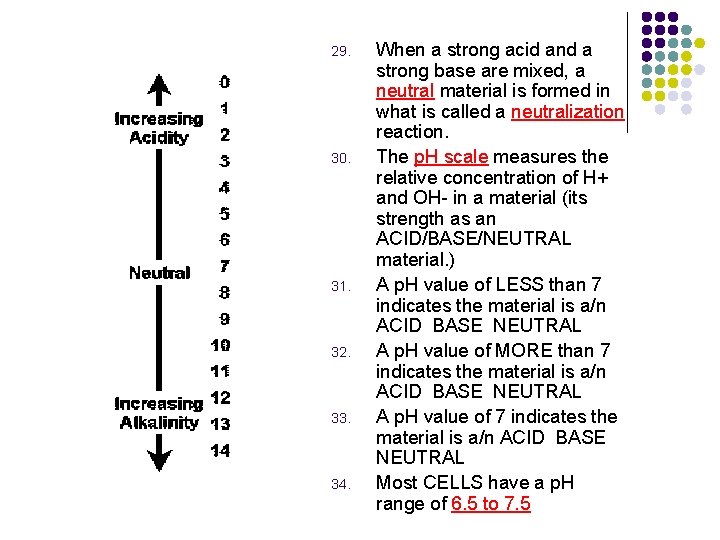
29. 30. 31. 32. 33. 34. When a strong acid and a strong base are mixed, a neutral material is formed in what is called a neutralization reaction. The p. H scale measures the relative concentration of H+ and OH- in a material (its strength as an ACID/BASE/NEUTRAL material. ) A p. H value of LESS than 7 indicates the material is a/n ACID BASE NEUTRAL A p. H value of MORE than 7 indicates the material is a/n ACID BASE NEUTRAL A p. H value of 7 indicates the material is a/n ACID BASE NEUTRAL Most CELLS have a p. H range of 6. 5 to 7. 5

35. • • • Determine whether the following p. H is a strong Acid/ weak Acid/ strong Base/ weak Base/ Neutral material: p. H=2. 3 strong acid p. H=7 neutral p. H=13. 4 strong base p. H=8. 2 weak base p. H=6. 1 weak acid

36. a. b. c. Determine whether the following is an ACID/BASE/NEUTRAL material: Blue Litmus paper and Red Litmus paper end up RED after they are placed in a solution acid Blue Litmus paper and Red Litmus paper end up BLUE after they are placed in a solution base Blue Litmus paper stays BLUE and Red Litmus paper stays RED after they are placed in solution neutral