CHEMISTRY Matter and Change Chapter 23 Substituted Hydrocarbons

CHEMISTRY Matter and Change Chapter 23: Substituted Hydrocarbons and Their Reactions

CHAPTER 23 Table Of Contents Section 23. 1 Alkyl Halides and Aryl Halides Section 23. 2 Alcohols, Ethers, and Amines Section 23. 3 Carbonyl Compounds Section 23. 4 Other Reactions of Organic Compounds Section 23. 5 Polymers Click a hyperlink to view the corresponding slides. Exit

2 3. 1 SECTION Alkyl Halides and Aryl Halides • Define functional group, and give examples. • Compare and contrast alkyl and aryl halide structures. • Evaluate the boiling points of organic halides. aliphatic compound: a nonaromatic hydrocarbon, such as an alkane, an alkene, or an alkyne

2 3. 1 SECTION Alkyl Halides and Aryl Halides functional group plastic halocarbon substitution reaction alkyl halide halogenation aryl halide A halogen atom can replace a hydrogen atom in some hydrocarbons.

2 3. 1 SECTION Alkyl Halides and Aryl Halides Functional Groups • In an organic molecule, a functional group is an atom or group of atoms that always reacts in a certain way. • Addition of a functional group to a hydrocarbon always produces a substance with different chemical and physical properties. • Double and triple bonds between carbon atoms are considered functional groups.
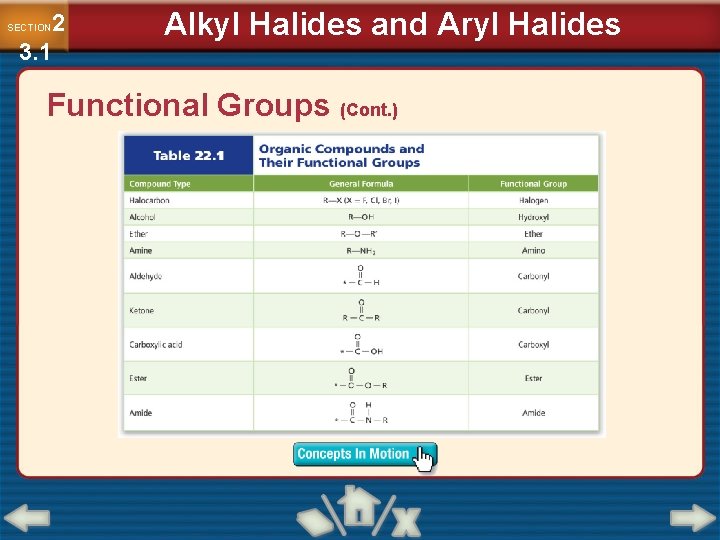
2 3. 1 SECTION Alkyl Halides and Aryl Halides Functional Groups (Cont. )

2 3. 1 SECTION Alkyl Halides and Aryl Halides Organic Compounds Containing Halogens • Any organic compound that contains a halogen substituent is called a halocarbon. • An alkyl halide is an organic compound containing a halogen atom covalently bonded to an aliphatic carbon atom. • An aryl halide is an organic compound containing a halogen bonded to an aromatic group.

Alkyl Halides and Aryl Halides 2 3. 1 SECTION Organic Compounds Containing Halogens (cont. ) • Organic molecules containing functional groups are given IUPAC names based on their main chain alkane structures. –For alkyl halides, a prefix indicating the halogen as follows: remove ine and add o. Ex. Fluoro, chloro, bromo, etc. –If there is more than a single halogen, they are listed alphabetically.

2 3. 1 SECTION Alkyl Halides and Aryl Halides Organic Compounds Containing Halogens (cont. ) • Halocarbons have higher boiling points because they have an increasing tendency to form temporary dipoles. • Dipoles attract each other, and require more energy to separate. • A plastic is a polymer that can be heated and molded while relatively soft.
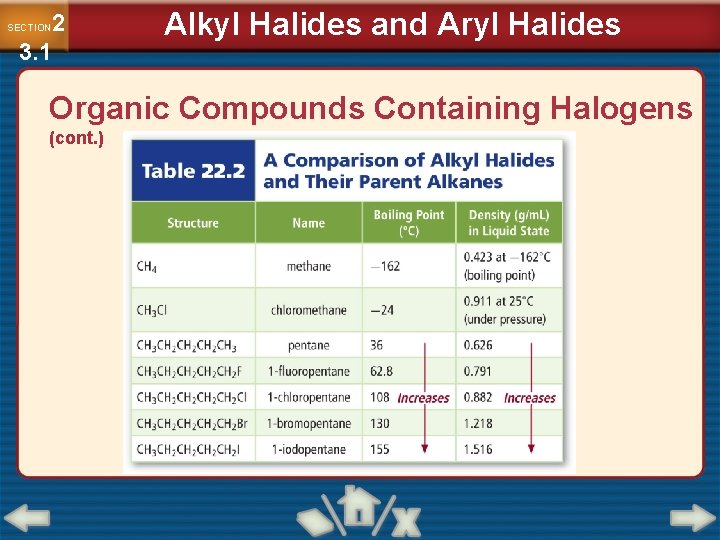
2 3. 1 SECTION Alkyl Halides and Aryl Halides Organic Compounds Containing Halogens (cont. )

2 3. 1 SECTION Alkyl Halides and Aryl Halides Substitution Reactions • Nearly all synthetic organic compounds begin with petroleum. • A substitution reaction is one in which one atom or a group of atoms in a molecule is replaced by another atom or group of atoms • Hydrogen atoms in an alkane can be replaced by atoms of halogens in a process called halogenation.

2 3. 1 SECTION Alkyl Halides and Aryl Halides Substitution Reactions (cont. )

2 3. 1 SECTION Alkyl Halides and Aryl Halides Substitution Reactions (cont. ) • Once halogenated, alkanes can undergo further reactions. • Reacting an alkyl halide with a basic solution results in replacement of the halogen atom with an –OH group to form an alcohol.

2 3. 1 SECTION Section Check Which of the following is NOT an alkyl halide? A. 2 -fluorobutane B. 1, 3 -dichlorobenzene C. sodium chloride D. bromoethane

2 3. 1 SECTION Section Check An alkane with one or more halogen substituents is called what? A. aryl halide B. metal halide C. chlorofluorocarbon D. alkyl halide


2 3. 2 SECTION Alcohols, Ethers, and Amines • Identify the functional groups that characterize alcohols, ethers, and amines. • Draw the structures of alcohols, ethers, and amines. • Discuss the properties and uses of alcohols, ethers, and amines. miscible: describes two liquids that are soluble in each other

2 3. 2 SECTION Alcohols, Ethers, and Amines hydroxyl group alcohol denatured alcohol ether amine Oxygen and nitrogen are two of the most-common atoms found in organic functional groups.

2 3. 2 SECTION Alcohols, Ethers, and Amines Alcohols • Oxygen commonly forms two covalent bonds to form a stable octet. • An oxygen-hydrogen group covalently bonded to a carbon atom is called a hydroxyl group. • An organic compound in which a hydroxyl group replaces a hydrogen is called an alcohol.
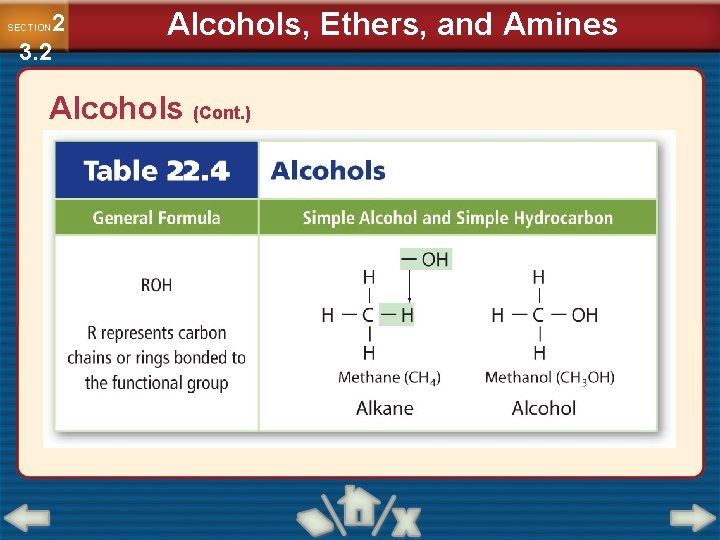
2 3. 2 SECTION Alcohols, Ethers, and Amines Alcohols (Cont. )

2 3. 2 SECTION Alcohols, Ethers, and Amines Alcohols (Cont. ) • Alcohol is polar and mixes completely with water. • Denatured alcohol is ethanol with small amounts of noxious materials added to it. • Alcohol names are based on the alkane names, with the ending –ol.
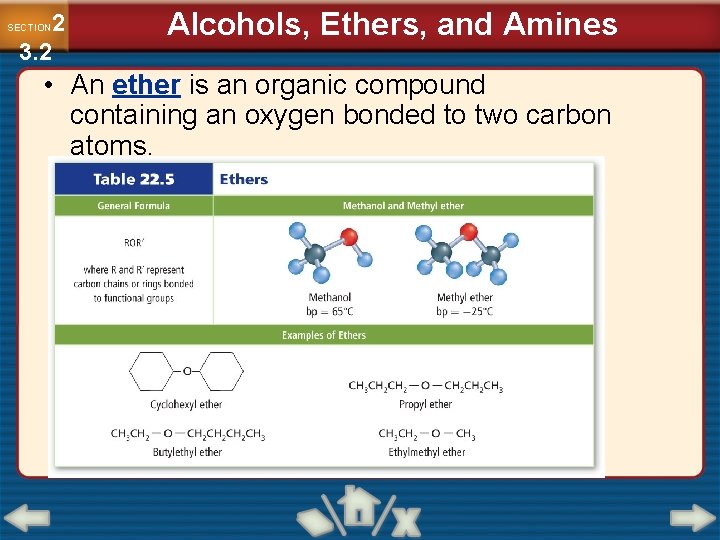
2 3. 2 SECTION Alcohols, Ethers, and Amines • An ether is an organic compound containing an oxygen bonded to two carbon atoms.

2 3. 2 SECTION Alcohols, Ethers, and Amines Ethers (Cont. ) • Ethers have no hydrogen atoms bonded to their oxygen atoms, so they cannot form hydrogen bonds with each other. • Ethers are volatile and have low boiling points. • Ethers with identical chains are named by the chain followed by the word ether. • Ethers with different chains are named in alphabetical order followed by ether.

2 3. 2 SECTION Alcohols, Ethers, and Amines • Amines contain nitrogen atoms bonded to carbon atoms in aliphatic chains or aromatic rings. • Amines are primary, secondary, or tertiary depending on if there are one, two, or three hydrogen atoms replaced by organic groups. • Amines have the suffix –amine. • If more than one amino group is present, the prefixes di, tri, tetra, etc. are used.

2 3. 2 SECTION Section Check Which of the following is an alcohol? A. cyclohexamine B. 1 -chlorobutanol C. bromobutene D. butylpropyl ether

2 3. 2 SECTION Section Check Which functional group is present in an alcohol? A. hydroxyl group B. halogen C. aromatic ring D. amino group


2 3. 3 SECTION Carbonyl Compounds • Identify the structures of carbonyl compounds, including aldehydes, ketones, carboxylic acids, esters, and amides. • Discuss the properties of compounds containing the carbonyl group. electronegative: indicates the relative ability of an element’s atoms to attract electrons in a chemical bond

Carbonyl Compounds 2 3. 3 SECTION carbonyl group carboxyl group aldehyde ester ketone amide carboxylic acid condensation reaction Carbonyl compounds contain a doublebonded oxygen in the functional group.

2 3. 3 SECTION Carbonyl Compounds Organic Compounds Containing the Carbonyl Group • An oxygen atom double-bonded to a carbon atom is a carbonyl group. • Aldehydes are organic compounds in which the carbonyl group is located at the end of the carbon chain and is bonded to a carbon atom on one side and a hydrogen on the other side. • Aldehydes are named with the suffix –al.
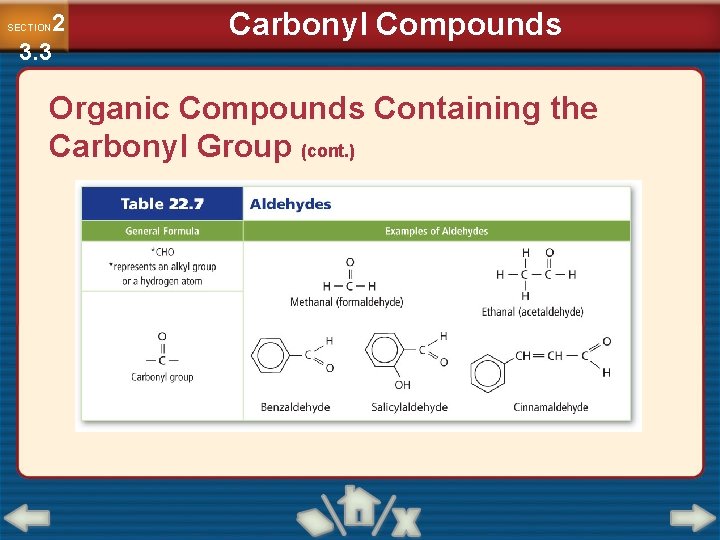
2 3. 3 SECTION Carbonyl Compounds Organic Compounds Containing the Carbonyl Group (cont. )

2 3. 3 SECTION Carbonyl Compounds Organic Compounds Containing the Carbonyl Group (cont. ) • A ketone is an organic compound in which the carbon of the carbonyl group is bonded to two other carbon atoms. • Ketones are named by changing the –e at the end of the alkane name to –one, and including a number before the name to indicate the position of the ketone group. • Ketones and aldehydes share many chemical and physical properties because their structures are similar.

2 3. 3 SECTION Carbonyl Compounds Organic Compounds Containing the Carbonyl Group (cont. ) • Ketones are popular solvents for moderately polar substances. • Ketones are somewhat soluble in water.

2 3. 3 SECTION Carbonyl Compounds Organic Compounds Containing the Carbonyl Group (cont. )

2 3. 3 SECTION Carbonyl Compounds Carboxylic Acids • Carboxylic acids are organic compounds that have a carboxyl group. • Carboxyl groups are carbonyls bonded to a hydroxyl group • Carboxylic acids are named by changing the –ane to –anoic acid.
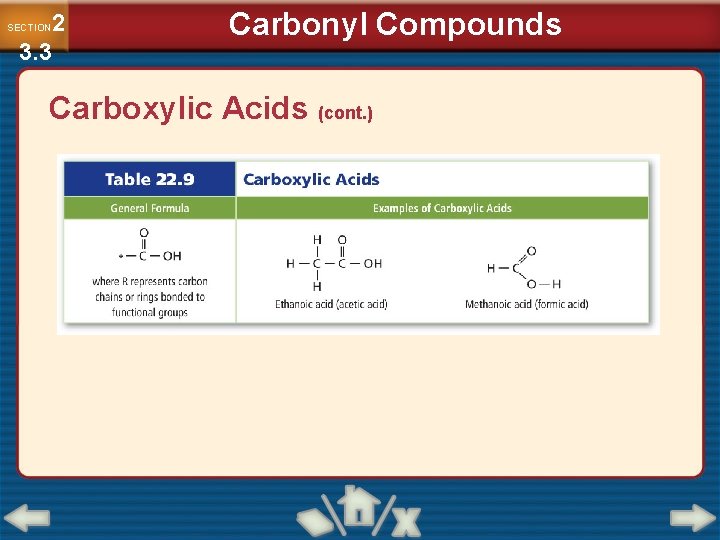
2 3. 3 SECTION Carbonyl Compounds Carboxylic Acids (cont. )

2 3. 3 SECTION Carbonyl Compounds Carboxylic Acids (cont. ) • Carboxylic groups are represented by –COOH. • Carboxylic acids are polar and reactive, and ionize in water to form acids.

2 3. 3 SECTION Carbonyl Compounds Organic Compounds Derived from Carboxylic Acids • An ester is any organic compound with a carboxyl group in which the hydrogen in the hydroxyl group is replaced by an alkyl chain.

2 3. 3 SECTION Carbonyl Compounds Organic Compounds Derived from Carboxylic Acids (Cont. ) • To name an ester, write the alkyl group followed by the name of the acid with the –oic acid ending replaced with –oate. • Esters are polar molecules and many are volatile and sweet smelling. • Many esters are found in fruits and flowers.

2 3. 3 SECTION Carbonyl Compounds Organic Compounds Derived from Carboxylic Acids (Cont. ) • An amide group is an organic compound in which the –OH group of a carboxylic acid is replaced by a nitrogen atom bonded to other atoms.

2 3. 3 SECTION Carbonyl Compounds Organic Compounds Derived from Carboxylic Acids (Cont. ) • The amide functional group is found repeated many times in natural proteins and some synthetic materials.
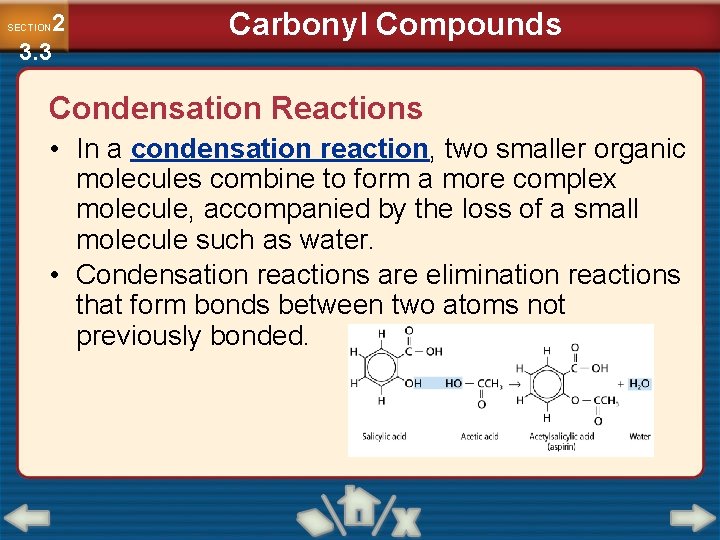
2 3. 3 SECTION Carbonyl Compounds Condensation Reactions • In a condensation reaction, two smaller organic molecules combine to form a more complex molecule, accompanied by the loss of a small molecule such as water. • Condensation reactions are elimination reactions that form bonds between two atoms not previously bonded.

2 3. 3 SECTION Section Check Which of the following is the carbonyl group? A. COOH B. C=O C. CONH 2 D. R-O-R′

2 3. 3 SECTION Section Check Which of the following does NOT contain a carbonyl group? A. ketones B. esters C. amines D. aldehydes


2 3. 4 SECTION Other Reactions of Organic Compounds • Classify an organic reaction into one of five categories: substitution, addition, elimination, oxidation -reduction, or condensation. • Use structural formulas to write equations for reactions of organic compounds. • Predict the products of common types of organic reactions. catalyst: a substance that increases the rate of a chemical reaction by lowering activation energies but is not consumed in the reaction

2 3. 4 SECTION Other Reactions of Organic Compounds elimination reaction addition reaction dehydrogenation reaction hydration reaction dehydration reaction hydrogenation reaction Classifying the chemical reactions of organic compounds makes predicting products of reactions much easier.

2 3. 4 SECTION Other Reactions of Organic Compounds Classifying Reactions of Organic Substances • Using combinations of reactions allows chemical industries to convert simple molecules from petroleum and natural gas into large, complex organic molecules.

2 3. 4 SECTION Other Reactions of Organic Compounds • The formation of alkenes from alkanes is an elimination reaction, where a combination of atoms is removed from two adjacent carbon atoms forming an additional bond between the two carbon atoms.

2 3. 4 SECTION Other Reactions of Organic Compounds • A reaction that eliminates two hydrogen atoms is called a dehydrogenation reaction.
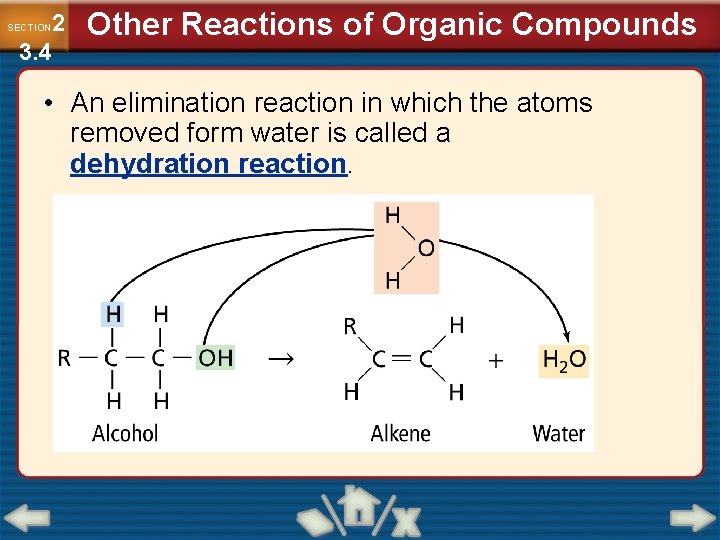
2 3. 4 SECTION Other Reactions of Organic Compounds • An elimination reaction in which the atoms removed form water is called a dehydration reaction.

2 3. 4 SECTION Other Reactions of Organic Compounds • Addition reactions result when other atoms bond to two atoms bonded by double or triple covalent bonds. • A hydration reaction is an addition reaction in which a hydrogen atom and hydroxyl group from a water molecule add to a double or triple bond.

2 3. 4 SECTION Other Reactions of Organic Compounds • A hydrogenation reaction involves the addition of hydrogen to atoms in a double or triple bond. • Catalysts are usually needed in hydrogenation reactions. • Hydrogenation reactions are commonly used to convert liquid unsaturated fats into saturated fats that are solid at room temperature.

2 3. 4 SECTION Other Reactions of Organic Compounds

2 3. 4 SECTION Other Reactions of Organic Compounds Classifying Reactions of Organic Substances (Cont. ) • Many organic compounds can be converted to other compounds by oxidation-reduction reactions.
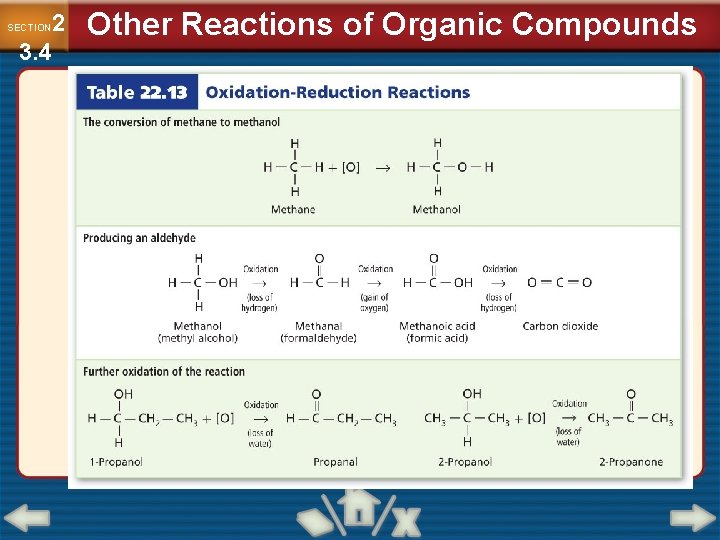
2 3. 4 SECTION Other Reactions of Organic Compounds

2 3. 4 SECTION Other Reactions of Organic Compounds Predicting Products of Organic Reactions • Each type of reaction—substitution, addition, elimination, oxidation-reduction, and condensation—can be used to predict the products of other organic reactions of the same types.

2 3. 4 SECTION Section Check Which type of reaction normally produces an alcohol from an alkene? A. substitution B. elimination C. hydration D. addition

2 3. 4 SECTION Section Check What type of reaction is the following? alkyl halide → alkene A. halogenation B. elimination C. addition D. substitution


2 3. 5 SECTION Polymers • Diagram the relationship between a polymer and the monomers from which it forms. • Classify polymerization reactions as addition or condensation. • Predict polymer properties based on their molecular structures and the presence of functional groups. molecular mass: the mass of one molecule of a substance

Polymers 2 3. 5 SECTION polymer condensation polymerization monomer polymerization reaction addition polymerization thermoplastic thermosetting Synthetic polymers are large organic molecules made up of repeating units that are linked together by addition or condensation reactions.

2 3. 5 SECTION Polymers The Age of Polymers • Polymers are large molecules consisting of many repeating structural units. • The turn of the 20 th century saw the advent of such polymers as rubber and celluloid.

2 3. 5 SECTION Polymers Reactions Used to Make Polymers • A monomer is a molecule from which a polymer is made. • A reaction in which monomer units are bonded together to form a polymer are called polymerization reactions. • In addition polymerization all of the atoms present in the monomers are retained in the polymer product.

2 3. 5 SECTION Polymers Reactions Used to Make Polymers (cont. )

2 3. 5 SECTION Polymers Reactions Used to Make Polymers (cont. )

2 3. 5 SECTION Polymers Reactions Used to Make Polymers (cont. )

2 3. 5 SECTION Polymers Reactions Used to Make Polymers (cont. ) • Condensation polymerization takes place when monomers containing at least two functional groups combine with the loss of a small by-product, usually water.

2 3. 5 SECTION Polymers Properties and Recycling of Polymers • Polymers are inexpensive and easy to make. • Polymers have a wide range of properties. • They are easy to mold into different shapes. • Thermoplastic polymers can be melted and molded repeatedly into shapes that are retained when cooled. • Thermosetting polymers can be molded when first prepared, but cannot be remelted.

2 3. 5 SECTION Polymers Properties and Recycling of Polymers (Cont. ) • As fossil fuel supplies are depleted, recycling polymers becomes more important.

2 3. 5 SECTION Section Check The smaller, repeating units that make up a polymer are called ____. A. structural units B. polyunits C. monomers D. building blocks

2 3. 5 SECTION Section Check One way polymers are synthesized is through ____ reactions. A. condensation B. elimination C. substitution D. dehydration


CHAPTER 23 Substituted Hydrocarbons and Their Reactions Resources Chemistry Online Study Guide Chapter Assessment Standardized Test Practice

2 3. 1 Alkyl Halides and Aryl Halides SECTION Study Guide Key Concepts • The substitution of functional groups for hydrogen in hydrocarbons creates a wide variety of organic compounds. • An alkyl halide is an organic compound that has one or more halogen atoms bonded to a carbon atom in an aliphatic compound.

2 3. 2 Alcohols, Ethers, and Amines SECTION Study Guide Key Concepts • Alcohols, ethers, and amines are formed when specific functional groups substitute for hydrogen in hydrocarbons. • Because they readily form hydrogen bonds, alcohols have higher boiling points and higher water solubilities than other organic compounds.

2 3. 3 Carbonyl Compounds SECTION Study Guide Key Concepts • Carbonyl compounds are organic compounds that contain the C=O group. • Five important classes of organic compounds containing carbonyl compounds are aldehydes, ketones, carboxylic acids, esters, and amides.

2 3. 4 SECTION Other Reactions of Organic Compounds Study Guide Key Concepts • Most reactions of organic compounds can be classified into one of five categories: substitution, elimination, addition, oxidation-reduction, and condensation. • Knowing the types of organic compounds reacting can enable you to predict the reaction products.

2 3. 5 Polymers SECTION Study Guide Key Concepts • Polymers are large molecules formed by combining smaller molecules called monomers. • Polymers are synthesized through addition or condensation reactions. • The functional groups present in polymers can be used to predict polymer properties.

CHAPTER 23 Substituted Hydrocarbons and Their Reactions Chapter Assessment Aryl halides contain what two groups? A. nitrogen and aliphatic hydrocarbon chain B. nitrogen and aromatic hydrocarbon ring C. halogen and aromatic hydrocarbon ring D. halogen and aliphatic hydrocarbon chain

CHAPTER 23 Substituted Hydrocarbons and Their Reactions Chapter Assessment Amines contain which functional group? A. –OH B. –Br C. R-O-R′ D. –NH 2

CHAPTER 23 Substituted Hydrocarbons and Their Reactions Chapter Assessment Which of the following does NOT contain the carbonyl group? A. ketones B. ethers C. aldehydes D. amides

CHAPTER 23 Substituted Hydrocarbons and Their Reactions Chapter Assessment What is an addition reaction in which a hydrogen atom and hydroxyl group from a water molecule add to a double or triple bond called? A. elimination reaction B. hydration reaction C. dehydration reaction D. hydrogenation reaction

CHAPTER 23 Substituted Hydrocarbons and Their Reactions Chapter Assessment A type of plastic that can be molded only when first produced is called a ____. A. thermoplastic B. polythermal C. setting plastic D. thermosetting

CHAPTER 23 Substituted Hydrocarbons and Their Reactions Standardized Test Practice What are the products of this reaction? CH 3 CH 2 Br + Na. OH → ____ A. CH 3 CH 2 Na + Br. OH B. CH 3 CH 2 OH + Na. Br C. CH 3 CH 2 OHNa. Br D. CH 3 + Na 3 Br. O

CHAPTER 23 Substituted Hydrocarbons and Their Reactions Standardized Test Practice What type of compound does this formula represent? CH 3 CH 2 COOH A. ester B. ether C. carboxylic acid D. ketone

CHAPTER 23 Substituted Hydrocarbons and Their Reactions Standardized Test Practice How many carbon atoms are present in 2 -ethyl pentane? A. 2 B. 5 C. 7 D. 8

CHAPTER 23 Substituted Hydrocarbons and Their Reactions Standardized Test Practice What are the oxidation numbers of the elements in H 2 SO 4? A. H = +1, S = +6, O = – 2 B. H = +2, S = +4, O = – 4 C. H = +1, S = +4, O = – 1 D. H = +1, S = – 10, O = +2

CHAPTER 23 Substituted Hydrocarbons and Their Reactions Standardized Test Practice Which type of compound is formed when a base accepts H+ ions? A. Brønsted-Lowery base B. Brønsted-Lowery acid C. conjugate base D. conjugate acid

This slide is intentionally blank.
- Slides: 90