Chemistry of Cells Has nothing to do with

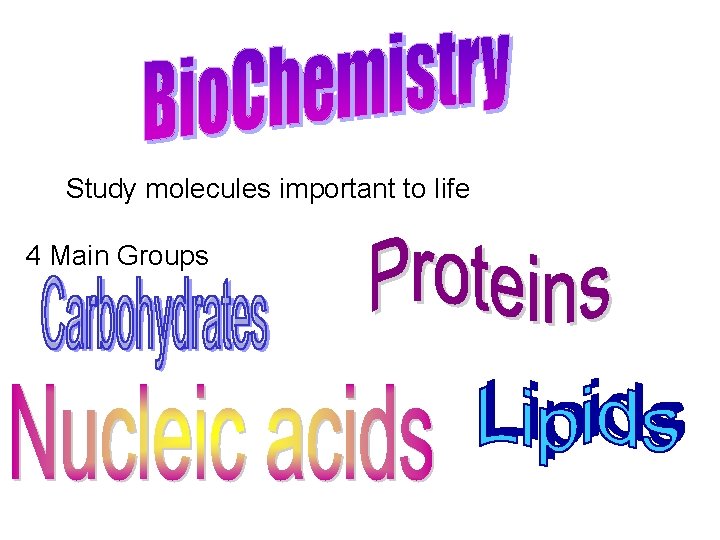








- Slides: 45

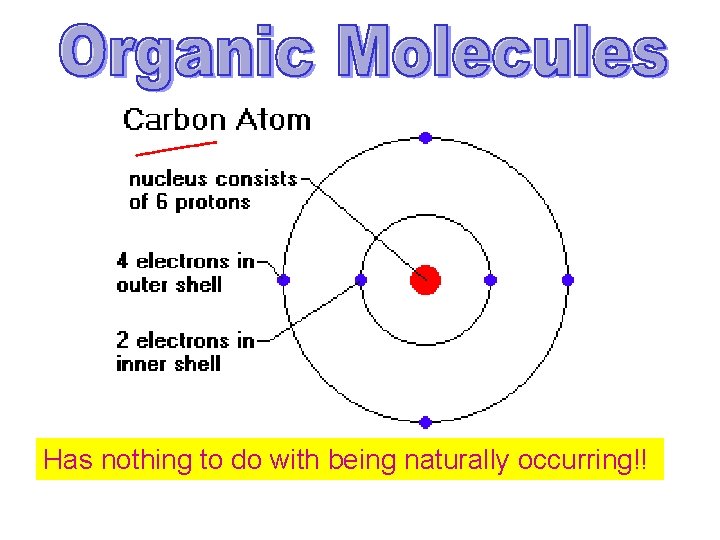
Chemistry of Cells

Has nothing to do with being naturally occurring!!

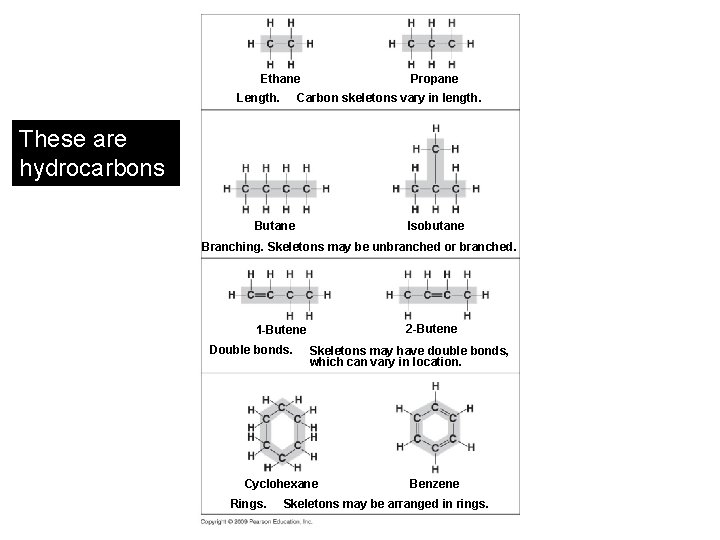
Propane Ethane Length. Carbon skeletons vary in length. These are hydrocarbons Isobutane Branching. Skeletons may be unbranched or branched. 2 -Butene 1 -Butene Double bonds. Skeletons may have double bonds, which can vary in location. Cyclohexane Rings. Benzene Skeletons may be arranged in rings.

Study molecules important to life 4 Main Groups

What elements do they contain? ? ? Carbon, Hydrogen, and Oxygen


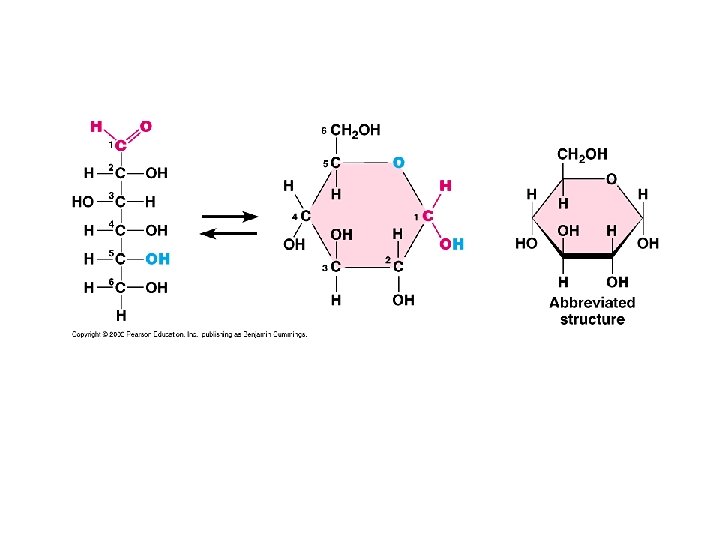
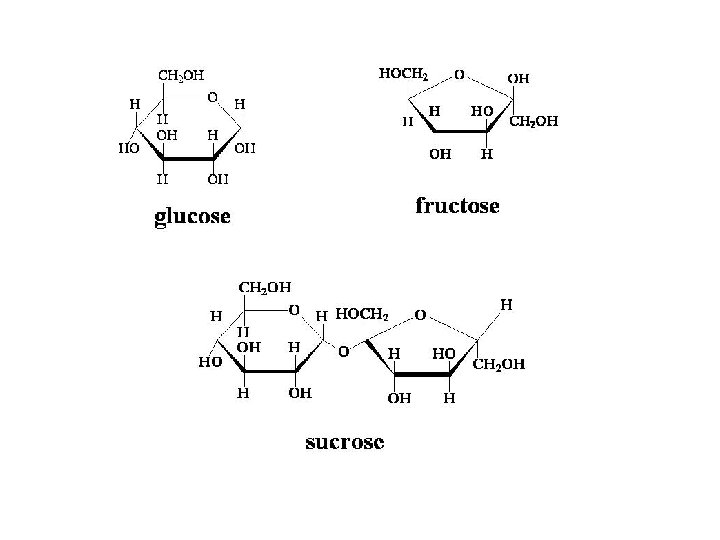
1) Monosaccharides (Simple sugars) Examples: -glucose BLOOD SUGAR -fructose FOUND IN FRUITS -Ribose found in RNA Function energy (readily available)

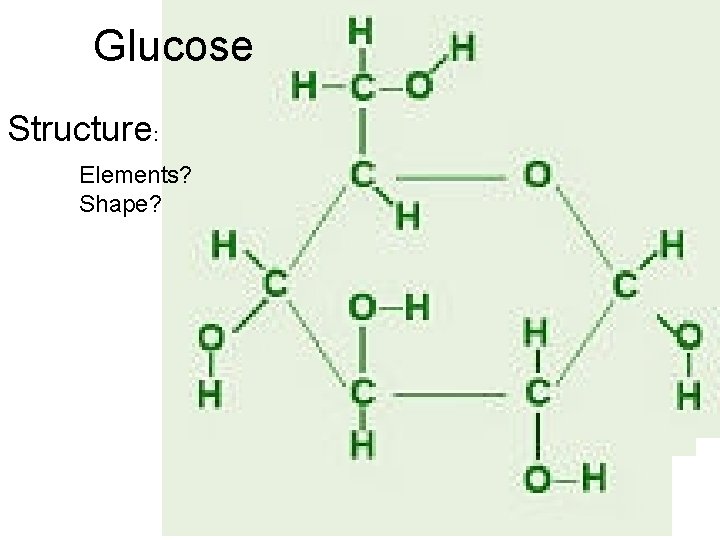
Glucose Structure: Elements? Shape?


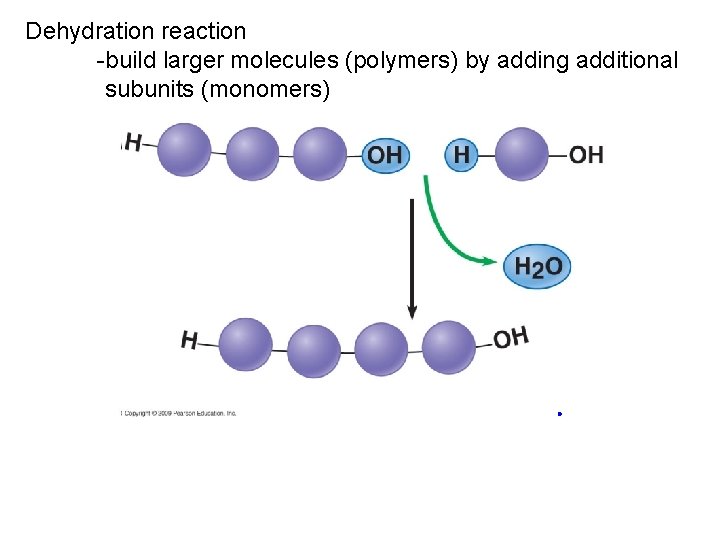
Dehydration reaction -build larger molecules (polymers) by adding additional subunits (monomers)

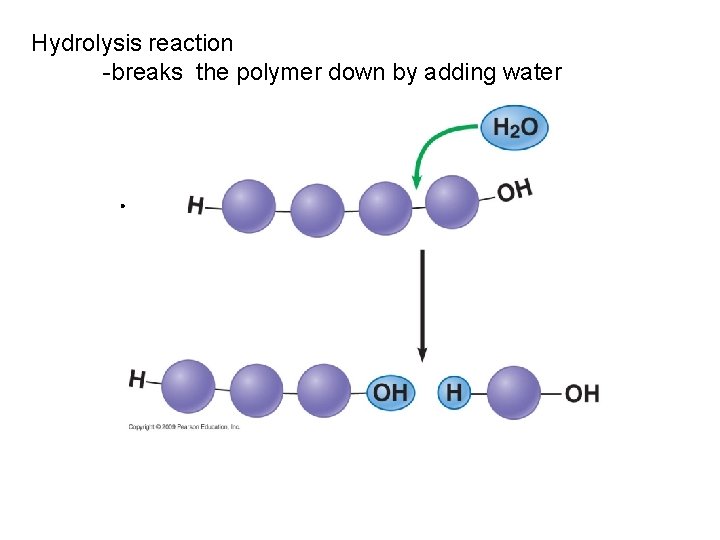
Hydrolysis reaction -breaks the polymer down by adding water

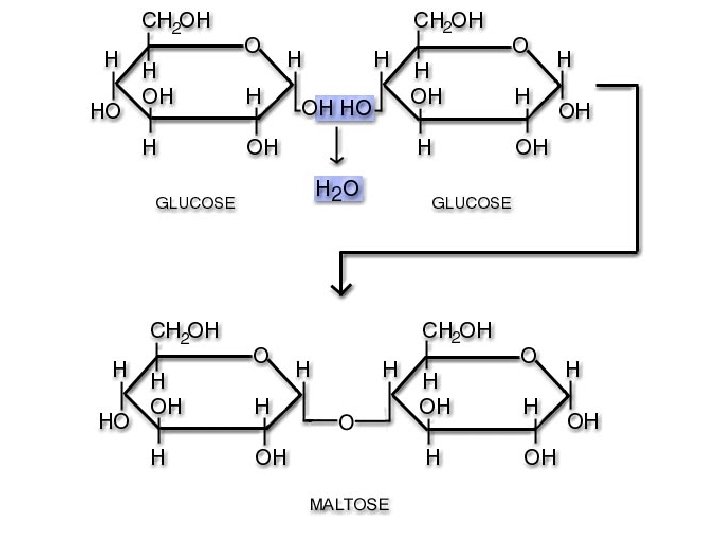
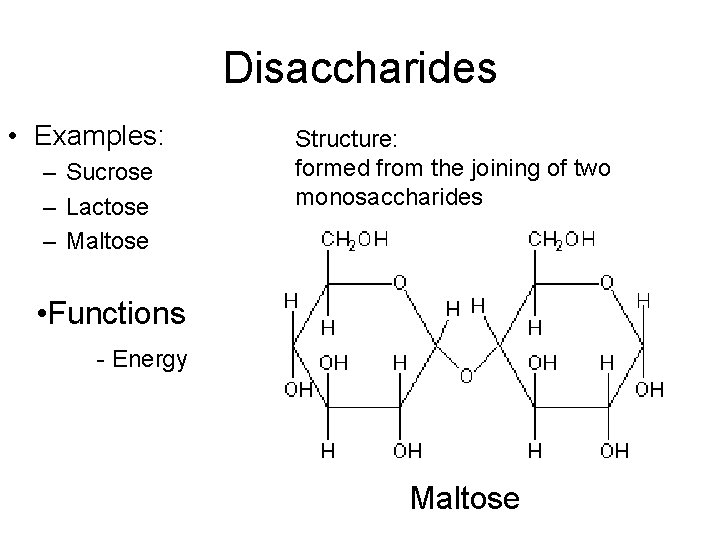
Disaccharides • Examples: – Sucrose – Lactose – Maltose Structure: formed from the joining of two monosaccharides • Functions - Energy Maltose


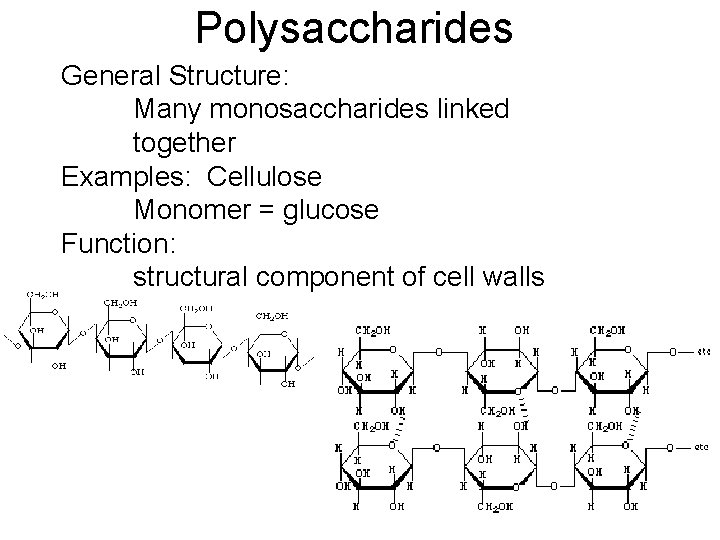
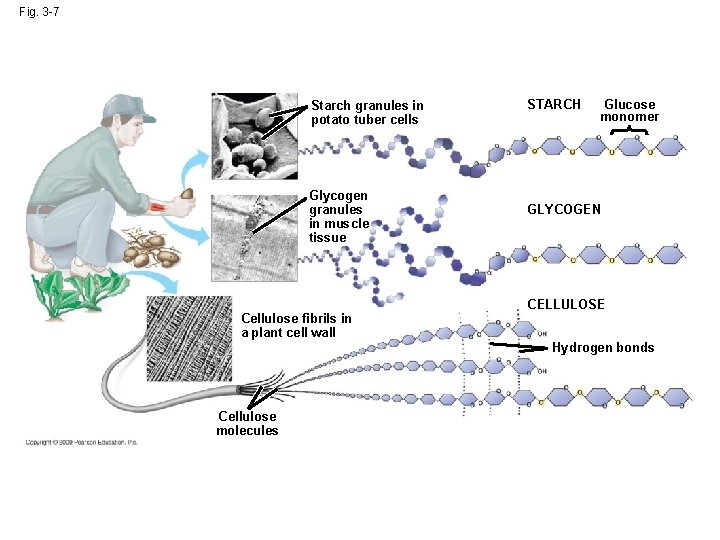
Polysaccharides General Structure: Many monosaccharides linked together Examples: Cellulose Monomer = glucose Function: structural component of cell walls

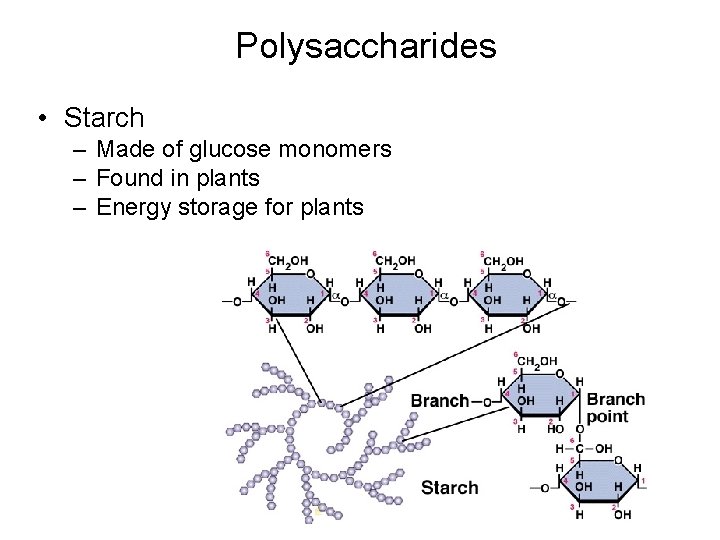
Polysaccharides • Starch – Made of glucose monomers – Found in plants – Energy storage for plants

Glycogen • Glucose monomers • How animals store glucose – In liver and muscles

Fig. 3 -7 Starch granules in potato tuber cells Glycogen granules in muscle tissue STARCH Glucose monomer GLYCOGEN CELLULOSE Cellulose fibrils in a plant cell wall Hydrogen bonds Cellulose molecules

Chitin • Found in the exoskeletons of arthropods • Function? ?

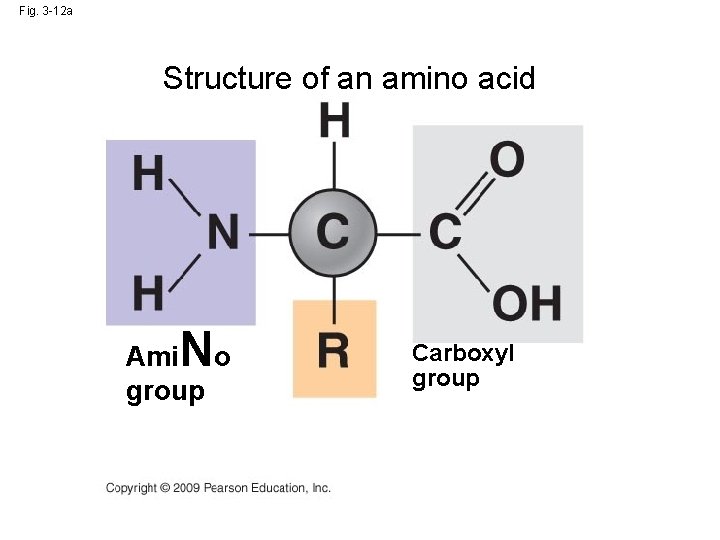
Proteins structure • Are polymers • Made from chains of amino acids • Linked by peptide bonds – Those bonds form through dehydration reaction

Fig. 3 -12 a Structure of an amino acid N Ami o group Carboxyl group

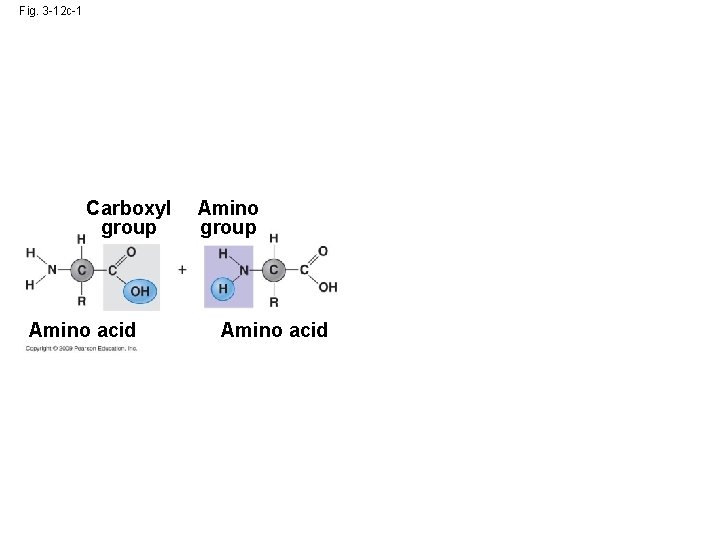
Fig. 3 -12 c-1 Carboxyl group Amino acid Amino group Amino acid

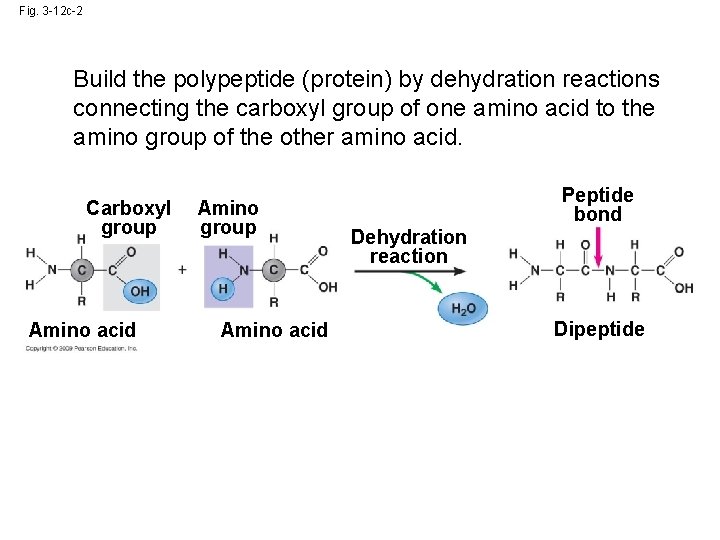
Fig. 3 -12 c-2 Build the polypeptide (protein) by dehydration reactions connecting the carboxyl group of one amino acid to the amino group of the other amino acid. Carboxyl group Amino acid Amino group Amino acid Peptide bond Dehydration reaction Dipeptide

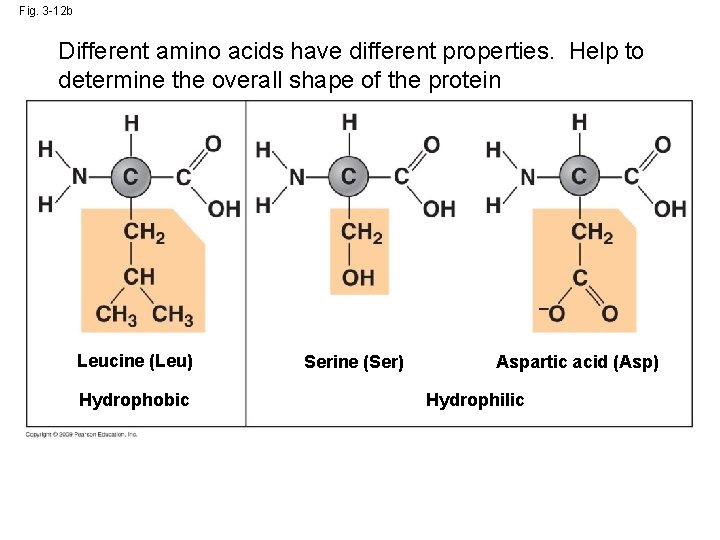
Fig. 3 -12 b Different amino acids have different properties. Help to determine the overall shape of the protein Leucine (Leu) Hydrophobic Serine (Ser) Aspartic acid (Asp) Hydrophilic

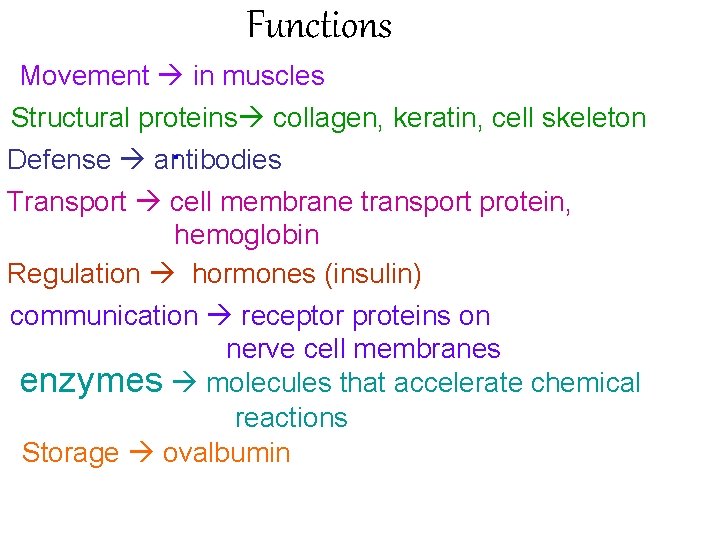
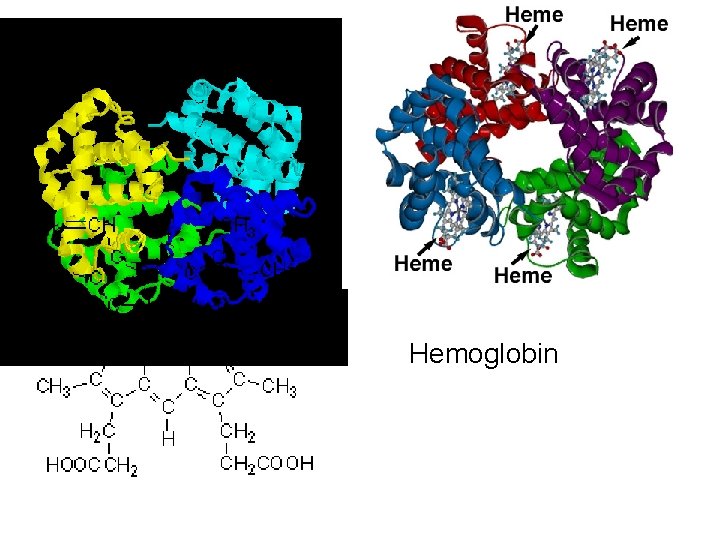
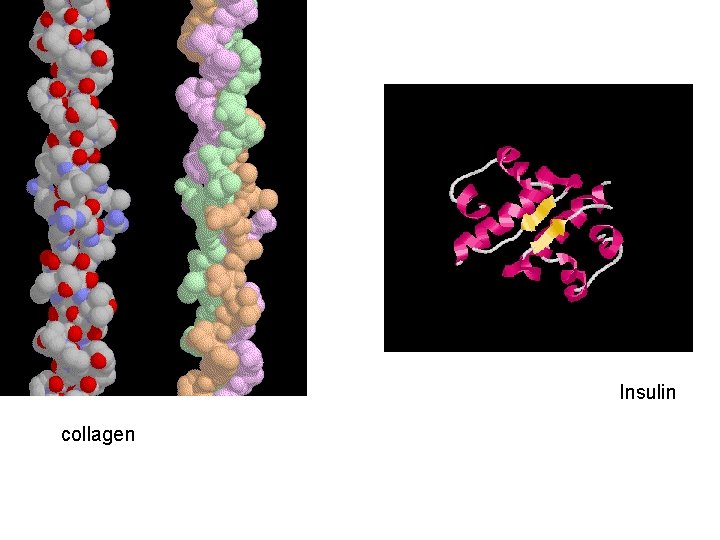
Functions Movement in muscles Structural proteins collagen, keratin, cell skeleton Defense antibodies Transport cell membrane transport protein, hemoglobin Regulation hormones (insulin) communication receptor proteins on nerve cell membranes enzymes molecules that accelerate chemical reactions Storage ovalbumin




Hemoglobin

Insulin collagen

actin myosin

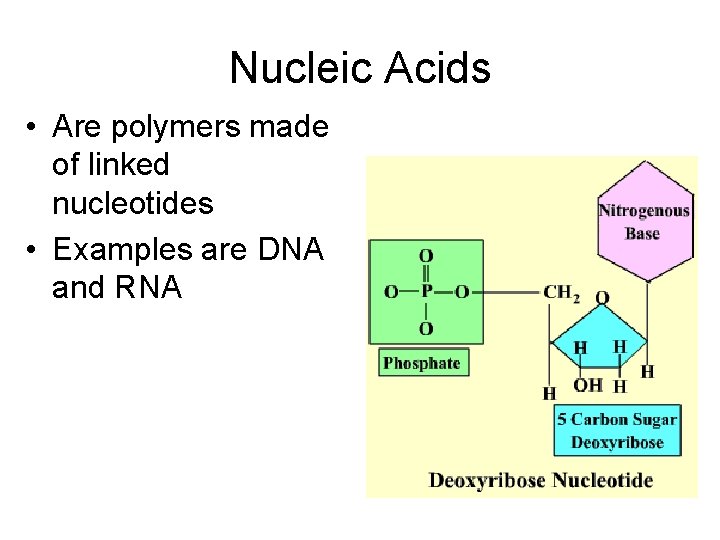
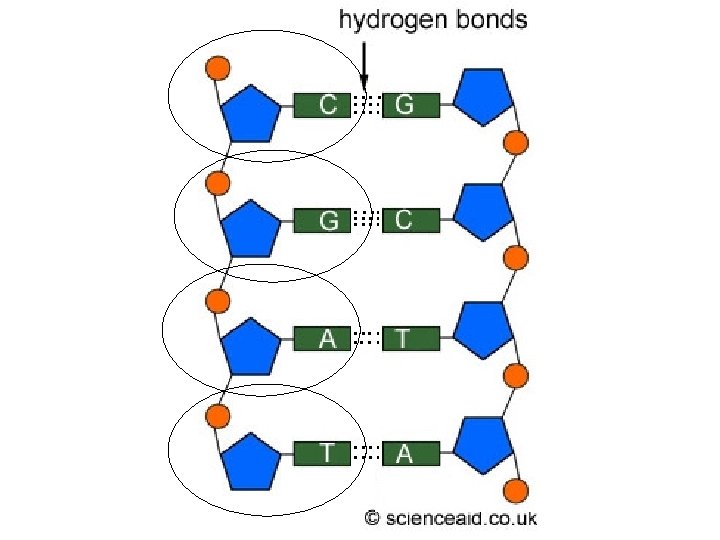
Nucleic Acids • Are polymers made of linked nucleotides • Examples are DNA and RNA


Fig. 3 -8 a

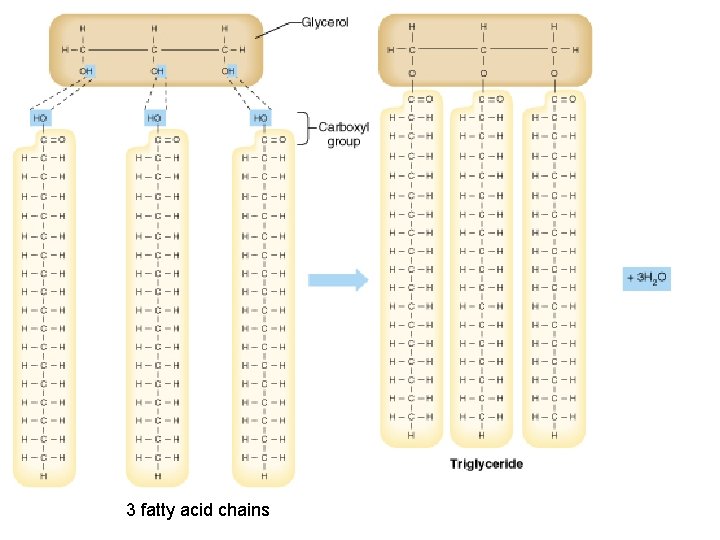
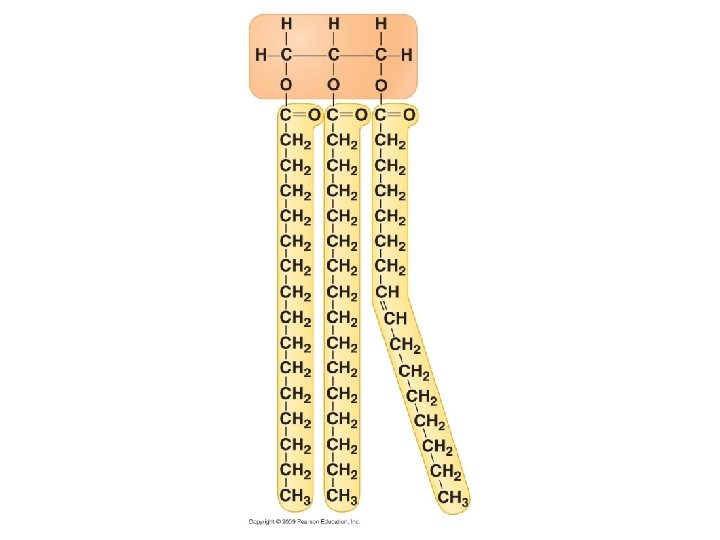
Lipids Include fats, phospholipids, and steroids • Diverse group of hydrophobic molecules General structure of a fat

3 fatty acid chains

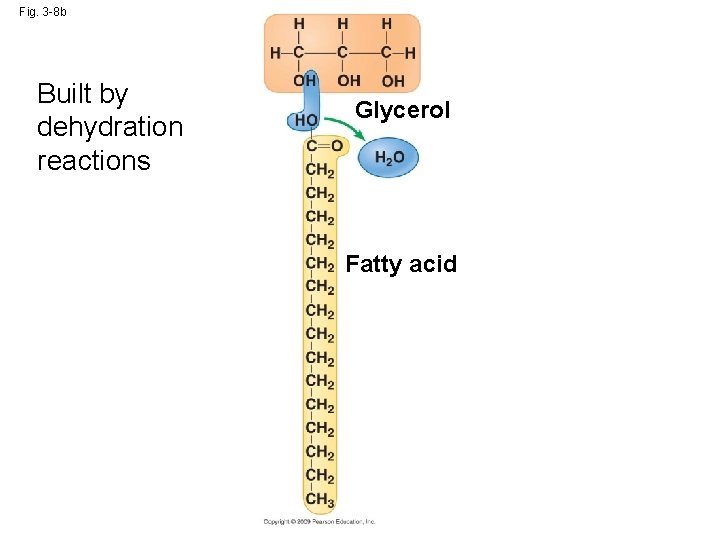
Fig. 3 -8 b Built by dehydration reactions Glycerol Fatty acid


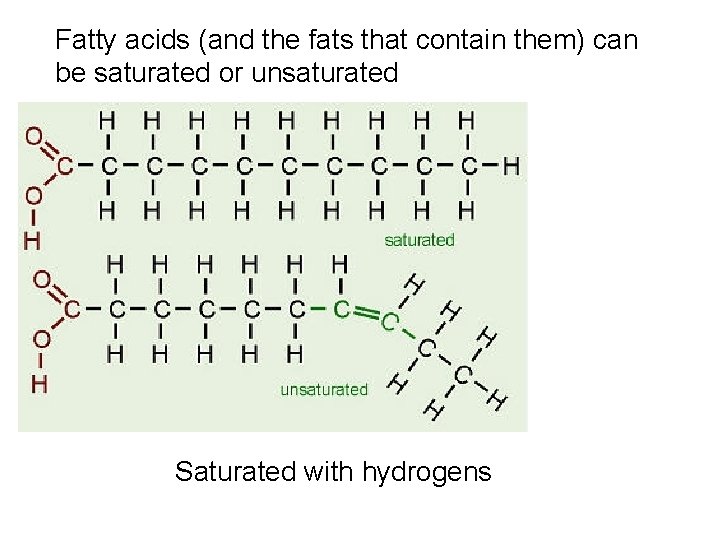
Fatty acids (and the fats that contain them) can be saturated or unsaturated Saturated with hydrogens

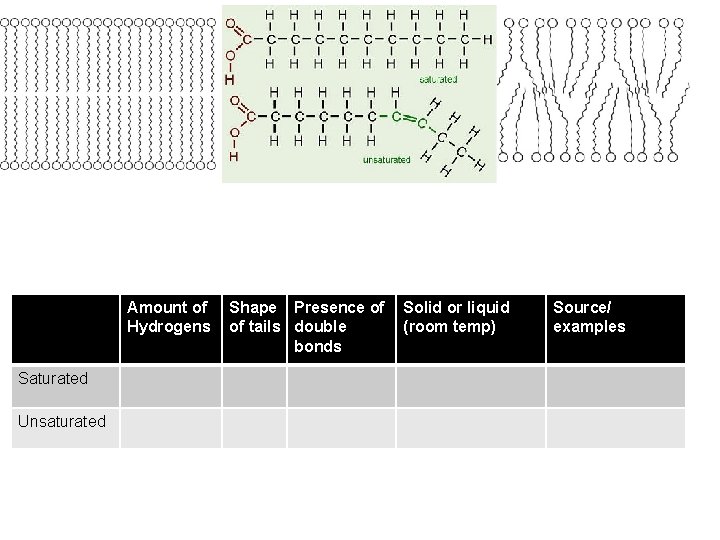
Amount of Hydrogens Saturated Unsaturated Shape Presence of of tails double bonds Solid or liquid (room temp) Source/ examples

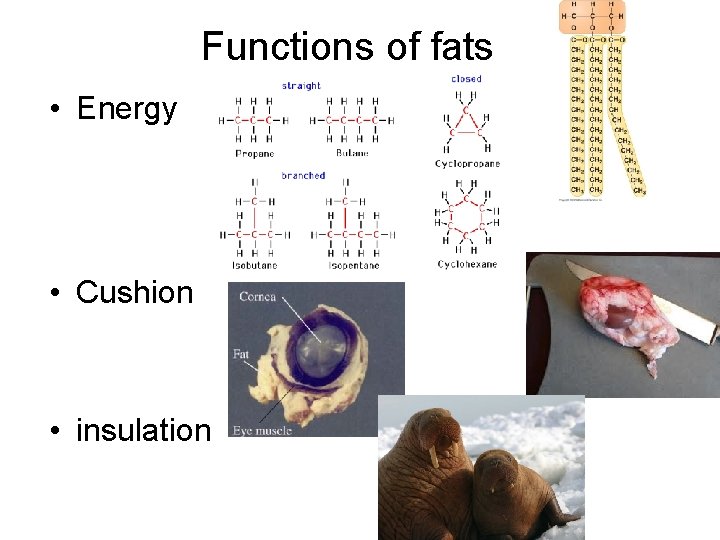
Functions of fats • Energy • Cushion • insulation

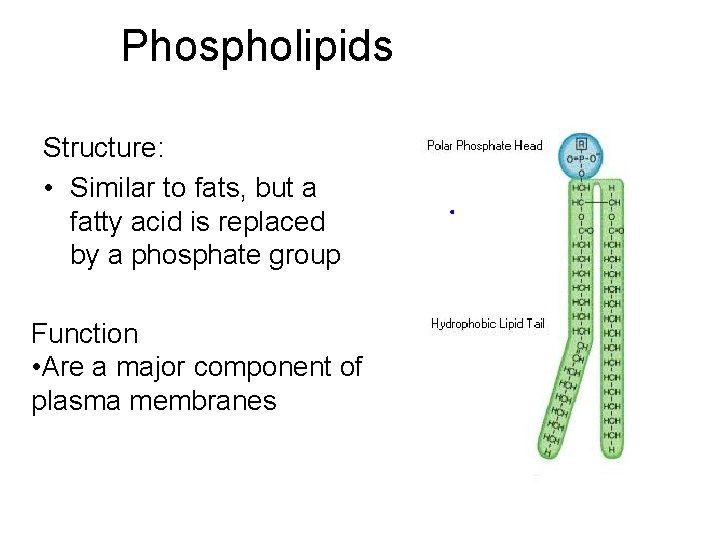
Phospholipids Structure: • Similar to fats, but a fatty acid is replaced by a phosphate group Function • Are a major component of plasma membranes

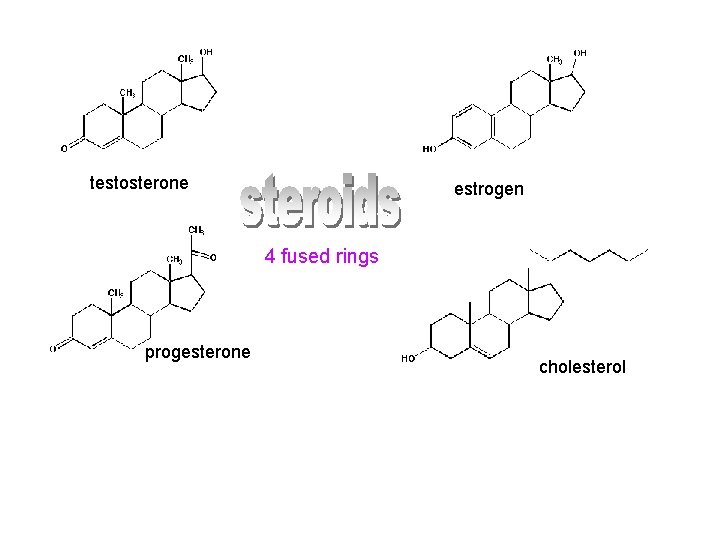
testosterone estrogen 4 fused rings progesterone cholesterol

Study molecules important to life 4 Main Groups