Chemistry Notes Chapter 2 Hydrogen Bonding animation http

Chemistry Notes Chapter 2

Hydrogen Bonding animation: • http: //www. classzone. com/cz/books/bio_09/reso urces/htmls/animated_biology/unit 1/bio_ch 02_0 041_ab_bonding. html

Energy and chemical reactions: • http: //www. classzone. com/cz/books/bio_09/reso urces/htmls/animated_biology/unit 1/bio_ch 02_0 052_ab_exoendo. html

Atoms and bonding: • http: //www. classzone. com/cz/books/bio_09/reso urces/htmls/animated_biology/unit 1/bio_ch 02_0 059_ab_atombond. html

I. Nature of Matter A. What is Matter? • Matter= anything that has mass and takes up space (has volume). B. What is NOT matter? • Light • Sound • Electricity

Think About it: What is matter? Give some examples of what is matter and what is Not matter.

C. Composition of Matter • The ATOM is the basic “building block” (or unit) of all matter. • What was the basic Unit of Life? • Atoms are the smallest units of matter.

II. The Atom Anatomy • Protons = + • Neutrons = 0 (neutral) • Electrons = • Nucleus = Protons and Neutrons • Electron Cloud (shells, orbital)= Electrons surrounding nucleus • Valence Electrons = electrons on outermost valence of atom. • Only protons and neutrons have substantial mass (= 1 AMU) So they make up an atoms weight or mass. Electrons have negligible mass
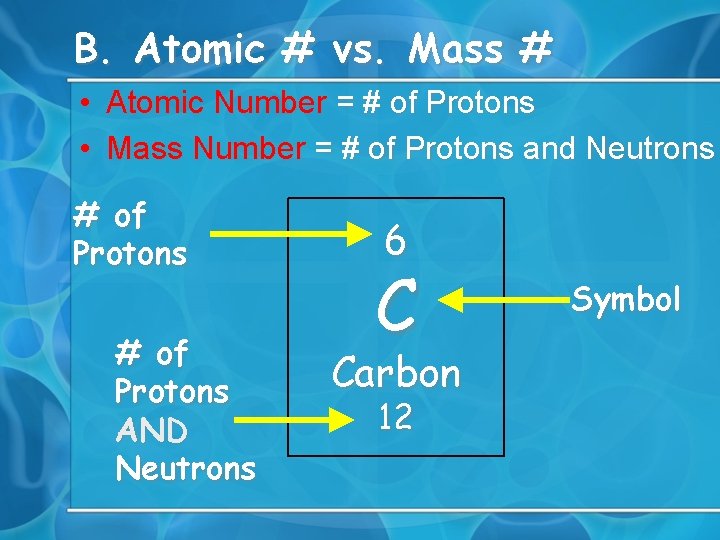
B. Atomic # vs. Mass # • Atomic Number = # of Protons • Mass Number = # of Protons and Neutrons # of Protons AND Neutrons 6 C Carbon 12 Symbol
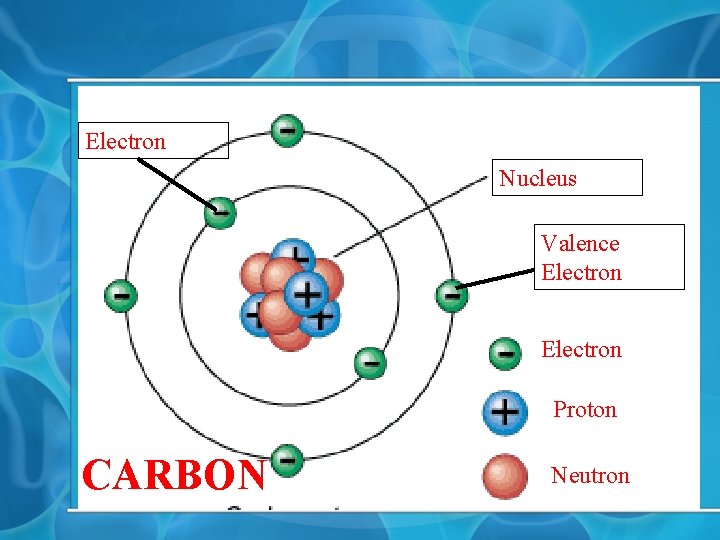
Electron Nucleus Valence Electron Proton CARBON Neutron

III. Elements A. Elements consist of entirely one type of atom. B. The # of protons determines the type of element. (Look at your table) C. Organized on the Periodic Table of Elements in order of increasing Atomic #. WHY? ?

What particles are found in the nucleus of an Atom? Neutrons and Protons!!

What subatomic particles are responsible for the bonding between atoms? Valence Electrons!!


IV. Common Biological Elements • You are responsible for knowing the following elements throughout the year: – Carbon *** – Hydrogen CHNOPS!!! – Nitrogen – Oxygen – Phosphorus – Sulfur * CHON = 96% of living matter

Think about it: What are the 6 most common biological elements?

What is the basic unit of all Matter? The Atom

V. Molecules • Molecule = the smallest unit any substance can be divided into (without losing its properties). – Example: 1 Water molecule! H 2 O 2 Hydrogen atoms + 1 Oxygen atom

VI. Compounds • Compounds = a combination of two or more different elements bonded together. – Example: Glucose! C 6 H 12 O 6 C= Black H = White O = Red

What’s the Difference? • A molecule is formed when two or more atoms join together chemically. • A compound is a molecule that contains at least two different elements. • All compounds are molecules but not all molecules are compounds.

Lets practice together!!

How many total atoms are found in one molecule of C 12 H 22 O 11? a. 3 b. 28 c. 35 d. 56
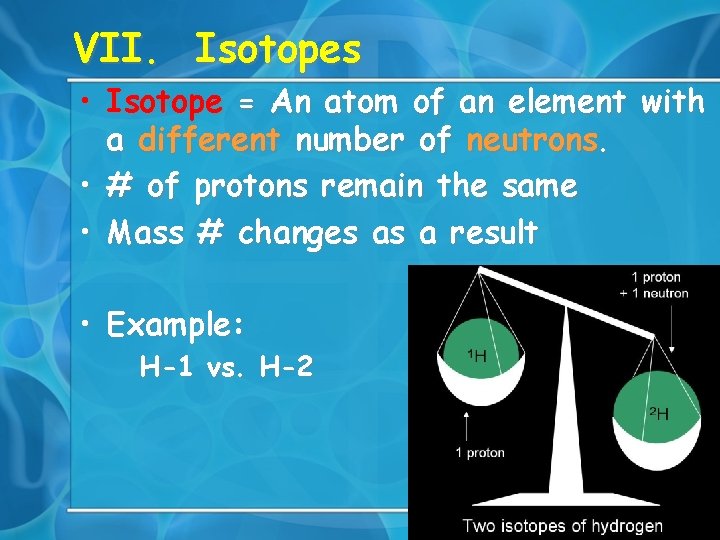
VII. Isotopes • Isotope = An atom of an element with a different number of neutrons. • # of protons remain the same • Mass # changes as a result • Example: H-1 vs. H-2

• Radioactive isotopes: – Occur when the nucleus of an atom begin to break down, releasing energy. – Used to find the age of extremely old organic matter. • Carbon 14 Dating

An atom of an element with a different number of neutrons is a ____? a. Element b. Carbon c. Compound d. Isotope

Which of the following is a radioactive isotope of carbon? a. C-12 b. C-13 c. C-14 d. C-15 e. C-16

Bonding and Chemical Reactions Notes

I. Bonds § Bonds= the force of attraction that holds atoms together. § Occurs between valence electrons that are oppositely charged (+ and -).
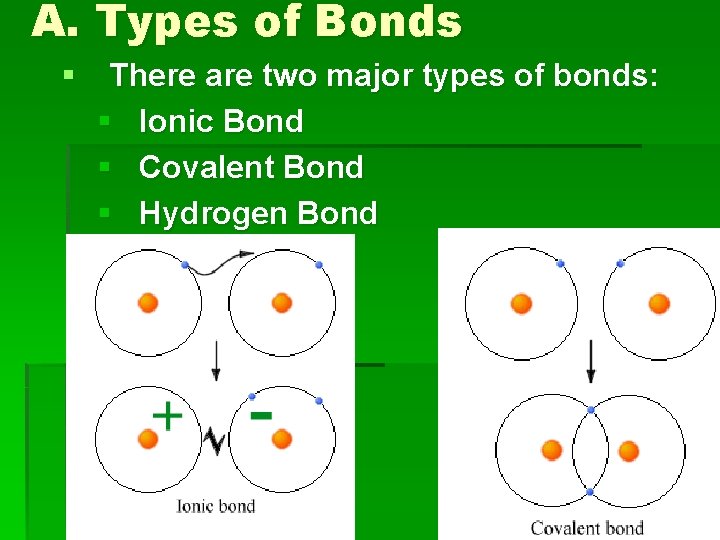
A. Types of Bonds § There are two major types of bonds: § Ionic Bond § Covalent Bond § Hydrogen Bond

1. Ionic Bonds § § § Ions: An atom or group of atoms that has an electrical charge (+ or -). Ion = an atom that has gained or lost an electron. Occur between a metal and a non-metal Positive Ions: § Lose an electron § More p+ than e. Negative Ions: § Gain an electron § Less p+ than e-
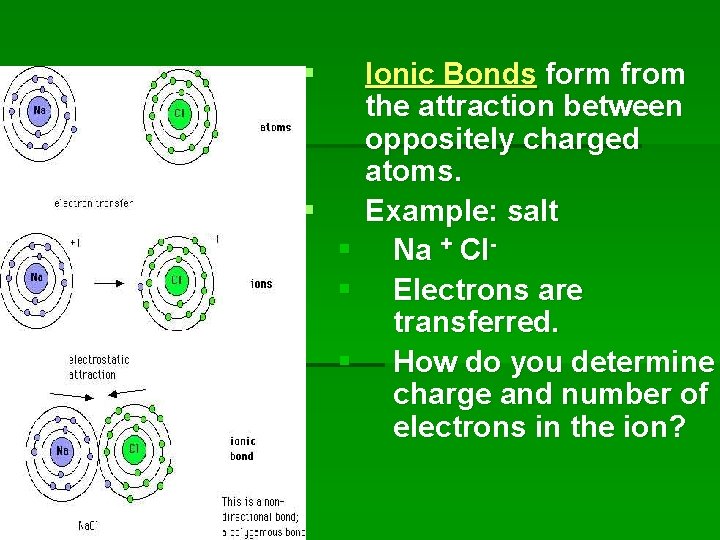
§ § § Ionic Bonds form from the attraction between oppositely charged atoms. Example: salt Na + Cl. Electrons are transferred. How do you determine charge and number of electrons in the ion?

2. Covalent Bonds § § Covalent Bonds form when two atoms SHARE electrons. Occurs between 2 non-metals The electrons shared are the outermost electrons—the VALENCE electrons. There are two types of covalent bonds: § Polar § Non-Polar

§ Polar Covalent Bonds = Covalent bonds where the electrons are SHARED unequally. § Example = H 20 § Non-Polar Covalent Bonds = Covalent bonds where the electrons are shared EQUALLY. § Example = CH 4 (Methane Gas!)

Which of the following bonds occurs when there is a transfer of electrons? a. polar covalent b. non-polar covalent c. ionic d. bipolar

Which of the following bonds occurs when there is an equal sharing of electrons? a. polar covalent c. ionic b. non-polar covalent d. bipolar

3. Hydrogen Bonds § Hydrogen Bonds= are bonds that form between Hydrogen atoms and Oxygen/ Nitrogen. § These bonds are weaker than covalent bonds.

Which of the following is bonded due to ionic bonds? a. H 2 O b. Na. Cl; metal and nonmetal c. CH 4; metal and metal d. all of them e. none of above b. Na. Cl; metal and nonmetal

II. Chemical Reactions § Definition: A chemical reaction occurs when substances undergo chemical changes to form NEW substances. § Break and form new bonds

A. Why Atoms Move § Every chemical reaction involves the rearrangement of elements as they react with one another.
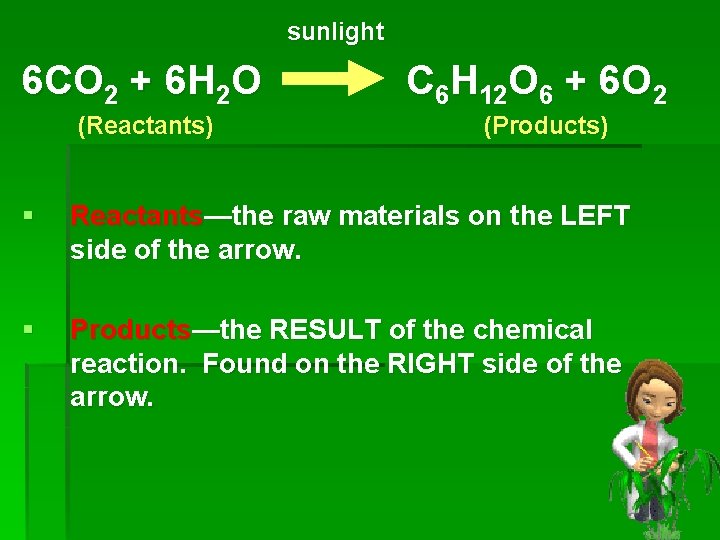
sunlight 6 CO 2 + 6 H 2 O (Reactants) C 6 H 12 O 6 + 6 O 2 (Products) § Reactants—the raw materials on the LEFT side of the arrow. § Products—the RESULT of the chemical reaction. Found on the RIGHT side of the arrow.
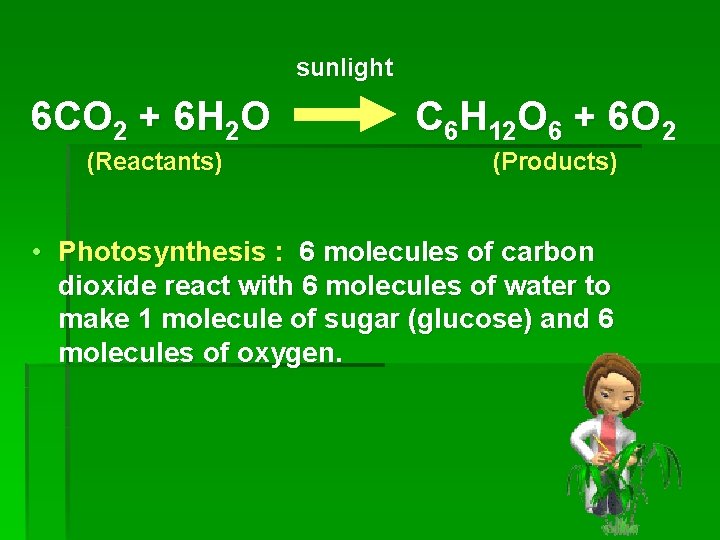
sunlight 6 CO 2 + 6 H 2 O (Reactants) C 6 H 12 O 6 + 6 O 2 (Products) • Photosynthesis : 6 molecules of carbon dioxide react with 6 molecules of water to make 1 molecule of sugar (glucose) and 6 molecules of oxygen.

B. Types of Energy (Why Bonds Form) § Catabolic Reaction= breaking bonds and energy is released. § Anabolic Reaction= when atoms join back together to form a new bonds and energy is stored in the bond.

1. Exothermic Reactions § § § exo = Out thermic = Heat Therefore, exothermic reactions release heat energy as they occur.

2. Endothermic Reactions § Endo = In § Therefore, endothermic reactions require the absorption of heat energy in order to occur.

If a reaction occurs and heat is released, is it an exothermic reaction or endothermic reaction?
Solution Chemistry

II. Solutions • Definition = the even distribution of substances dissolved in H 2 O. • Example: – H 2 O and Na. Cl

A. Solvents= the substance that breaks apart other substances. – H 2 O is the “universal” solvent. B. Solutes= the substance that is broken apart. - Kool Aid! C. Concentration = how much solute is in the solution.

What is the substance that breaks apart other substances? a. Solute b. Solvent c. H-Bond d. Suspension

III. Acids • Acids contain hydrogen ions: H+ • Acids donate hydrogen ions (H+) to water (H 2 O) to form H 3 O+ • H 3 O+ is known as a hydronium ion

A. Properties of Acids • Acids taste sour • Acids conduct electricity. • Acids turn litmus paper RED.
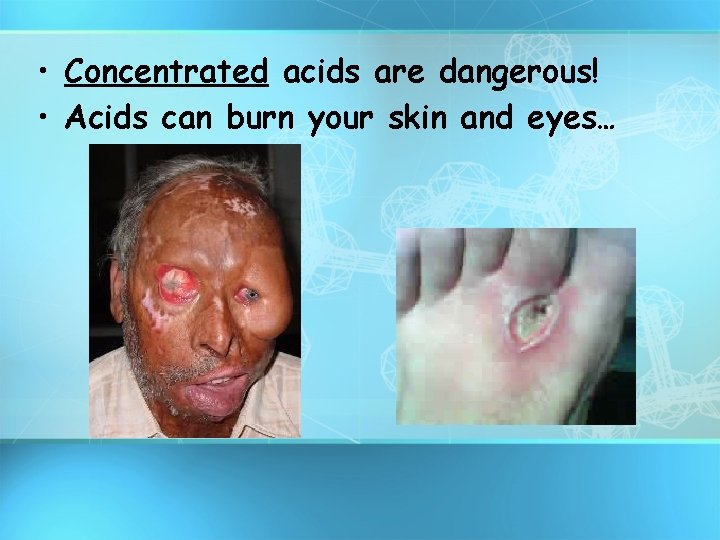
• Concentrated acids are dangerous! • Acids can burn your skin and eyes…

IV. Bases • Bases contain hydroxide ions: OH- A. Properties of Bases • Bases taste bitter. • Bases feel slippery • Bases turn litmus paper BLUE.
![V. p. H A. p. H is the measure of hydronium [H 3 O+] V. p. H A. p. H is the measure of hydronium [H 3 O+]](http://slidetodoc.com/presentation_image_h2/3ca529081698b9faddd352e468fded52/image-54.jpg)
V. p. H A. p. H is the measure of hydronium [H 3 O+] ions in a solution. B. The p. H scale is a much more accurate measure of how acidic or basic a solution is.

VI. p. H Scale • • • The p. H scale ranges from 0 -14 0 -6 is Acidic (more H+) 8 -14 is Basic (more OH-) 0 = VERY acidic [H 3 O+] 7 = neutral [H 2 O] (equal H+ and OH-) 14 = VERY basic [OH-] 0 7 14 Acidic Neutral Basic

• Low p. H is acid – Lots of H 3 O+ – Little OH • High p. H is base – Little H 3 O+ – Lots of OH-

Which of the following has the p. H that is most acidic? a. b. c. d. e. 3. 5 6. 3 2. 8 1. 5 11. 0

Which of the following has the p. H that is most basic? a. b. c. d. e. 7. 4 8. 3 13. 7 9. 4 12. 5

Why is pure water neutral? Please explain in words.
- Slides: 59