Chemistry Mrs Brittain Mrs Tew Ms Siddique Miss

Chemistry Mrs Brittain, Mrs Tew, Ms Siddique & Miss Hargreaves

Why choose to study …. ? • A Level Chemistry A is an excellent base for a university degree in healthcare such as medicine, pharmacy and dentistry as well as the biological sciences, physics, mathematics, pharmacology and analytical chemistry. • Chemistry is also taken by many law applicants as it shows you can cope with difficult concepts. • Chemistry can also complement a number of arts subjects. • A range of career opportunities including chemical, manufacturing and pharmaceutical industries and in areas such as forensics, environmental protection and healthcare. • The problem solving skills are useful for many other areas, too, such as general management and finance.

AS Level Chemistry OCR – H 032(AS) AS Level: Two 1 ½ hour examinations, modules 1 -4 Paper 1 - Breadth in chemistry (01)* 70 marks - 1 hour 30 minutes written paper (50%), multiple choice, short-medium length response answers. Tests recall and application. Paper 2 - Depth in chemistry (02)* 70 marks 1 hour 30 minutes written paper (50%), short-longer answer questions. Tests application, analysis & practical skill. *Both components include synoptic assessment

A Level Chemistry OCR – H 432 (A 2) A Level: Three examinations, 2 x 2 ¼ hour, 1 x 1 ½ hour Paper 1 – Modules 1, 2, 3 & 5 (01) 100 marks, 2 hour 15 minutes written paper (37%), multiple choice, short and longer response answers. Paper 2 – Modules 1, 2, 4 & 6 (02) 100 marks, 2 hour 15 minutes written paper (37%), multiple choice, short and longer response answers. Paper 3 - Synoptic paper (03) 70 marks, 1 hour 20 minutes (26%), short and longer response answers.

Starter: How many different ways can you represent a compound that contains 2 carbon atoms and 4 hydrogen atoms?
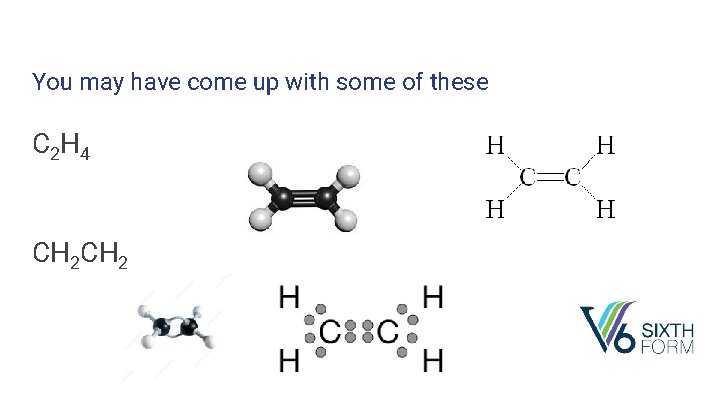
You may have come up with some of these C 2 H 4 CH 2

Representing & Making Organic Compounds Easier Represent organic substances in different ways Research information about Esters Find out the rules for naming organic compounds and apply them How do Chemists represent substances? Molecular formula, empirical formula, displayed formula, isomers, esters, alkenes Harder

Isomers A molecular formula shows how many atoms of each element are in a compound, e. g. C 5 H 10. However, there are nine different ways of putting together 5 carbon atoms and 10 hydrogen atoms. These are known as structural isomers. Some of them have one C=C bond and some are ring like structures. Can you find all of the isomers? Draw each one you find as a displayed formula e. g. C 2 H 4 Remember: Carbon makes 4 bonds and hydrogen only 1
Challenge! What is IUPAC? Look up the rules for naming organic compounds (particularly alkanes, cyclic alkanes and alkenes). Try to name the substances you drew on the previous slide.

Esters are a really interesting group of organic compounds. Find out the following about them: -What are they used for -Name some that naturally occur -Which one is responsible for the flavour of the sweets ‘Pear Drops’? What chemicals would you need to make it? - What are polyesters? How are they the same as/different to esters?

Pre-Course Reading and summer work See the reading list and separate booklet, ‘summer assignment’.
- Slides: 11