Chemistry of the Atmosphere Structure and Chemical Composition

































- Slides: 44

Chemistry of the Atmosphere

Structure and Chemical Composition Photochemistry Units of Atmospheric Concentration Atmospheric Lifetimes Mixing time vs residence time Atmospheric Emissions Questions • What are the species (gases & particles) present in the atmosphere? What are their natural and anthropogenic sources? • What chemical reactions do these species undergo in the atmosphere? Sulfur Nitrogen oxides Ammonia • What are the products from the atmospheric transformations of these species? Organic nitrogen Types of Atmospheric Reactions Thermodynamic approaches Kinetic approaches Photochemical Reactions • What effects do the presence of these species and their chemical transformation products have on the atmosphere, climate, and human health? Free Radical Reactions 2 12/12/2021 The Atmosphere CHEM 196 Petrucci

Structure and Chemical Composition

Structure and Chemical Composition Photochemistry Units of Atmospheric Concentration Atmospheric Lifetimes Mixing time vs residence time Atmospheric Emissions Sulfur Nitrogen oxides Ammonia Organic nitrogen Types of Atmospheric Reactions Thermodynamic approaches Kinetic approaches Photochemical Reactions Free Radical Reactions 4 12/12/2021 Chemical Composition • Three gases make up ~99% of total atmospheric mass (~5. 3 x 1018 kg)* • N 2 (78%) – diatomic nitrogen • O 2 (21%) – diatomic oxygen • Ar (1%) – argon • These 3 gases are relatively un-reactive and their mean residence times are much longer that the rate of atmospheric mixing • As a result, their concentrations are relatively uniform globally • Water vapor is the next most abundant constituent • Found in the lowest part of the atmosphere • Concentrations are variable • Can be as high as 3% in some areas • Remaining gaseous constituents, trace gases, represent <1% of atmosphere, but play critical role in Earth’s radiative balance Composition CHEM 196 Petrucci

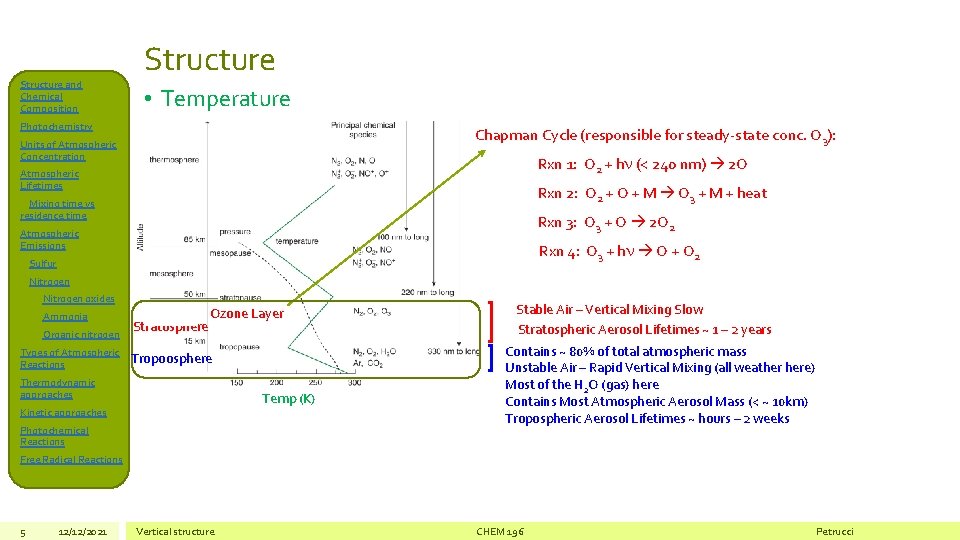
Structure and Chemical Composition Structure • Temperature Photochemistry Chapman Cycle (responsible for steady-state conc. O 3): Units of Atmospheric Concentration Rxn 1: O 2 + hn (< 240 nm) 2 O Atmospheric Lifetimes Rxn 2: O 2 + O + M O 3 + M + heat Mixing time vs residence time Rxn 3: O 3 + O 2 O 2 Atmospheric Emissions Rxn 4: O 3 + hn O + O 2 Sulfur Nitrogen oxides Ammonia Organic nitrogen Types of Atmospheric Reactions Stratosphere Ozone Layer Tropoosphere Thermodynamic approaches Temp (K) Kinetic approaches Photochemical Reactions Stable Air – Vertical Mixing Slow Stratospheric Aerosol Lifetimes ~ 1 – 2 years Contains ~ 80% of total atmospheric mass Unstable Air – Rapid Vertical Mixing (all weather here) Most of the H 2 O (gas) here Contains Most Atmospheric Aerosol Mass (< ~ 10 km) Tropospheric Aerosol Lifetimes ~ hours – 2 weeks Free Radical Reactions 5 12/12/2021 Vertical structure CHEM 196 Petrucci

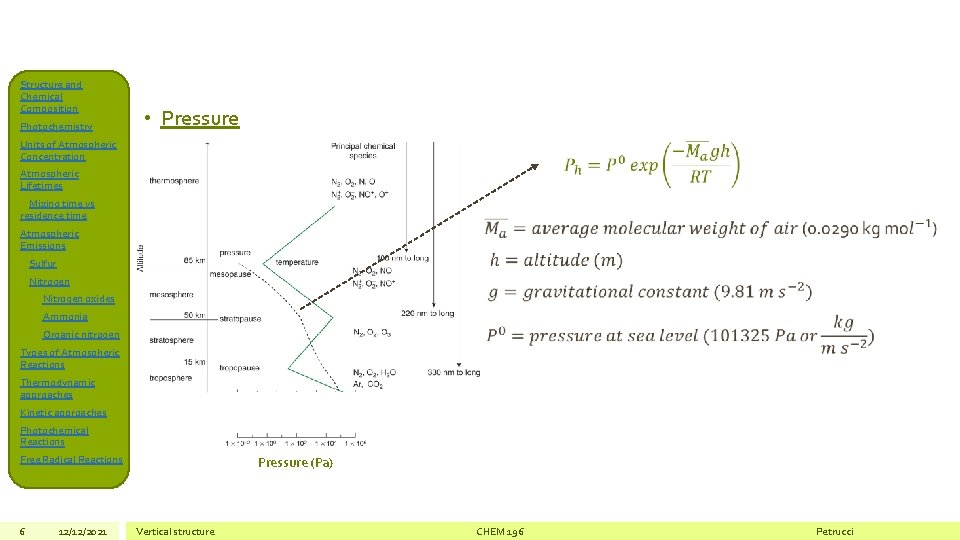
Structure and Chemical Composition Photochemistry • Pressure Units of Atmospheric Concentration Atmospheric Lifetimes Mixing time vs residence time Atmospheric Emissions Sulfur Nitrogen oxides Ammonia Organic nitrogen Types of Atmospheric Reactions Thermodynamic approaches Kinetic approaches Photochemical Reactions Free Radical Reactions 6 12/12/2021 Pressure (Pa) Vertical structure CHEM 196 Petrucci

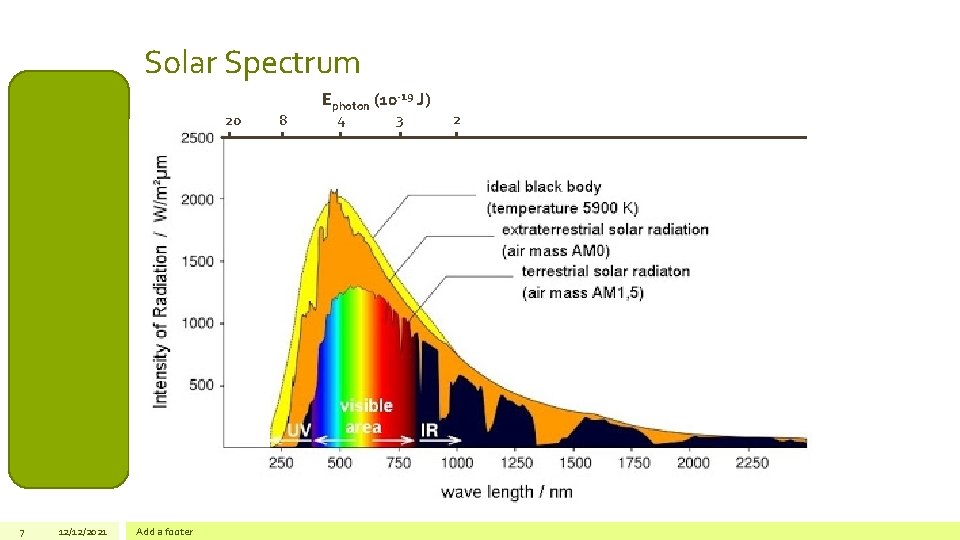
Solar Spectrum 20 7 12/12/2021 Add a footer 8 Ephoton (10 -19 J) 4 3 2

Structure and Chemical Composition Photochemistry Units of Atmospheric Concentration Atmospheric Lifetimes Mixing time vs residence time Atmospheric Emissions Sulfur Nitrogen oxides Photochemistry • Solar energy has a large influence on the chemical composition of the atmosphere • Influence depends on photon energies available • Mesosphere: >100 nm (2 x 10 -18 J) • Stratosphere: > 220 nm (9 x 10 -19 J) • Troposphere: > 320 nm (6 x 10 -19 J) Ammonia Bond dissociation energy (k. J/mol) Bond dissociation energy (J/bond) λthreshold (nm) O 2 498 8. 3 x 10 -19 239 N 2 945 1. 6 x 10 -18 124 O-O* 364 6. 0 x 10 -19 331 N-O* 300 5. 0 x 10 -19 398 C-C 350 5. 8 x 10 -19 343 Organic nitrogen Types of Atmospheric Reactions Thermodynamic approaches Kinetic approaches Photochemical Reactions Free Radical Reactions 8 12/12/2021 Atmospheric lifetimes CHEM 196 hν * Reactants Products Petrucci

Structure and Chemical Composition Units of Atmospheric Concentration Photochemistry Units of Atmospheric Concentration Atmospheric Lifetimes • Mixing time vs residence time Atmospheric Emissions Sulfur Nitrogen oxides Ammonia Organic nitrogen Types of Atmospheric Reactions Thermodynamic approaches Kinetic approaches Photochemical Reactions Free Radical Reactions 9 12/12/2021 Atmospheric concentrations CHEM 196 Petrucci

Structure and Chemical Composition of the Atmosphere Photochemistry Units of Atmospheric Concentration Atmospheric Lifetimes Mixing time vs residence time Atmospheric Emissions Sulfur Nitrogen oxides Ammonia Organic nitrogen Types of Atmospheric Reactions Thermodynamic approaches Kinetic approaches Photochemical Reactions Free Radical Reactions 10 12/12/2021 Atmospheric concentrations CHEM 196 Petrucci

Structure and Chemical Composition Photochemistry Units of Atmospheric Concentration • Concentration (mol m-3) • depends on pressure and temperature (through IGL) • Mass loading or concentration (µg m-3) Atmospheric Lifetimes Mixing time vs residence time Atmospheric Emissions Sulfur Nitrogen oxides Ammonia For example, at STP, 390 410 ppm CO 2 in atmosphere corresponds to Organic nitrogen Types of Atmospheric Reactions Thermodynamic approaches Kinetic approaches Photochemical Reactions Free Radical Reactions 11 12/12/2021 So, while CO 2 comprises only 0. 041% of the atmosphere by volume, its fraction by mass is 0. 065%. Atmospheric concentrations CHEM 196 Petrucci

Structure and Chemical Composition Photochemistry Units of Atmospheric Concentration • Atmospheric Lifetimes Mixing time vs residence time Atmospheric Emissions Sulfur Nitrogen oxides Ammonia saturation vapor pressure of water at that temperature Organic nitrogen Types of Atmospheric Reactions Thermodynamic approaches Kinetic approaches Photochemical Reactions Free Radical Reactions 12 12/12/2021 Atmospheric concentrations CHEM 196 Petrucci

Atmospheric Lifetimes

Structure and Chemical Composition Photochemistry Units of Atmospheric Concentration Atmospheric Lifetimes Mixing time vs residence time Atmospheric Emissions Sulfur • Atmospheric lifetime tells us on average how long a representative molecule/particle will stay in the atmosphere before it is removed. • Only two possible exits of species from atmosphere: • Precipitation (wet deposition) • Settling (dry deposition) Nitrogen oxides Ammonia Organic nitrogen Types of Atmospheric Reactions Thermodynamic approaches Kinetic approaches Photochemical Reactions Free Radical Reactions 14 12/12/2021 Atmospheric lifetimes CHEM 196 Petrucci

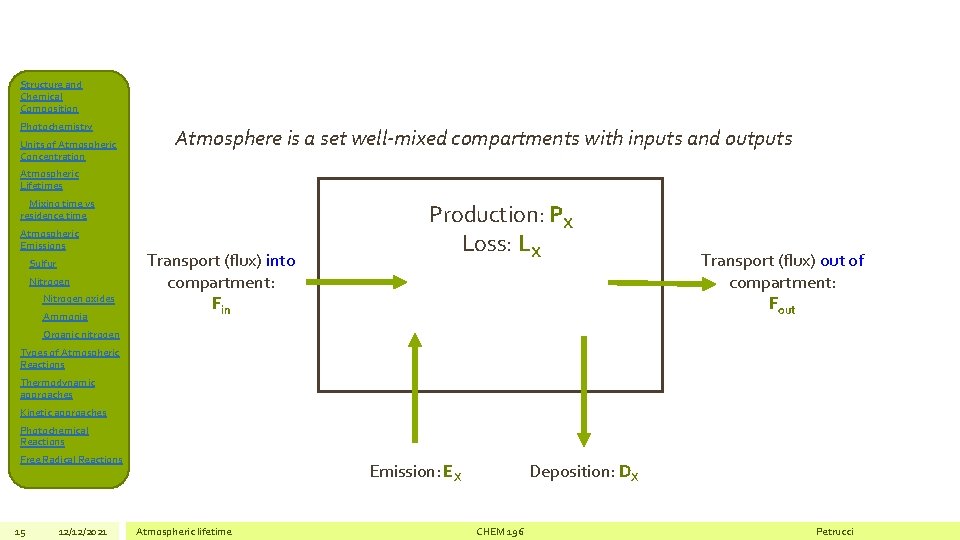
Structure and Chemical Composition Photochemistry Units of Atmospheric Concentration Atmosphere is a set well-mixed compartments with inputs and outputs Atmospheric Lifetimes Mixing time vs residence time Atmospheric Emissions Sulfur Nitrogen oxides Ammonia Transport (flux) into compartment: Fin Production: PX Loss: LX Transport (flux) out of compartment: Fout Organic nitrogen Types of Atmospheric Reactions Thermodynamic approaches Kinetic approaches Photochemical Reactions Free Radical Reactions 15 12/12/2021 Emission: EX Atmospheric lifetime Deposition: DX CHEM 196 Petrucci

Structure and Chemical Composition Photochemistry Units of Atmospheric Concentration Mixing time vs residence time Balance of the two defines mixing state of species i in the atmosphere Atmospheric Lifetimes Mixing time vs residence time Atmospheric Emissions Characteristic mixing time, τM, is the time needed to thoroughly mix a chemical in that compartment of air Sulfur Nitrogen oxides Ammonia Organic nitrogen Types of Atmospheric Reactions Thermodynamic approaches Kinetic approaches A compartment is poorly mixed if τM > τi. Mixing state is therefore species specific through τi. Mixing state is different for vertical (~1 week) and horizontal year) directions (~1 Photochemical Reactions Free Radical Reactions 18 12/12/2021 Atmospheric lifetime CHEM 196 Petrucci

Structure and Chemical Composition Photochemistry Units of Atmospheric Concentration Atmospheric Lifetimes Mixing time vs residence time Atmospheric Emissions Sulfur Nitrogen oxides Ammonia Organic nitrogen Types of Atmospheric Reactions Thermodynamic approaches Emissions of Trace Gases & Other Constituents (Aerosols) • TRACE GASES, represent <1% of atmosphere but play CRITICAL role in Earth’s radiative balance and atmospheric chemistry • Major groupings: • Sulfur-containing compounds • Nitrogen-containing compounds • Carbon-containing compounds • Halogen-containing compounds • Aerosols • Ozone (O 3) Kinetic approaches Photochemical Reactions Free Radical Reactions 19 12/12/2021 Atmospheric trace gases CHEM 196 Petrucci

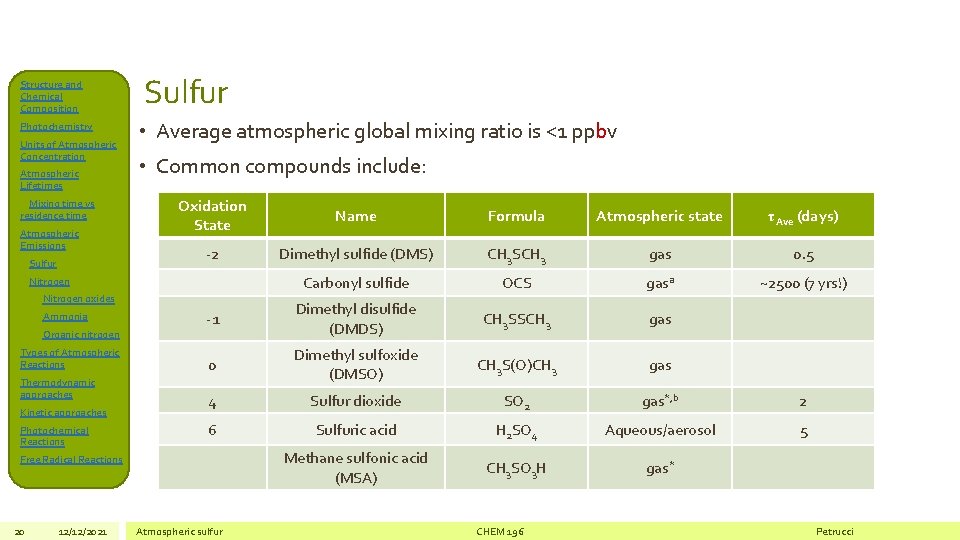
Structure and Chemical Composition Photochemistry Units of Atmospheric Concentration Atmospheric Lifetimes Mixing time vs residence time Atmospheric Emissions Sulfur • Average atmospheric global mixing ratio is <1 ppbv • Common compounds include: Oxidation State Name Formula Atmospheric state τAve (days) -2 Dimethyl sulfide (DMS) CH 3 SCH 3 gas 0. 5 Carbonyl sulfide OCS gasa ~2500 (7 yrs!) -1 Dimethyl disulfide (DMDS) CH 3 SSCH 3 gas 0 Dimethyl sulfoxide (DMSO) CH 3 S(O)CH 3 gas 4 Sulfur dioxide SO 2 gas*, b 2 6 Sulfuric acid H 2 SO 4 Aqueous/aerosol 5 Methane sulfonic acid (MSA) CH 3 SO 3 H gas* Nitrogen oxides Ammonia Organic nitrogen Types of Atmospheric Reactions Thermodynamic approaches Kinetic approaches Photochemical Reactions Free Radical Reactions 20 12/12/2021 Atmospheric sulfur CHEM 196 Petrucci

Structure and Chemical Composition Sulfur emissions Photochemistry Units of Atmospheric Concentration Atmospheric Lifetimes Mixing time vs residence time Atmospheric Emissions Sulfur Nitrogen Almost entirely DMS Nitrogen oxides Ammonia Organic nitrogen Types of Atmospheric Reactions Thermodynamic approaches Kinetic approaches Photochemical Reactions Free Radical Reactions 21 12/12/2021 These emission rates and τi, ave ~ 1 -7 days give ξSO 2 ~ 100 -1000 pptv Atmospheric sulfur CHEM 196 Petrucci

Structure and Chemical Composition Photochemistry Units of Atmospheric Concentration Atmospheric Lifetimes Mixing time vs residence time Atmospheric Emissions Sulfur Nitrogen oxides Ammonia Organic nitrogen Types of Atmospheric Reactions • Dimethyl sulfide (DMS, CH 3 SCH 3): is dominant S species emitted from world’s oceans; produced by marine phytoplankton • DMS reacts primarily with OH and NO 3 radicals; oxidation of DMS is only source of MSA and SO 2 in marine atmosphere (which ultimately generates H 2 SO 4 aerosol) • Carbonyl Sulfide (OCS): is most abundant S gas in the global background atmosphere because of its low reactivity in the troposphere • Only S compound that survives to enter stratosphere (besides SO 2 from volcanoes) • responsible for maintenance of normal stratospheric SO 42 - aerosol layer • Sulfur Dioxide (SO 2): predominant anthropogenic S-containing air pollutant; its gas-phase oxidation by OH radicals can produce H 2 SO 4 Thermodynamic approaches Kinetic approaches Photochemical Reactions Free Radical Reactions 23 12/12/2021 Atmospheric sulfur CHEM 196 Petrucci

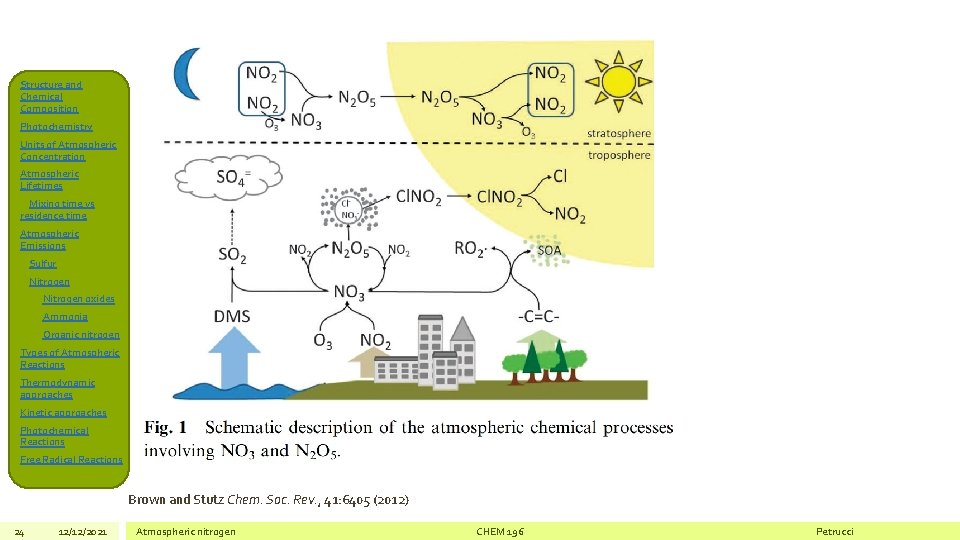
Structure and Chemical Composition Photochemistry Units of Atmospheric Concentration Atmospheric Lifetimes Mixing time vs residence time Atmospheric Emissions Sulfur Nitrogen oxides Ammonia Organic nitrogen Types of Atmospheric Reactions Thermodynamic approaches Kinetic approaches Photochemical Reactions Free Radical Reactions Brown and Stutz Chem. Soc. Rev. , 41: 6405 (2012) 24 12/12/2021 Atmospheric nitrogen CHEM 196 Petrucci

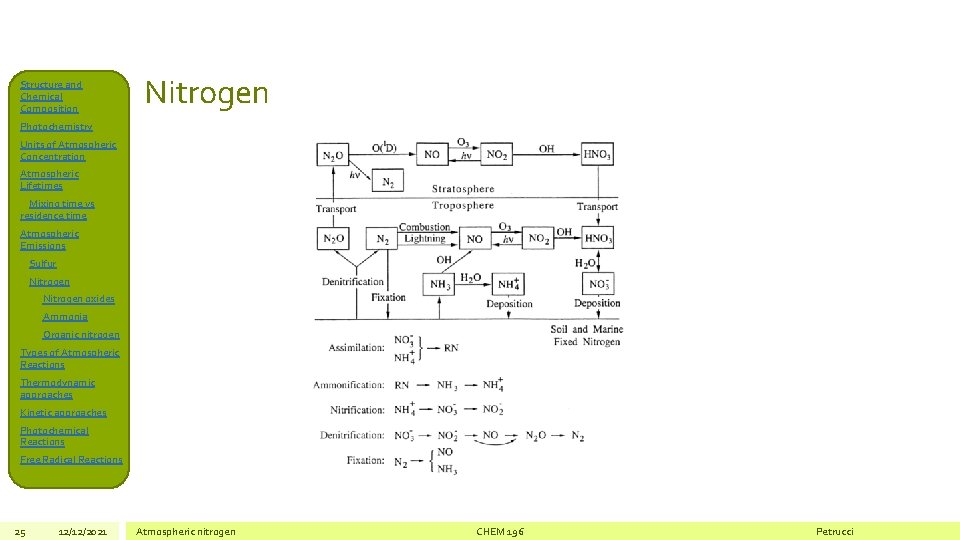
Structure and Chemical Composition Nitrogen Photochemistry Units of Atmospheric Concentration Atmospheric Lifetimes Mixing time vs residence time Atmospheric Emissions Sulfur Nitrogen oxides Ammonia Organic nitrogen Types of Atmospheric Reactions Thermodynamic approaches Kinetic approaches Photochemical Reactions Free Radical Reactions 25 12/12/2021 Atmospheric nitrogen CHEM 196 Petrucci

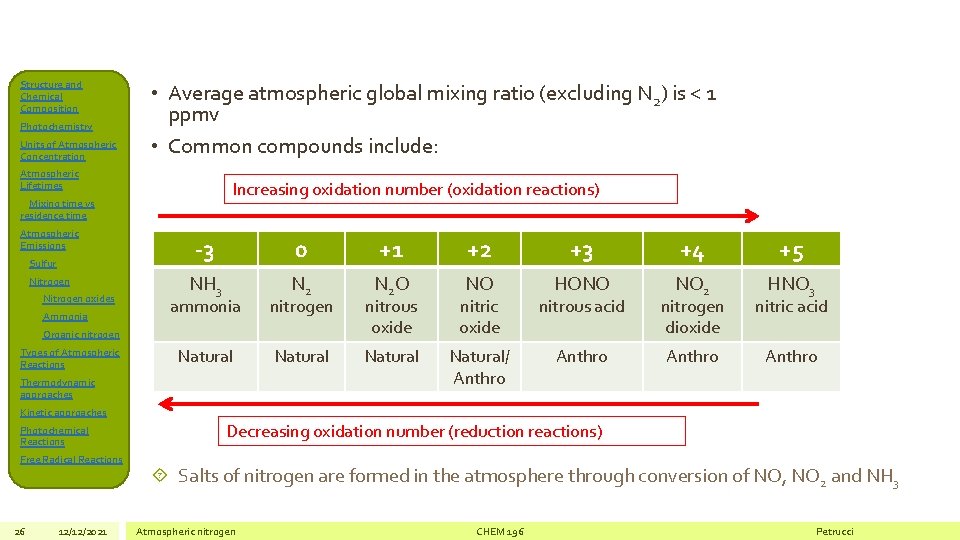
Structure and Chemical Composition Photochemistry Units of Atmospheric Concentration • Average atmospheric global mixing ratio (excluding N 2) is < 1 ppmv • Common compounds include: Atmospheric Lifetimes Increasing oxidation number (oxidation reactions) Mixing time vs residence time Atmospheric Emissions Sulfur Nitrogen oxides Ammonia -3 0 +1 +2 +3 +4 +5 NH 3 N 2 O NO HONO NO 2 HNO 3 ammonia nitrogen nitrous oxide nitric oxide nitrous acid nitrogen dioxide nitric acid Natural/ Anthro Organic nitrogen Types of Atmospheric Reactions Thermodynamic approaches Kinetic approaches Photochemical Reactions Free Radical Reactions 26 12/12/2021 Decreasing oxidation number (reduction reactions) Salts of nitrogen are formed in the atmosphere through conversion of NO, NO 2 and NH 3 Atmospheric nitrogen CHEM 196 Petrucci

Structure and Chemical Composition Photochemistry Units of Atmospheric Concentration Atmospheric Lifetimes Mixing time vs residence time Atmospheric Emissions Sulfur Nitrogen oxides Ammonia Organic nitrogen Types of Atmospheric Reactions Thermodynamic approaches Kinetic approaches Nitrous oxide • Potent greenhouse gas • very large absorptivity within solar spectrum • Per unit mass, global warming potential (GWP) 300 x that of CO 2 • concentrations (~300 ppbv) much lower than CO 2 and H 2 O, but • non-reactive (τi ~ 131 years!) • major atmospheric sink is photodissociation in stratosphere (90%) and rxn with excited oxygen atoms, O(1 D) (10%); • the latter provides major input of nitric oxide (NO) to stratosphere – we will return to this in detail soon!! • Pre-industrial (i. e. , natural) emission rate ~ 10 Tg(N) yr-1 • Current flux from anthropogenic sources ~ 6. 9 Tg(N) yr-1 Photochemical Reactions Free Radical Reactions 27 12/12/2021 ØAnnual growth rate ~ 3. 6 Tg(N) yr-1 Atmospheric nitrogen CHEM 196 Petrucci

Structure and Chemical Composition Nitrogen oxides (NOx = NO + NO 2) Photochemistry Units of Atmospheric Concentration Atmospheric Lifetimes Mixing time vs residence time Atmospheric Emissions Sulfur Nitrogen oxides Ammonia Organic nitrogen Types of Atmospheric Reactions Kinetic approaches • Typical mixing ratios range from 1000 ppbv in rural areas to 0. 02 ppbv in remote environments like Amazon Forest, Mace Head (Ireland) or Cape Grim, Tasmania Photochemical Reactions • Among the most important molecules in atmospheric chemistry Thermodynamic approaches Free Radical Reactions 28 12/12/2021 • Major sources: Antropogenic, fossil fuel combustion in Troposphere and reaction of N 2 O in Stratosphere Atmospheric nitrogen CHEM 196 Petrucci

Structure and Chemical Composition Photochemistry Units of Atmospheric Concentration Atmospheric Lifetimes Mixing time vs residence time Atmospheric Emissions Sulfur Nitrogen oxides Ammonia Organic nitrogen Types of Atmospheric Reactions Thermodynamic approaches Kinetic approaches Photochemical Reactions Free Radical Reactions

Structure and Chemical Composition Photochemistry Units of Atmospheric Concentration Atmospheric Lifetimes Ammonia • Primary basic gas in the atmosphere • most abundant after N 2 and N 2 O Mixing time vs residence time Atmospheric Emissions Sulfur Nitrogen oxides Ammonia Organic nitrogen Types of Atmospheric Reactions Thermodynamic approaches Kinetic approaches Photochemical Reactions Free Radical Reactions 30 12/12/2021 • Typical mixing ratios between 0. 1 and 10 ppbv over continents • Sinks: Wet (precipitation or wet surfaces) and dry (to aerosol or surfaces) deposition Atmospheric nitrogen CHEM 196 Petrucci

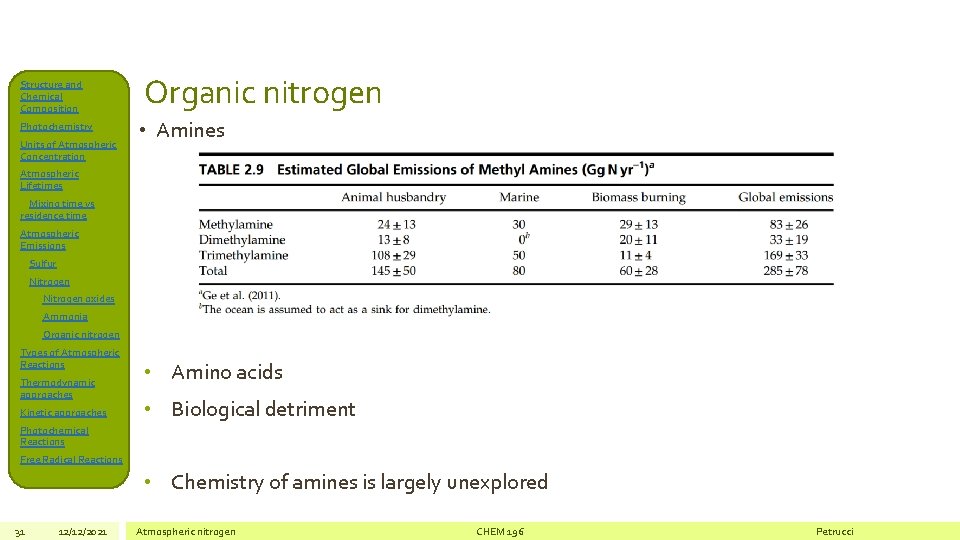
Structure and Chemical Composition Photochemistry Units of Atmospheric Concentration Organic nitrogen • Amines Atmospheric Lifetimes Mixing time vs residence time Atmospheric Emissions Sulfur Nitrogen oxides Ammonia Organic nitrogen Types of Atmospheric Reactions Thermodynamic approaches Kinetic approaches • Amino acids • Biological detriment Photochemical Reactions Free Radical Reactions • Chemistry of amines is largely unexplored 31 12/12/2021 Atmospheric nitrogen CHEM 196 Petrucci

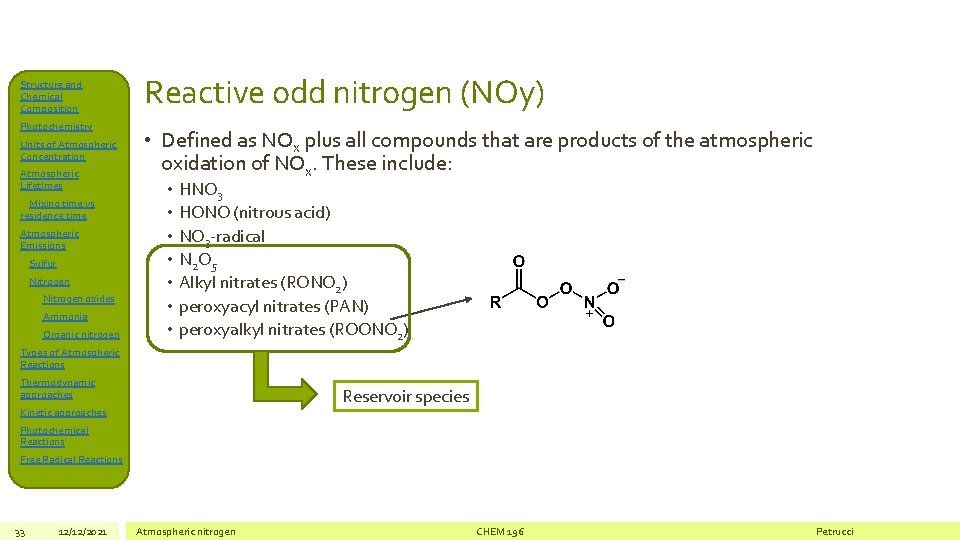
Structure and Chemical Composition Photochemistry Units of Atmospheric Concentration Atmospheric Lifetimes Mixing time vs residence time Atmospheric Emissions Sulfur Nitrogen oxides Ammonia Organic nitrogen Reactive odd nitrogen (NOy) • Defined as NOx plus all compounds that are products of the atmospheric oxidation of NOx. These include: • • HNO 3 HONO (nitrous acid) NO 3 -radical N 2 O 5 Alkyl nitrates (RONO 2) peroxyacyl nitrates (PAN) peroxyalkyl nitrates (ROONO 2) Types of Atmospheric Reactions Thermodynamic approaches Reservoir species Kinetic approaches Photochemical Reactions Free Radical Reactions 33 12/12/2021 Atmospheric nitrogen CHEM 196 Petrucci

Structure and Chemical Composition Photochemistry Units of Atmospheric Concentration Types of Atmospheric Reactions • Reactions where thermodynamic calculations are appropriate Atmospheric Lifetimes • Reactions where kinetic considerations are important Mixing time vs residence time • Free radical reactions Atmospheric Emissions Sulfur Nitrogen oxides Ammonia Organic nitrogen Types of Atmospheric Reactions Thermodynamic approaches Kinetic approaches Photochemical Reactions Free Radical Reactions 35 12/12/2021 Types of atmospheric reactions CHEM 196 Petrucci

Thermodynamic approaches Structure and Chemical Composition • Photochemistry Units of Atmospheric Concentration Atmospheric Lifetimes Mixing time vs residence time Atmospheric Emissions Sulfur Nitrogen oxides Ammonia Organic nitrogen Types of Atmospheric Reactions Thermodynamic approaches Kinetic approaches Photochemical Reactions Free Radical Reactions 36 12/12/2021 Types of atmospheric reactions CHEM 196 Petrucci

Structure and Chemical Composition Photochemistry Units of Atmospheric Concentration Atmospheric Lifetimes • However, there is also a positive entropy change associated with this reaction, so we must consider the temperature of the reaction as well Mixing time vs residence time Atmospheric Emissions Sulfur Nitrogen oxides Assuming a car cylinder temperature of 2770 K, Ammonia Organic nitrogen Types of Atmospheric Reactions Thermodynamic approaches Kinetic approaches Although this is still positive (ie. , not spontaneous to the right), we can estimate the equilibrium constant for the reaction from Photochemical Reactions Free Radical Reactions 37 12/12/2021 Types of atmospheric reactions CHEM 196 Petrucci

Structure and Chemical Composition Photochemistry Units of Atmospheric Concentration • Now, we can estimate the mixing ratio of NO in the cylinder from the Kp: Atmospheric Lifetimes Mixing time vs residence time Atmospheric Emissions Sulfur Nitrogen oxides Ammonia • Assume that after combustion, there remains a PO 2= 1. 0 k. Pa and PN 2= 650 k. Pa (remember, N 2 doesn’t participate in the combustion). • x k. Pa of O 2 react with x k. Pa of N 2 to form 2 x k. Pa of NO (assuming IGL behavior) Organic nitrogen Types of Atmospheric Reactions Thermodynamic approaches Kinetic approaches Photochemical Reactions • Plugging into Kp expression and solving for “x” then gives x = 0. 66 k. Pa and so PNO = 2 x = 1. 32 k. Pa • In the cylinder then, the mixing ratio of NO is Free Radical Reactions 38 12/12/2021 Types of atmospheric reactions CHEM 196 Petrucci

Structure and Chemical Composition • Photochemistry Units of Atmospheric Concentration Atmospheric Lifetimes We calculated this earlier Mixing time vs residence time Atmospheric Emissions Sulfur Review intro chem Nitrogen oxides Ammonia Organic nitrogen Types of Atmospheric Reactions Thermodynamic approaches Kinetic approaches Photochemical Reactions Free Radical Reactions 39 12/12/2021 Types of atmospheric reactions CHEM 196 Petrucci

Structure and Chemical Composition Photochemistry Units of Atmospheric Concentration Atmospheric Lifetimes Mixing time vs residence time Atmospheric Emissions Sulfur Nitrogen oxides • Measured atmospheric NO mixing ratios are typically around 100 ppbv, more than 9 oom than we just estimated! • Recall that thermodynamic approaches are only valid when the chemical system approaches or reaches a true equilibrium • Equilibrium in an engine piston is reached quickly, • In the atmosphere, where Temp, pressure and concentrations are low, equilibrium is almost never reached Ammonia Organic nitrogen Types of Atmospheric Reactions Thermodynamic approaches Kinetic approaches Photochemical Reactions Free Radical Reactions 40 12/12/2021 Types of atmospheric reactions CHEM 196 Petrucci

Structure and Chemical Composition Photochemistry Units of Atmospheric Concentration Atmospheric Lifetimes Mixing time vs residence time Kinetic approaches • Let’s look at the NO system when equilibrium is not reached, and kinetic issues become important. • If there remains significant NO in the atmosphere, then the reverse of the formation reaction must be extremely slow: Atmospheric Emissions Sulfur Nitrogen oxides • The temperature dependent 2 nd order rate constant for this reaction has been measured experimentally to be Ammonia Organic nitrogen Types of Atmospheric Reactions Thermodynamic approaches Kinetic approaches Photochemical Reactions So, at 25 o. C, k 2 = 4. 3 x 10 -41 m 3 mol-1 s-1 – Extremely small • We can now use this rate constant to estimate the rate of decomposition of NO in the atmosphere according to this reaction Free Radical Reactions 41 12/12/2021 Types of atmospheric reactions CHEM 196 Petrucci

Structure and Chemical Composition Photochemistry Units of Atmospheric Concentration Atmospheric Lifetimes Mixing time vs residence time Atmospheric Emissions Sulfur • NOTE: Let’s keep in mind that, while the use of mixing ratios is convenient when talking about atmospheric “concentrations, ” we must use “chemical concentrations” when dealing with rates of reactions. • Using the IGL, we can convert the partial pressure of NO to a chemical concentration (mol m-3) Nitrogen oxides Ammonia Organic nitrogen Types of Atmospheric Reactions • Now, the rate of decomposition of NO can be estimated as Thermodynamic approaches Kinetic approaches Photochemical Reactions • We can also estimate the atmospheric half life of NO to be Free Radical Reactions 42 12/12/2021 Types of atmospheric reactions CHEM 196 Petrucci

Structure and Chemical Composition Photochemistry Units of Atmospheric Concentration • Atmospheric Lifetimes Mixing time vs residence time Atmospheric Emissions Sulfur Nitrogen oxides Ammonia Organic nitrogen Types of Atmospheric Reactions Thermodynamic approaches Kinetic approaches Photochemical Reactions Free Radical Reactions 43 12/12/2021 Types of atmospheric reactions CHEM 196 Petrucci

Structure and Chemical Composition Photochemistry Units of Atmospheric Concentration • If we use a kinetic approach, on the other hand, Atmospheric Lifetimes Mixing time vs residence time Atmospheric Emissions Sulfur Nitrogen oxides Ammonia Organic nitrogen Types of Atmospheric Reactions Thermodynamic approaches • Note: Here we used pressures of reactants because experimentally determined 3 rd order rate constant is also given in terms of pressure Kinetic approaches Photochemical Reactions Free Radical Reactions 44 12/12/2021 • Takeaway: For any given atmospheric chemical process, eithermodynamic or kinetic control is possible. Types of atmospheric reactions CHEM 196 Petrucci

Structure and Chemical Composition Photochemistry Units of Atmospheric Concentration Atmospheric Lifetimes Mixing time vs residence time Atmospheric Emissions Sulfur Nitrogen oxides Ammonia Organic nitrogen Photochemical Reactions • A variety of chemical processes in the atmosphere can lead to the formation of NO 2 (other than oxidation by O 2). • We might ask then, “Can decomposition of NO 2 lead to significant production of NO? ” • Absorption of solar radiation can provide the energy to stimulate atmospheric chemical reactions that would otherwise be very slow or inaccessible. • Photochemical reactions typically involve two fundamental steps: Types of Atmospheric Reactions hν M X+Y* t tan Kinetic approaches or s Photochemical Reactions XY* reac Thermodynamic approaches products Free Radical Reactions XY 45 12/12/2021 Types of atmospheric reactions CHEM 196 Petrucci

Structure and Chemical Composition Photochemistry Units of Atmospheric Concentration • Atmospheric Lifetimes Mixing time vs residence time Atmospheric Emissions Sulfur Nitrogen oxides Ammonia Organic nitrogen Types of Atmospheric Reactions Thermodynamic approaches Kinetic approaches Photochemical Reactions Free Radical Reactions 46 12/12/2021 Types of atmospheric reactions CHEM 196 Petrucci

Structure and Chemical Composition Photochemistry Units of Atmospheric Concentration Atmospheric Lifetimes • Back to our question of NO 2 dissociation as a source of atmospheric NO • NO 2 can decompose (bimolecularly) to form NO and O 2: Mixing time vs residence time Atmospheric Emissions Sulfur Nitrogen • The second order rate constant for this reaction is 12. 5 M-1 s-1 • Assuming an atmospheric [NO 2] of 50 ppb, the rate of decomposition is Nitrogen oxides Ammonia Organic nitrogen Types of Atmospheric Reactions Thermodynamic approaches Kinetic approaches • If we now consider the photochemical rate of dissociation Photochemical Reactions Free Radical Reactions • So, photodissociation rate is about 109 greater than thermal dissociation rate 47 12/12/2021 Types of atmospheric reactions CHEM 196 Petrucci

Free Radical Reactions Structure and Chemical Composition • Photochemistry Units of Atmospheric Concentration Atmospheric Lifetimes Mixing time vs residence time Atmospheric Emissions Sulfur Nitrogen oxides Ammonia Organic nitrogen Types of Atmospheric Reactions Thermodynamic approaches Kinetic approaches Photochemical Reactions Free Radical Reactions 48 12/12/2021 Types of atmospheric reactions CHEM 196 Petrucci

Structure and Chemical Composition Photochemistry Units of Atmospheric Concentration • Atmospheric Lifetimes Mixing time vs residence time Atmospheric Emissions Sulfur Nitrogen oxides Ammonia Organic nitrogen Types of Atmospheric Reactions Thermodynamic approaches Kinetic approaches Photochemical Reactions Free Radical Reactions 49 12/12/2021 Types of atmospheric reactions CHEM 196 Petrucci