Chemistry Matter Anything that occupies space Solids matter




















































- Slides: 54

Chemistry

Matter Anything that occupies space

Solids matter with definite weight, volume and shape

Ice

liquids matter with definite weight and volume but no definite shape

Liquid Nitrogen

liquid

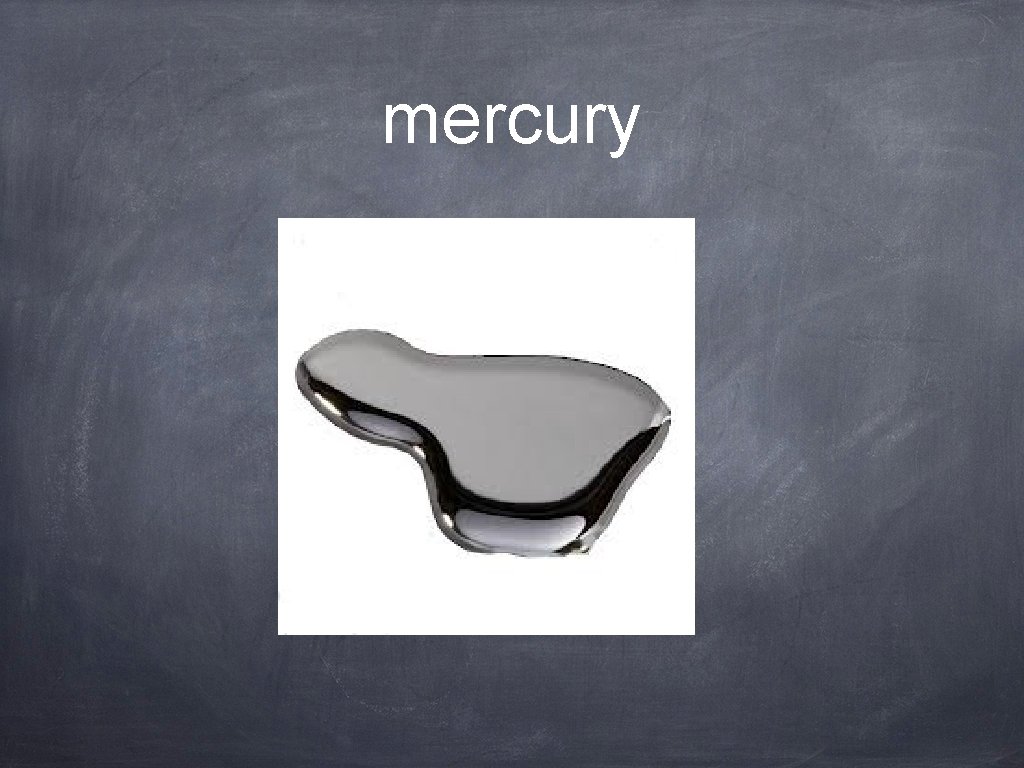
mercury

Gases Matter with definite weight but indefinite volume and shape

neon gas

Gases in many forms. . .

Physical change a change in the physical characteristics of a substance without creating a new substance

Chemical change in a substance that creates a new substance

The difference between the two http: //www. youtube. com/watch? v=g. Cbqjs-pq. Jo

Chemistry the scientific study of matter and the physical and chemical changes of matter

Organic Chemistry deals with all matter that is now living or was alive at one time, with carbon present such as plants and animals

inorganic chemistry studies all matter that is not alive, has never been alive and does not contain carbon, such as rocks, water and minerals

Elements basic substances that cannot be broken down int simpler substances

hydrogen gas atomic number 1 H

Helium atomic number 2 gas He

lithium atomic number 3 solid Li

Carbon Atomic number 6 C solid

Nitrogen Atomic number 7 gas N

Oxygen Atomic number 8 gas O

Aluminum Atomic number 13 solid Al

Sulfur Atomic number 16 solid S

Mercury Atomic number 80 liquid H

Atomic Number http: //www. youtube. com/watch? v=ufe. ZCeu_TS o

Protons Nuetrons Electrons http: //www. youtube. com/watch? v=Vi 91 qyjukn. M

Molecule two or more atoms joined by a chemical bond

Molecule http: //www. youtube. com/watch? v=qmg. E 0 w 6 E 6 ZI

Chemical Bonds http: //www. youtube. com/watch? v=_M 9 khs 87 x Q 8

Amino Acids compound consisting of carbon, oxygen hydrogen and nitrogen. 22 common aminos

Hair is a form of protein. . . Keratin 97% Keratin 3% trace minerals

Peptide Bonds http: //www. youtube. com/watch? v=va 0 DNJId_C M

Polypeptide Bonds Backbone of all protien molecules When two amino acids are positioned end to end, the acide endo of one amino accid attaches to the amino end of another amino acid

NO HAirdryer for Sodium hydroxide relaxer looses the characteristic of hair

Side bonds Hydrogen salt disulfide vander Waal’s forces

Hydrogen Bond http: //www. youtube. com/watch? v=tv 4 Jrc 06 y. LA

Salt Bond result in attraction of unlike charges

Disulfide Bond http: //www. youtube. com/watch? v=O 6 e. Ku. Sbn. O MA

van der Waal’s Forces role in bonding protien chains

experiment http: //www. youtube. com/watch? v=5 n 7 k. Bc. Xg o

Page 117 Review Hair’s chemical structure

Ph Potential Hydrogen

ph explained http: //www. youtube. com/watch? v=M 8 t. TELZD 5 Ek

The scale

Chemistry of Cosmetics solutions suspensions emulsions ointments soaps Powders

Scavenger Hunt Bring back examples from clinic floor

Water the universal solvent capable of dissolving more sublstanses than any other solvent

Shampoos
Go Go Go!! http: //www. youtube. com/watch? v=i-h. Xc. Rtbj 1 Y

Artist Access video

Albert E http: //www. youtube. com/watch? v=HZ_W 3 EAfp 6 I