Chemistry Notes Miss Bergin 2010 2011 Substances can

Chemistry Notes Miss Bergin 2010 -2011 Substances can be classified in three principal categories: • solids, liquids or gases • elements (metals and non-metals), compounds and mixtures • acidic, neutral or basic. Matter is anything that takes up space and has mass. Matter is made up of very small particles (which we call molecules, atoms or ions). States of Matter: Solid, liquid and gas. Particle theory Molecules are always vibrating (jiggling). It is this movement that determines whether the object is a solid, a liquid or a gas. In a solid Molecules jiggle/vibrate around a fixed position but don’t actually move anywhere. In a liquid molecules they have more energy therefore vibrate faster, they are able to leave their fixed position and they can slide/flow over each other. In a gas molecules vibrate even faster, the molecules have enough energy to separate completely from each other and move off in all directions Melting Point: The melting point of a solid is the temperature at which both the solid and the liquid states exist together Boliing Point: The boiling point of a liquid is the temperature at which evaporation begins to occur throughout the liquid.
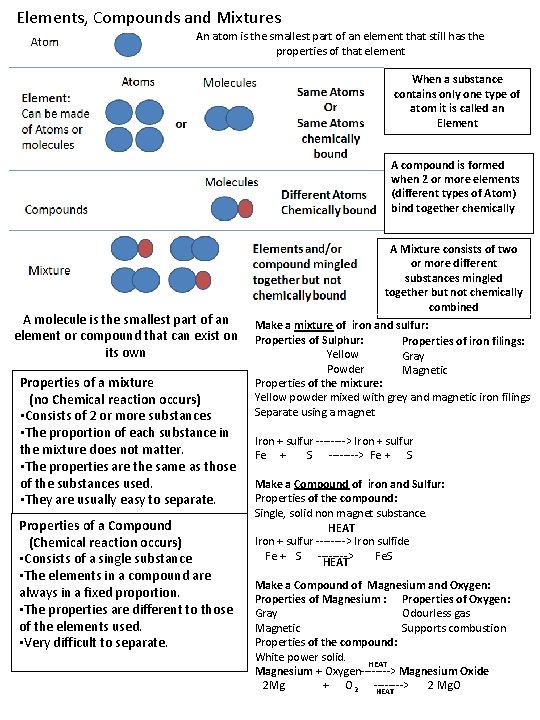
Elements, Compounds and Mixtures An atom is the smallest part of an element that still has the properties of that element When a substance contains only one type of atom it is called an Element A compound is formed when 2 or more elements (different types of Atom) bind together chemically A molecule is the smallest part of an element or compound that can exist on its own Properties of a mixture (no Chemical reaction occurs) • Consists of 2 or more substances • The proportion of each substance in the mixture does not matter. • The properties are the same as those of the substances used. • They are usually easy to separate. Properties of a Compound (Chemical reaction occurs) • Consists of a single substance • The elements in a compound are always in a fixed proportion. • The properties are different to those of the elements used. • Very difficult to separate. A Mixture consists of two or more different substances mingled together but not chemically combined Make a mixture of iron and sulfur: Properties of Sulphur: Properties of iron filings: Yellow Gray Powder Magnetic Properties of the mixture: Yellow powder mixed with grey and magnetic iron filings Separate using a magnet Iron + sulfur ----> Iron + sulfur Fe + S ----> Fe + S Make a Compound of iron and Sulfur: Properties of the compound: Single, solid non magnet substance. HEAT Iron + sulfur ----> Iron sulfide Fe + S ----> Fe. S HEAT Make a Compound of Magnesium and Oxygen: Properties of Magnesium : Properties of Oxygen: Odourless gas Gray Supports combustion Magnetic Properties of the compound: White power solid. HEAT Magnesium + Oxygen----> Magnesium Oxide 2 Mg + O 2 ----> 2 Mg. O HEAT
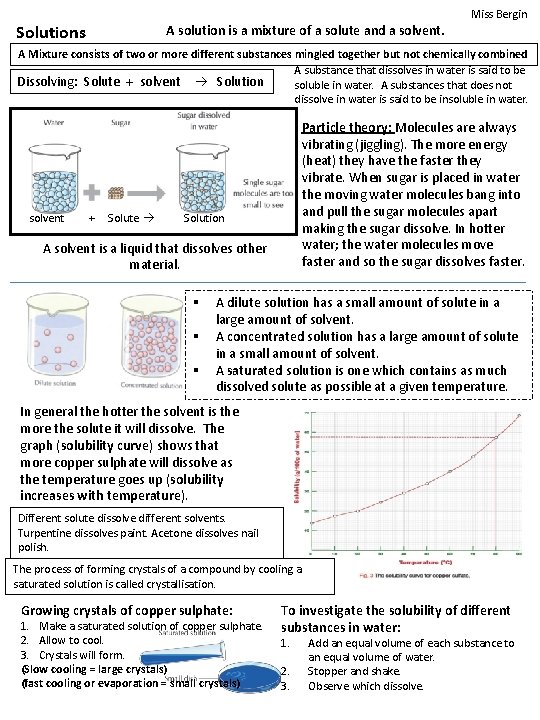
Miss Bergin Solutions A solution is a mixture of a solute and a solvent. A Mixture consists of two or more different substances mingled together but not chemically combined A substance that dissolves in water is said to be Dissolving: Solute + solvent Solution soluble in water. A substances that does not dissolve in water is said to be insoluble in water. solvent + Solute Particle theory: Molecules are always vibrating (jiggling). The more energy (heat) they have the faster they vibrate. When sugar is placed in water the moving water molecules bang into and pull the sugar molecules apart making the sugar dissolve. In hotter water; the water molecules move faster and so the sugar dissolves faster. Solution A solvent is a liquid that dissolves other material. § § § A dilute solution has a small amount of solute in a large amount of solvent. A concentrated solution has a large amount of solute in a small amount of solvent. A saturated solution is one which contains as much dissolved solute as possible at a given temperature. In general the hotter the solvent is the more the solute it will dissolve. The graph (solubility curve) shows that more copper sulphate will dissolve as the temperature goes up (solubility increases with temperature). Different solute dissolve different solvents. Turpentine dissolves paint. Acetone dissolves nail polish. The process of forming crystals of a compound by cooling a saturated solution is called crystallisation. Growing crystals of copper sulphate: 1. Make a saturated solution of copper sulphate. 2. Allow to cool. 3. Crystals will form. (Slow cooling = large crystals) (fast cooling or evaporation = small crystals) To investigate the solubility of different substances in water: 1. 2. 3. Add an equal volume of each substance to an equal volume of water. Stopper and shake. Observe which dissolve.
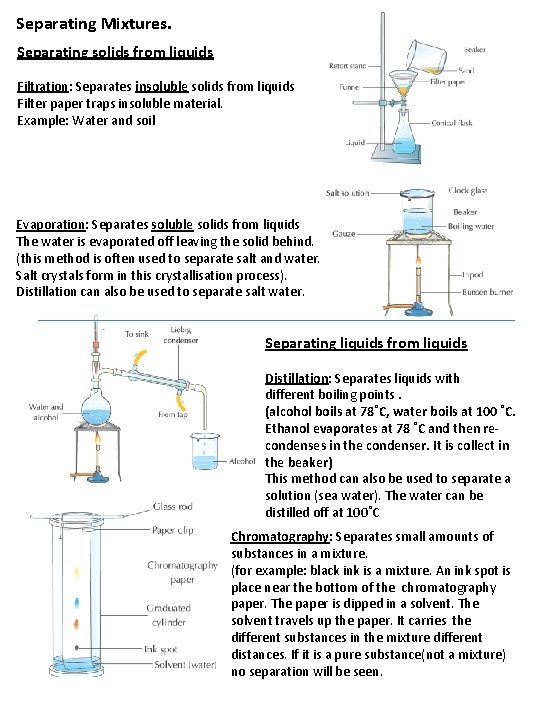
Separating Mixtures. Separating solids from liquids Filtration: Separates insoluble solids from liquids Filter paper traps insoluble material. Example: Water and soil Evaporation: Separates soluble solids from liquids The water is evaporated off leaving the solid behind. (this method is often used to separate salt and water. Salt crystals form in this crystallisation process). Distillation can also be used to separate salt water. Separating liquids from liquids Distillation: Separates liquids with different boiling points. (alcohol boils at 78˚C, water boils at 100 ˚C. Ethanol evaporates at 78 ˚C and then recondenses in the condenser. It is collect in the beaker) This method can also be used to separate a solution (sea water). The water can be distilled off at 100˚C Chromatography: Separates small amounts of substances in a mixture. (for example: black ink is a mixture. An ink spot is place near the bottom of the chromatography paper. The paper is dipped in a solvent. The solvent travels up the paper. It carries the different substances in the mixture different distances. If it is a pure substance(not a mixture) no separation will be seen.

Acids and Bases (An alkali is a base that dissolves in water) Common laboratory acids: Hydrochloric Acid HCl Sulfuric Acid H 2 SO 4 Common laboratory bases: Sodium Hydroxide Na. OH Calcium Hydroxide Ca(OH)2 Common acids: Carbonic acid in fizzy drinks Citric acid in fruit Ethanoic acid in vinegar All acids have a sharp sour taste. Common bases: Toothpaste Soap Window Cleaner Alkalis have a soapy feel. Corrosive An indicator is a substance that shows, by changing colour, whether a chemical is an acid or a base. Paper with litmus indicator soaked into it is called litmus paper. Blue litmus paper goes red in acid (Remember Acid-Red-Danger) Red litmus paper goes blue in an alkali Universal indicator paper can be used to find the p. H of a substance (A p. H meter or p. H sensor may also be used) A solution whose p. H is 7 is neutral. A solution whose p. H is < 7 is acidic. A solution whose p. H is > 7 is basic. Exp: How to measure the p. H of different substances • Set up apparatus shown in diagram. • Dip the universal indicator paper into each liquid. • Compare the colour of the universal indicator paper to the colour code provided. Litmus can only tell us if a substance is an acid or a base (alkali). Litmus cannot tell us how strong the acid or base is. The p. H scale is a scale used to measure the how acidic or a basic a substance is. It goes from 0 -14

The air and Oxygen The air is a mixture of gasses. Composition of the air Nitrogen (N 2)78% Oxygen (O 2) 21% Carbon dioxide (CO 2) and inert gases 1% Water vapour (H 2 O) variable depending on weather. • Water is the only substance that turns blue cobalt chloride paper pink • CO 2 is the only gas that turns limewater milky Physical properties of Oxygen. • Colourless, Odourless, Tasteless gas. . • Slightly soluble in water Chemical properties. • Relights glowing splint • Oxygen is neutral. To show that air contains water vapour 1. Set up apparatus shown. 2. The water vapour in the air condenses when it touches the cold test tube. 3. The blue cobalt chloride paper turns pink. 4. This proves that the air contains water vapour No change to litmus. • Combines with carbon to form carbon dioxide. C +O 2 -> C O 2 turns blue litmus red: Acid • Combines with magnesium to form magnesium oxide. 2 Mg +O 2 -> 2 Mg. O turns Red litmus blue: base Uses of oxygen. • Oxygen us needed for breathing. • Oxygen is needed for burning or combustion. When a substance combines with oxygen and releases heat this is called combustion. Fuel + O 2 -> Heat + C O 2 + H 2 O To show the presence of carbon dioxide CO 2 in the air 1. Set up apparatus shown in diagram. 2. The suction pump pulls the air from the room into the clear lime water. 3. The lime water soon turns milky white. To show that approximately one fifth of the air is oxygen 1. One syringe is filled to 100 cm 3 with air. 2. The copper in the tube is heated strongly. 3. The air is passed over the copper. 4. The copper reacts with the oxygen in the air to produce copper oxide. 5. 79% of the air remains. 21% of the air was oxygen. Copper + Oxygen Copper Oxide Preparation of oxygen gas O 2 1. Set up apparatus shown in diagram. 2. Open the tap of the funnel and let the Hydrogen Peroxide fall onto the Manganese Dioxide. 3. Oxygen gas forms and collects as shown. Manganese dioxide A catalyst speeds Hydrogen Peroxide -> Water + Oxygen up a chemical catalyst reaction but is not used up in the Mn 02 reaction 2 H 2 O 2 -> 2 H 2 O + O 2 catalyst
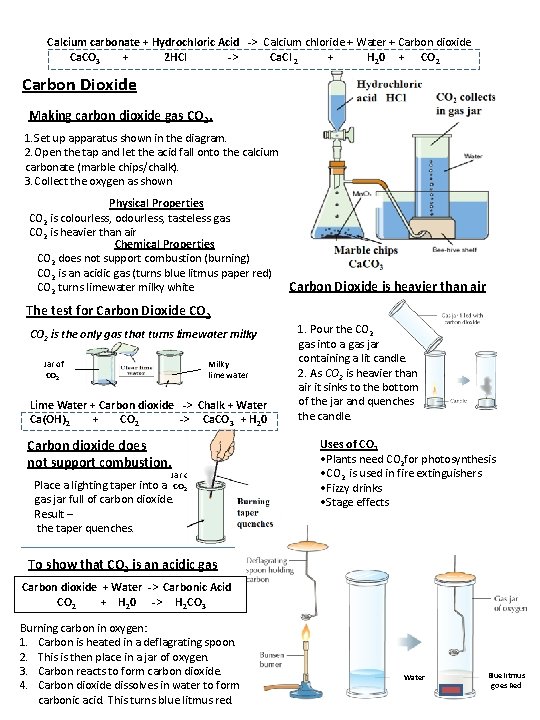
Calcium carbonate + Hydrochloric Acid -> Calcium chloride + Water + Carbon dioxide Ca. CO 3 + 2 HCl -> Ca. Cl 2 + H 20 + CO 2 Carbon Dioxide Making carbon dioxide gas CO 2. 1. Set up apparatus shown in the diagram. 2. Open the tap and let the acid fall onto the calcium carbonate (marble chips/chalk). 3. Collect the oxygen as shown Physical Properties CO 2 is colourless, odourless, tasteless gas. CO 2 is heavier than air Chemical Properties CO 2 does not support combustion (burning) CO 2 is an acidic gas (turns blue litmus paper red) CO 2 turns limewater milky white Carbon Dioxide is heavier than air The test for Carbon Dioxide CO 2 is the only gas that turns limewater milky Jar of CO 2 Milky lime water Lime Water + Carbon dioxide -> Chalk + Water Ca(OH)2 + CO 2 -> Ca. CO 3 + H 20 Carbon dioxide does not support combustion. Jar of Place a lighting taper into a CO 2 gas jar full of carbon dioxide. Result – the taper quenches. 1. Pour the CO 2 gas into a gas jar containing a lit candle. 2. As CO 2 is heavier than air it sinks to the bottom of the jar and quenches the candle. Uses of CO 2 • Plants need CO 2 for photosynthesis • CO 2 is used in fire extinguishers • Fizzy drinks • Stage effects To show that CO 2 is an acidic gas Carbon dioxide + Water -> Carbonic Acid CO 2 + H 20 -> H 2 CO 3 Burning carbon in oxygen: 1. Carbon is heated in a deflagrating spoon. 2. This is then place in a jar of oxygen. 3. Carbon reacts to form carbon dioxide. 4. Carbon dioxide dissolves in water to form carbonic acid. This turns blue litmus red. Water Blue litmus goes Red

Water is made of the elements Hydrogen and Oxygen. Water is the most common compound on earth. Properties of water. • Pure water boils at 100 0 C • Pure water freezes at 00 C • Water is the only liquid that expands as it freezes (water freezes from the top down). • Water is an excellent solvent. • Ice is less dense that water therefore it floats • Density of water is 1 g/cm 3 The water cycle: How water is recycled on the planet. Testing for water: Blue cobalt chloride paper turns pink in the presence of water. • The meniscus is the curved dip found on the surface of water in a vessel. • The meniscus is formed because water is attracted to the sides of the vessel it is placed into. • When reading the level of water in a vessel you read the bottom of the meniscus and at eye level 1. Evaporation of water from oceans. 2. Water vapour condenses into small droplets. 3. When these droplets cool more they fall as rain. 4. Rain travels in rivers to the oceans. . Filtration -this removes silt and clay Steps involved in water treatment. There are 5 steps used in the treatment of water: 1. Screening -removal of large floating objects. 2. Settling -left in tanks so that sand etc can fall to bottom 3. Filtration -this removes silt and clay 4. Chlorination -this kills bacteria in the water. 5. Fluoridation - this prevents tooth decay. Soft water is water that forms a lather easily with soap. Hard water is water that does not form a lather with soap easily. Hardness is caused by the presence of calcium ions dissolved in the water. Limestone (containing calcium carbonate) + Rainwater -> Calcium ions in solution (acidic) (Hard water) Advantages of hard water. • It tastes nice to drink. • It contains calcium for healthy bones. • It is used in the making of beer, and for tanning leather. Advantages of soft water. • As it forms a lather easily less soap is needed • It does not clog pipes and elements of kettles with limescale. How rainwater becomes hard water: • Falling rain dissolves some CO 2 and becomes slightly acidic • The rain dissolves some calcium carbonate in limestone rocks. • The dissolved calcium ions enters the rivers etc. with the rainwater. Disadvantages of hard water. • It wastes soap. • It deposits limescale in pipes and kettles when boiled. Disadvantages of soft water. • It does not taste nice to drink. • It does not provide calcium for building bones.
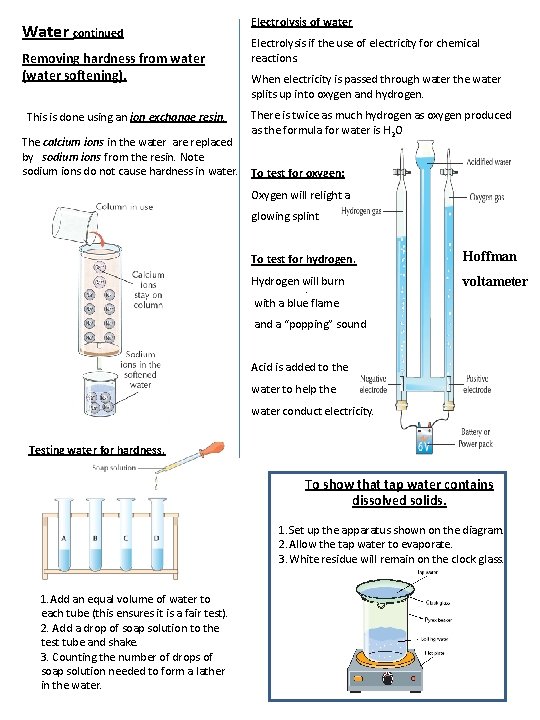
Water continued Removing hardness from water (water softening). This is done using an ion exchange resin. The calcium ions in the water are replaced by sodium ions from the resin. Note sodium ions do not cause hardness in water. Electrolysis of water Electrolysis if the use of electricity for chemical reactions. When electricity is passed through water the water splits up into oxygen and hydrogen. There is twice as much hydrogen as oxygen produced as the formula for water is H 2 O To test for oxygen: Oxygen will relight a glowing splint To test for hydrogen. Hoffman Hydrogen will burn. with a blue flame voltameter and a “popping” sound Acid is added to the water to help the water conduct electricity. Testing water for hardness. To show that tap water contains dissolved solids. 1. Set up the apparatus shown on the diagram. 2. Allow the tap water to evaporate. 3. White residue will remain on the clock glass. 1. Add an equal volume of water to each tube (this ensures it is a fair test). 2. Add a drop of soap solution to the test tube and shake. 3. Counting the number of drops of soap solution needed to form a lather in the water.

Acids and Bases (An alkali is a base that dissolves in water) Common laboratory acids: Hydrochloric Acid HCl Sulfuric Acid H 2 SO 4 Neutralisation. • When an acid reacts with a base in the correct proportions salt and water are formed. • This type of reaction is called a neutralisation reaction. Common laboratory bases: • The p. H of the salt and water produced is neutral. Sodium Hydroxide Na. OH Calcium Hydroxide. Lime Water Titration Experiment: To neutralise an acid with a base to produce salt and water 1. Setup apparatus shown in diagram. 2. Measure an exact volume of the base Na. OH into the flask using the pipette (base in the basement). 3. Add some litmus indicator to the flask. 4. Fill the burette to the zero mark with the acid HCl (acid in the attic) 5. Open the tap of the burette and slowly add the acid to the base swirling to mix contents. 6. Continue adding acid to base until the indicator just changes colour. This is the volume of acid needed to neutralise the base. 7. Repeat the neutralisation reaction using the same volumes but omitting the indicator. Ca(OH)2 Pipette Reactions of acids: Acid and base: Acid + Base -> Salt + Water Hydrochloric Acid + Sodium Hydroxide -> Sodium Chloride + Water HCl + Na. OH -> Na. Cl + H 2 O Reactions of acids: Acid and Carbonate: As used for CO 2 production Acid + Hydrochloric Acid + 2 HCl + Carbonate -> Salt + Water + Carbon dioxide Calcium carbonate -> Calcium chloride + Water + Carbon dioxide Ca. CO 3 -> Ca. Cl 2 + H 20 Reactions of acids: Acid and metal: Acid + Hydrochloric Acid 2 HCl + Metal -> + Zinc -> Zn Salt + Hydrogen Zinc chloride + Hydrogen -> Zn. Cl 2 + H 2 + CO 2

Fossil fuels and acid rain. A fuel is any substance that burns in oxygen to produce heat. Fuel + O 2 -> Heat + C O 2 + H 2 O Fossil fuels are the remains of plants and animals that lived millions of years ago. Fossil fuels contain the elements Carbon (C), Hydrogen (H), and Oxygen(O). Fossil fuels are called Hydrocarbons because they contain the elements Hydrogen and Carbon Burning is a chemical reaction. When a substance combines with oxygen and releases heat this is called combustion. Fuel + O 2 -> Heat + C O 2 + H 2 O CO 2 + H 2 O are wastes produced of combustion. We burn fossil fuels to heat our homes, to cook our food and to power our cars, factories, airplanes etc. Acid rain is very damaging to the environment: 1. Acid rain kills trees. 2. Acid rain causes the rivers to become acidic this kills fish and plants in waterways. 3. Acid rain corrodes limestone buildings and metal structure like bridges. Examples of fossil fuels. Coal, oil, gas and turf are fossil fuels. Methane (CH 4 ) is a fossil fuel often called natural gas. When methane (CH 4) burns it produces only CO 2 and H 2 O and heat energy. Methane + oxygen -> Heat + C O 2 + H 2 O CH 4 + 2 O 2 -> Heat + C O 2 +2 H 2 O Coal and oil also contain sulfur (S) which produces sulfur dioxide (SO 2) when you burn them. Coal + oxygen -> Heat + C O 2 + H 2 O + SO 2 Oil+ oxygen -> Heat + C O 2 + H 2 O + SO 2 sulfur dioxide combines with rainwater to form acid rain. sulfur dioxide + rain water -> Sulfurous acid SO 2 + H 2 O -> H 2 SO 3 Controlling acid rain. All cars, lorries , trains planes and factory chimneys are fitted with catalytic convertors these remove the harmful poisonous gasses from the exhaust fumes before they are released into the air. To show C O 2 and H 2 O are formed when hydrocarbons are burned 1. Set up the apparatus as shown. 2. The burner uses methane gas (CH 4). 3. Ice is added to aid condensation. Cobalt chloride paper is used to test for water. 4. Limewater is used to test for CO 2
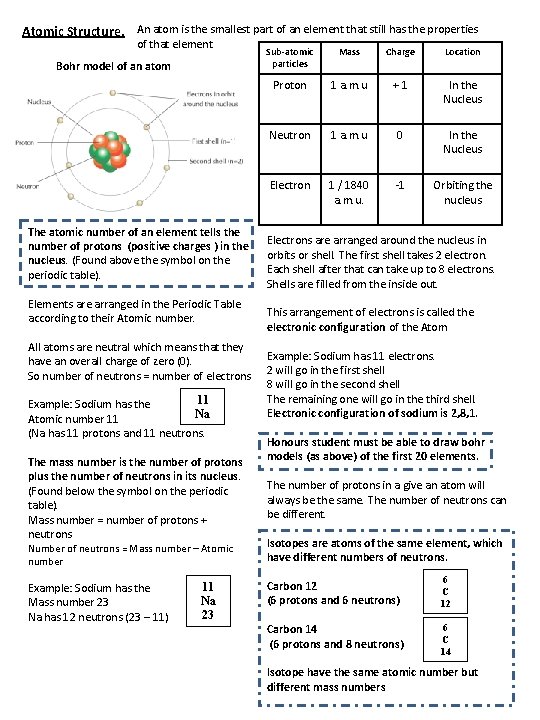
Atomic Structure. An atom is the smallest part of an element that still has the properties of that element Bohr model of an atom The atomic number of an element tells the number of protons (positive charges ) in the nucleus. (Found above the symbol on the periodic table). Elements are arranged in the Periodic Table according to their Atomic number. All atoms are neutral which means that they have an overall charge of zero (0). So number of neutrons = number of electrons 11 Example: Sodium has the Na Atomic number 11 (Na has 11 protons and 11 neutrons. The mass number is the number of protons plus the number of neutrons in its nucleus. (Found below the symbol on the periodic table). Mass number = number of protons + neutrons Number of neutrons = Mass number – Atomic number Example: Sodium has the Mass number 23 Na has 12 neutrons (23 – 11) 11 Na 23 Sub-atomic particles Mass Charge Location Proton 1 a. m. u +1 In the Nucleus Neutron 1 a. m. u 0 In the Nucleus Electron 1 / 1840 a. m. u. -1 Orbiting the nucleus Electrons are arranged around the nucleus in orbits or shell. The first shell takes 2 electron. Each shell after that can take up to 8 electrons. Shells are filled from the inside out. This arrangement of electrons is called the electronic configuration of the Atom Example: Sodium has 11 electrons. 2 will go in the first shell 8 will go in the second shell The remaining one will go in the third shell. Electronic configuration of sodium is 2, 8, 1. Honours student must be able to draw bohr models (as above) of the first 20 elements. The number of protons in a give an atom will always be the same. The number of neutrons can be different. Isotopes are atoms of the same element, which have different numbers of neutrons. Carbon 12 (6 protons and 6 neutrons) 6 C 12 Carbon 14 (6 protons and 8 neutrons) 6 C 14 Isotope have the same atomic number but different mass numbers
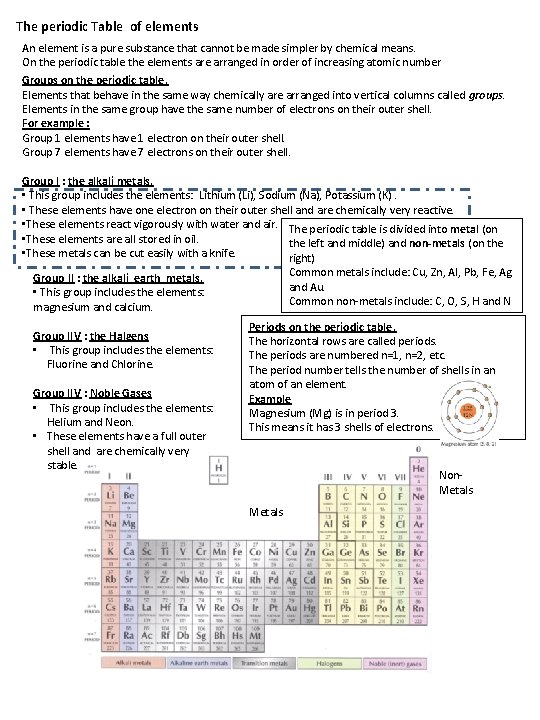
The periodic Table of elements An element is a pure substance that cannot be made simpler by chemical means. On the periodic table the elements are arranged in order of increasing atomic number Groups on the periodic table. Elements that behave in the same way chemically are arranged into vertical columns called groups. Elements in the same group have the same number of electrons on their outer shell. For example : Group 1 elements have 1 electron on their outer shell. Group 7 elements have 7 electrons on their outer shell. Group I : the alkali metals. • This group includes the elements: Lithium (Li), Sodium (Na), Potassium (K). • These elements have one electron on their outer shell and are chemically very reactive. • These elements react vigorously with water and air. The periodic table is divided into metal (on • These elements are all stored in oil. the left and middle) and non-metals (on the • These metals can be cut easily with a knife. right) Common metals include: Cu, Zn, Al, Pb, Fe, Ag and Au. Common non-metals include: C, O, S, H and N Group II : the alkali earth metals. • This group includes the elements: magnesium and calcium. Group IIV : the Halgens • This group includes the elements: Fluorine and Chlorine. Group IIV : Noble Gases • This group includes the elements: Helium and Neon. • These elements have a full outer shell and are chemically very stable. Periods on the periodic table. The horizontal rows are called periods. The periods are numbered n=1, n=2, etc. The period number tells the number of shells in an atom of an element. Example Magnesium (Mg) is in period 3. This means it has 3 shells of electrons. Non. Metals

Chemical Bonding A chemical bond holds atoms together in a molecule When Atoms combine they follow rules. In HCl Here will always be 1 H with 1 Cl = HCl All atoms are neutral which means that they have an overall charge of zero (0). So number of neutrons = number of electrons Elements that have 8 electrons in their outer shell are very unreactive. Eg. The Noble gases (Group VIII) Octet rule: when bonding occurs, atoms tend to reach an electron arrangement with 8 electrons in the outer shell. • • • An ionic bond is formed when atoms give or take electrons. An ionic bond is the force of attraction between oppositely-charged ions in a molecule. An ion is a charged atom or a charged group of atoms. When an atom looses electrons it comes positively charged. (it has lost negative charge) When an atom gains electrons it comes negatively charged. (it has gained negative charge ) 11 Na The electronic configuration of sodium is 2, 8, 1 Sodium losses its outer electron To become a sodium ion (Na +) The electronic configuration of Chlorine is 2, 8, 7 Chlorine gains this electron To become a Chlorine ion (Cl-) 17 Cl A sodium atom gives its outer electron to a chlorine atom to form Na. Cl: An ionic bond has formed and both now have a full outer shell. In the same way, a magnesium atom forms an ionic bond with an oxygen atom by giving it its 2 outer electrons. The ionic compound magnesium oxide (Mg. O) is formed.
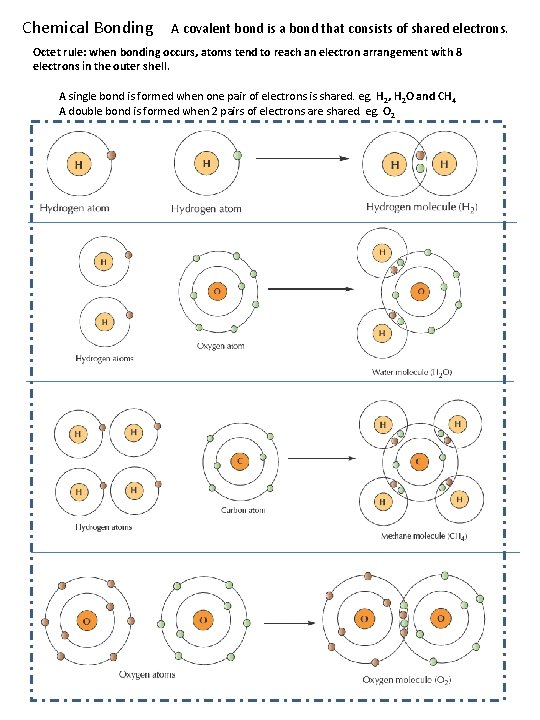
Chemical Bonding A covalent bond is a bond that consists of shared electrons. Octet rule: when bonding occurs, atoms tend to reach an electron arrangement with 8 electrons in the outer shell. A single bond is formed when one pair of electrons is shared. eg. H 2, H 2 O and CH 4 A double bond is formed when 2 pairs of electrons are shared. eg. O 2

Ionic and covalent compounds: Non- Metals Ionic bonds are formed between metals and non-metals Ionic bonds consist of many oppositely charged ions held together strongly to form a crystal lattice. A covalent substance is made up of separate single molecules. The attraction between the individual molecules is fairly week. crystal lattices. Properties of Ionic compounds. (Na. Cl and Mg. O) 1. 2. 3. 4. 5. Consist of crystal lattices. They form crystalline solids High melting and boiling points. They usually dissolve in water. They conduct electricity when dissolved in water. Properties of covalent compounds. (H 2, H 2 O, CH 4 and O 2 1. 2. 3. 4. 5. Consist of separate molecules Covalent compounds are usually liquids and gasses. Covalent compounds have low melting and boiling points. Covalent compounds are usually insoluble in water. Covalent compounds do not conduct electricity. To investigate if ionic and covalent compound conduct electricity. 1. Set up apparatus shown in diagram. 2. Place some covalent compounds in the beaker. (e. g. distilled water, paraffin oil, methylated sprits) 3. Result: The bulb will not light because covalent compounds cannot conduct electricity. 1. Place some ionic compounds (e. g. sodium chloride, copper sulphate, sodium hydroxide) in the beaker. Result: The bulb will light because ionic compounds can conduct electricity.

Chemical Formulas and chemical equations. A chemical formula consists of the symbols of the elements in the molecule. e. g H 2 O contains the elements Hydrogen and oxygen. A chemical formula also indicates the ratio of the different atoms in a compound. e. g H 2 O contains 2 Hydrogen atoms and 1 oxygen atom. Ion formation: Group I. Happy to loss 1 electron to become positive ions. Electronic configuration H Li Na K 1 2, 8, 8, 1 H+ Li + Na+ K+ Group II. Happy to loss 2 electron to become positive ions. Electronic configuration Be Mg Ca 2, 2 2, 8, 8, 2 Group VII. Happy to gain an electron to become negative ions. Electronic configuration F Cl 2, 7 2, 8, 7 FCl - Group VI. Happy to gain 2 electron to become negative ions. Electronic configuration O s 2, 6 2, 8, 6 O 2 S 2 - Calculating formulas of ionic compounds For Sodium Chloride: Ions: Na+ and Cl - (one positive and one negative = neutral) So Sodium Chloride is Na. Cl For Calcium Chloride: Ions: Ca 2+ and Cl - (two positive needs 2 negatives = neutral) So Calcium Chloride is Ca. Cl 2 Be 2+ Mg 2+ Ca 2+ Balancing chemical equations. Chemical formula do not change. e. g. in Fe. O there is always one atom of iron (Fe) bound to one atom of oxygen (O). In a chemical reaction atoms cannot vanish or appear from nowhere. The atoms may now be in new substances but all the atoms at the start of the reaction must still exist at the end of the reaction. In other words the equation must balance. Example Fe + O 2 -> Fe. O (have one atom of Fe and 2 atoms of oxygen on the left; one atom of Fe and one atom of O on the right) – not balanced Cannot change Fe. O. If we add a 2 before Fe. O we get 2 Fe. O. The 2 indicates 2 molecules of Fe. O formed Fe + O 2 -> 2 Fe. O (have one atom of Fe and 2 atoms of oxygen on the left; 2 atoms of Fe and 2 atom of O on the right) – not balanced. If we add a 2 before Fe on the left. 2 Fe + O 2 -> 2 Fe. O (have 2 atoms of Fe and 2 atoms of oxygen on the left; 2 atoms of Fe and 2 atoms of O on the right) –balanced In rusting (Fe. O formation) 2 atoms of Fe combine with one molecule of oxygen to give 2 molecules of Fe. O
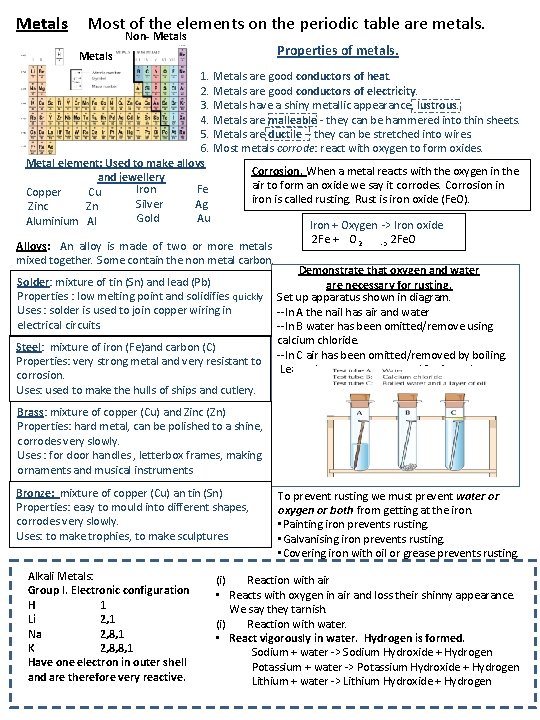
Metals Most of the elements on the periodic table are metals. Non- Metals Properties of metals. Metals 1. Metals are good conductors of heat. 2. Metals are good conductors of electricity. 3. Metals have a shiny metallic appearance, lustrous. 4. Metals are malleable - they can be hammered into thin sheets. 5. Metals are ductile – they can be stretched into wires. 6. Most metals corrode: react with oxygen to form oxides. Metal element: Used to make alloys Corrosion. When a metal reacts with the oxygen in the and jewellery air to form an oxide we say it corrodes. Corrosion in Iron Fe Copper Cu iron is called rusting. Rust is iron oxide (Fe. O). Silver Ag Zinc Zn Gold Au Aluminium Al Iron + Oxygen -> Iron oxide Alloys: An alloy is made of two or more metals mixed together. Some contain the non metal carbon. Solder: mixture of tin (Sn) and lead (Pb) Properties : low melting point and solidifies quickly Uses : solder is used to join copper wiring in electrical circuits. Steel: mixture of iron (Fe)and carbon (C) Properties: very strong metal and very resistant to corrosion. Uses: used to make the hulls of ships and cutlery. 2 Fe + O 2 -> 2 Fe. O Demonstrate that oxygen and water are necessary for rusting. Set up apparatus shown in diagram. --In A the nail has air and water --In B water has been omitted/remove using calcium chloride. --In C air has been omitted/removed by boiling. Leave the apparatus to stand for 1 week Brass: mixture of copper (Cu) and Zinc (Zn) Properties: hard metal, can be polished to a shine, corrodes very slowly. Uses : for door handles , letterbox frames, making ornaments and musical instruments Bronze: mixture of copper (Cu) an tin (Sn) Properties: easy to mould into different shapes, corrodes very slowly. Uses: to make trophies, to make sculptures. Alkali Metals: Group I. Electronic configuration H 1 Li 2, 1 Na 2, 8, 1 K 2, 8, 8, 1 Have one electron in outer shell and are therefore very reactive. To prevent rusting we must prevent water or oxygen or both from getting at the iron. • Painting iron prevents rusting. • Galvanising iron prevents rusting. • Covering iron with oil or grease prevents rusting. (i) Reaction with air • Reacts with oxygen in air and loss their shinny appearance. We say they tarnish. (i) Reaction with water. • React vigorously in water. Hydrogen is formed. Sodium + water -> Sodium Hydroxide + Hydrogen Potassium + water -> Potassium Hydroxide + Hydrogen Lithium + water -> Lithium Hydroxide + Hydrogen
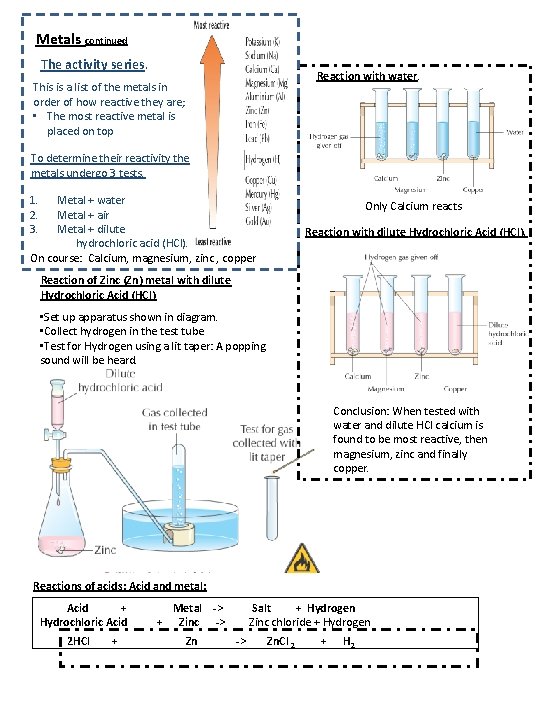
Metals continued The activity series. Reaction with water. This is a list of the metals in order of how reactive they are; • The most reactive metal is placed on top To determine their reactivity the metals undergo 3 tests. 1. 2. 3. Metal + water Metal + air Metal + dilute hydrochloric acid (HCl). On course: Calcium, magnesium, zinc , copper Only Calcium reacts Reaction with dilute Hydrochloric Acid (HCl). Reaction of Zinc (Zn) metal with dilute Hydrochloric Acid (HCl) • Set up apparatus shown in diagram. • Collect hydrogen in the test tube • Test for Hydrogen using a lit taper: A popping sound will be heard. Conclusion: When tested with water and dilute HCl calcium is found to be most reactive, then magnesium, zinc and finally copper. Reactions of acids: Acid and metal: Acid + Hydrochloric Acid 2 HCl + Metal -> + Zinc -> Zn Salt + Hydrogen Zinc chloride + Hydrogen -> Zn. Cl 2 + H 2

Plastics Plastic is a man made material. Plastics are produced from crude oil. There are many different kinds of plastics Types of plastics: polythene, nylon, PVC (poly vinylchloride), polystyrene and perspex. Properties of plastics. 1. 2. 3. 4. 5. 6. 7. 8. Plastics are lightweight. Plastics are durable. Plastics are waterproof. Plastics are easy to clean. Plastics are easily moulded. Plastics are cheap to produce. Plastics can be very strong. Plastics can be very flexible. Properties of perspex. Perspex is a clear , lightweight, tough plastic Uses of perspex. Perspex is used for the “glass” in watchs, light covers on cars, protective shields, Perspex is used to make pens and rulers. Properties of nylon. Nylon can be formed into fibres. Nylon is very strong, very hardwearing and is resistant to water and lots of chemicals. Nylon is used for: Making bristles on brushes. , fabric and clothes. Making guitar strings, ropes and rainwear. • PVC (Poly-Vinyl-Chloride) properties Easily moulded into different shapes. Doesn’t corrode and can be cleaned easily. It is also long lasting. Uses of PVC Window frames, Piping and guttering Impact of plastics on the environment. Most plastics bio-degrade very slowly. Plastic drinks bottles can take 700 years to bio degrade. Plastics when they are put into landfill stay there for a long time and cause the land fill sites to fill up very quickly. This means new sites have to be found. There is a limit to the amount of land that can be used for land fill. Plastic waste. To reduce the amount of plastics going into land fill we are now recycling them. This is good for the environment (litter and landfill). Recycling plastics saves oil. Recycled plastic can be used to produce fencing, furniture, pathways etc. Chemical reaction are very useful to us in everyday life. Burning is a chemical reaction. We burn fossil fuels to heat our homes, to cook our food and to power our cars, factories, airplanes etc. Chemists produce a wide variety of drugs for treating headaches and cancer etc. Fertilisers for growing crops can be produced by a chemical process. Chemicals called pesticides are used to kill pests on food crops.
- Slides: 20