CHEMISTRY Last day before the SOLs cram day

CHEMISTRY Last day before the SOL’s cram day

Most important topics on the SOL ¢ ¢ ¢ Naming (ionic, covalent, transition metal) Bonding (ionic, covalent) Measurement (estimate last digit) Significant figures (+ and -, x and ÷) Subatomic particles (p+, n 0, e-, mass, atomic #) Chemical and physical properties and changes MOLES Reactions and Equations Stoichiometry Molarity and dilutions Gas Laws Thermochemistry
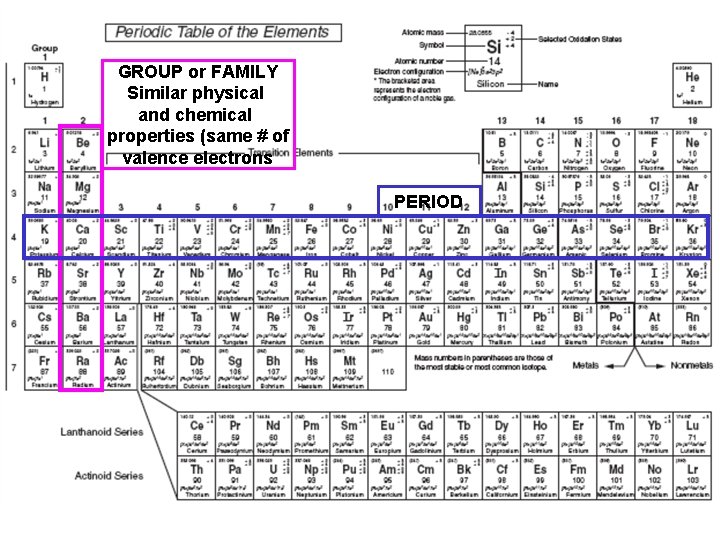
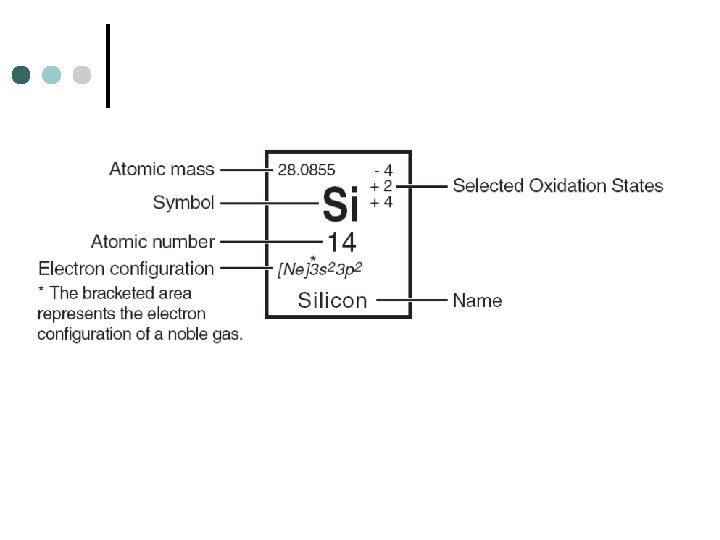
GROUP or FAMILY Similar physical and chemical properties (same # of valence electrons PERIOD
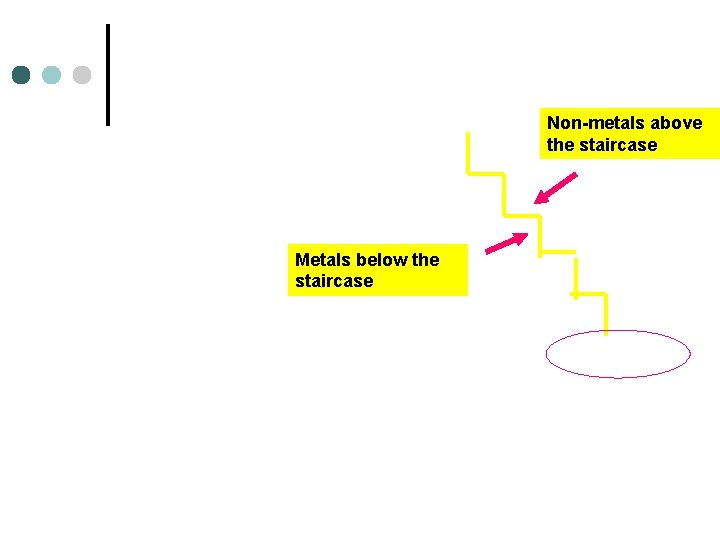
Non-metals above the staircase Metals below the staircase

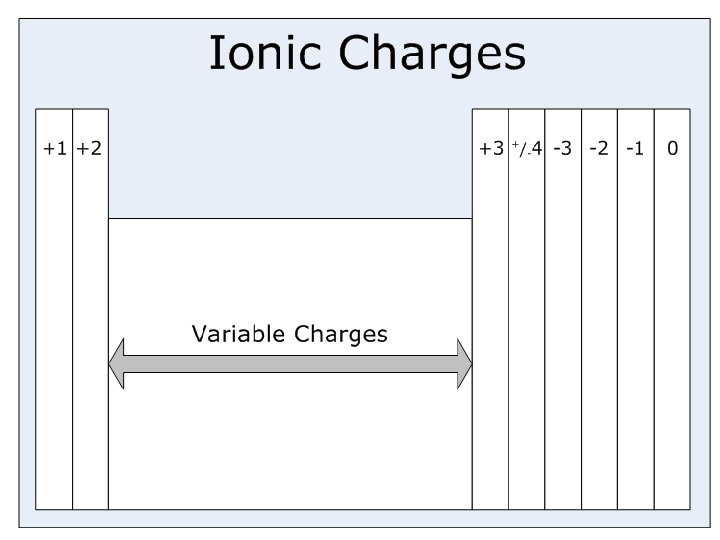
Naming (ionic, covalent, transition metal) ¢ Oxidation number: the charge of a monatomic ion l Indicates the number of electrons an atom has lost(+) or gained(-) l Metals tend to lose electrons (+ oxidation numbers) ¢ Cations with more than one oxidation number: l l ¢ ¢ Indicate the oxidation number with a Roman numeral Example: Fe 2+ = iron(II) ion Fe 3+ = iron(III) ion Ionic: Metal + nonmetal ex: Na. Cl (sodium chloride) Covalent: CO 2 (carbon dioxide) use prefixes
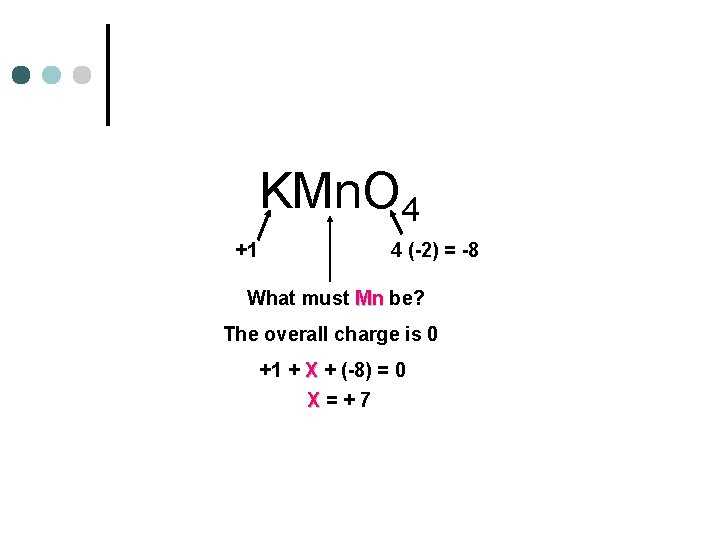
KMn. O 4 +1 4 (-2) = -8 What must Mn be? The overall charge is 0 +1 + X + (-8) = 0 X=+7

¢ Molecular compounds are composed of two non-metals (above the staircase) ¢ Indicate # of each atom using prefixes (mono, di, tri, tetra, penta, hexa, hepta, octa, nona, deca) ¢ The first element does not use mono if there’s only one. ¢ Examples: l OF 2 is named oxygen diflouride l N 2 O is named dinitrogen monoxide
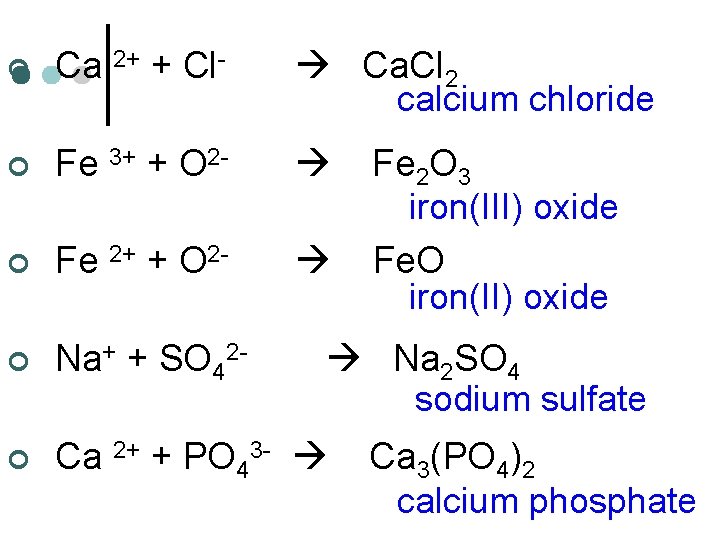
¢ Ca 2+ + Cl- Ca. Cl 2 calcium chloride ¢ Fe 3+ + O 2 - ¢ Fe 2+ + O 2 - ¢ Na+ + SO 42 - ¢ Ca 2+ + PO 43 - Fe 2 O 3 iron(III) oxide Fe. O iron(II) oxide Na 2 SO 4 sodium sulfate Ca 3(PO 4)2 calcium phosphate
Bonding (ionic, covalent) I C
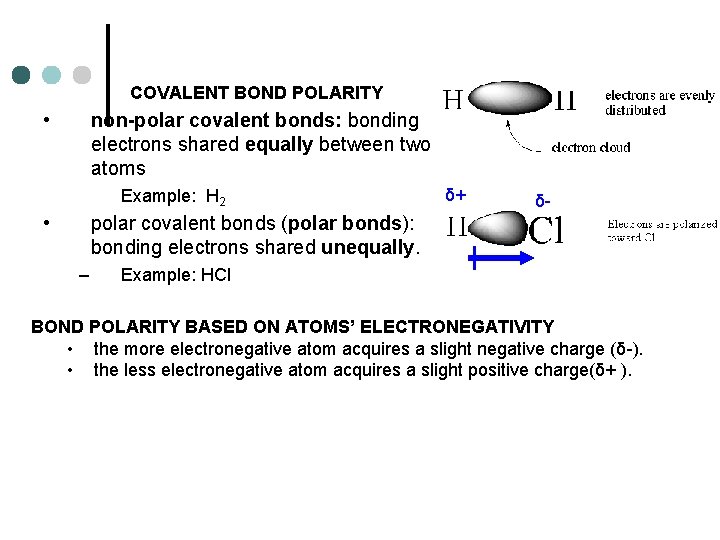
COVALENT BOND POLARITY • non-polar covalent bonds: bonding electrons shared equally between two atoms Example: H 2 • δ+ δ- polar covalent bonds (polar bonds): bonding electrons shared unequally. – Example: HCl BOND POLARITY BASED ON ATOMS’ ELECTRONEGATIVITY • the more electronegative atom acquires a slight negative charge (δ-). • the less electronegative atom acquires a slight positive charge(δ+ ).
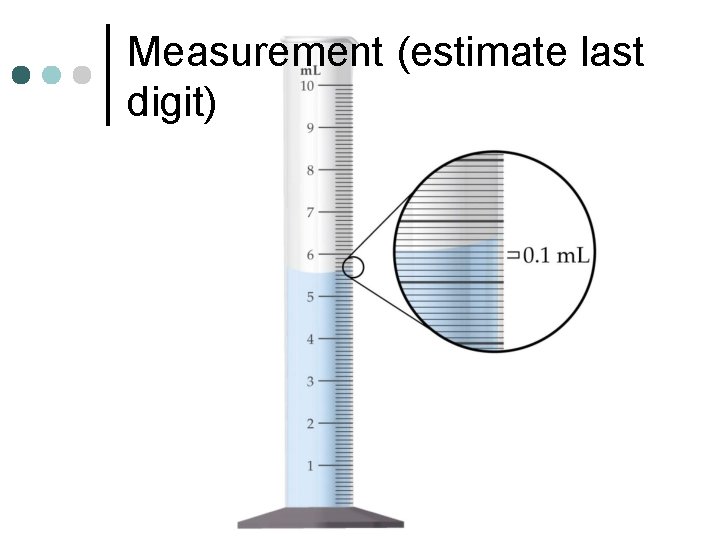
Measurement (estimate last digit)

Significant figures (+ and -, x and ÷) • Adding/subtracting lowest number of decimal places • Multiplying/dividing lowest number of significant figures

Subatomic particles (p+, n 0, e -, mass, atomic #) ¢ ¢ Isotopes – atoms with the same number of protons but different numbers of neutrons Ions- atoms that have lost or gained electrons making them have a positive or negative charge Atomic number (# of protons) l ANION: negative ion (gained an e-) for an atom never changes… l CATION: positive ion (lost an e-) that’s how we identify an atom

Chemical and physical properties ¢ ¢ Physical Properties – descriptions which do not involve a change in composition (color, density, texture, etc. ) Chemical Properties – Descriptions of a change in composition of a substance (reactivity, combustibility, etc. )

Chemical and physical change ¢ ¢ Physical change l does not alter the composition of the substance l Includes changes in state of matter, or shape Chemical Change l Alters the chemical structure of the substance l Cannot get original substance back through physical means • Evolution of a gas • Production of heat / light • Formation of a precipitate • Unexpected color change

MOLES Know Your Triangles! ¢ 1 mole= 6. 02 x 1023 particles ¢ 1 mole = molar mass in grams ¢ 1 mole = 22. 4 L of gas at STP ¢ When in doubt about what to do convert what you are given into moles and see if you can convert moles into what you need. ¢

Reactions Synthesis A + B AB ¢ Decomposition AB A+B ¢ Single Replacement A + BX B + AX ¢ Combustion Cx. Hy + O 2 CO 2+H 2 O ¢ Double Replacement ¢ • AB + XY AY + BX HX + MOH MX + H 2 O (neutralization)

Equations Balance in order to obey Law of Conservation of Mass ¢ Same number of atoms of each type on both sides ¢ Reactants Products ¢

Stoichiometry Change given quantity to moles ¢ Use mole ratio from Equation ¢ Change moles to answer quantity OR ¢ Change the coefficients (if needed) by multiplying by molar mass, molar volume, or avogadro’s number ¢ Set up a proportion and solve. ¢

Stoichiometry Cont. ¢ Limiting Reactant- The one that runs out first l ¢ Calculate twice. The Limiting Reactant gives you the least amount of product Percent Yield: [(Actual Yield) /( Theoretical Yield)] x 100%

Molarity and dilutions • Molarity = moles Liters Change masses to moles and m. L to L • Dilutions: M 1 V 1 = M 2 V 2 • M = Molarity • 1 = concentrated solution V = Volume 2 = diluted solution

Gas Laws 1. General Properties of Gases There is a lot of “free” space in a gas. Gases can be expanded infinitely. Gases fill containers uniformly and completely. Gases diffuse and mix rapidly. The Combined Gas Law P 1 V 1 = P 2 V 2 T 1 Boyle’s Law Inverse relationship Charles Law T 2 P 1 V 1 = P 2 V 2 V 1 = T 1 Lussac’s Law P 1 T 1 V 2 T 2 = P 2 T 2 Always use degrees Kelvin °C + 273 = K

The Ideal Gas Law Memorize: PV = n. RT • P= pressure in k. Pa • V= liters • n= moles • T= temperature in Kelvin • R = universal gas law constant = 8. 314 k. Pa x L Moles x K • The SOL test uses Dalton’s Law of Partial Pressures Memorize: Ptotal = P 1 + P 2 + P 3 + …

Gas pressure is measured in atmospheres, kilopascals (k. Pa), or mm Hg One atmosphere = 101. 3 k. Pa = 760 mm Hg Assumptions relating to gases: 1. Gas particles have negligible volume compared to container size 2. Gas particles do not attract or repel each other* 3. Gas particle move constantly, rapidly and randomly 4. All collisions are perfectly elastic (particles collide like billiard balls, not marshmallows) However, gas particles really do attract each other due to intermolecular forces
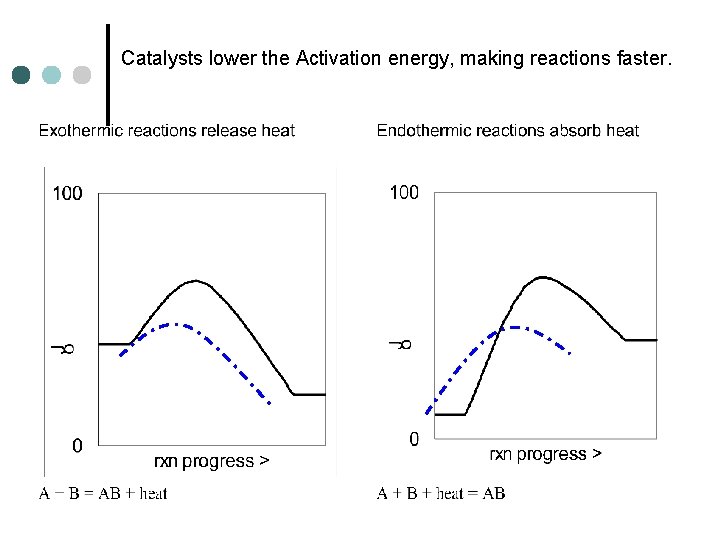
Catalysts lower the Activation energy, making reactions faster.

Reversible Reactions N 2(g) + 3 H 2(g) 2 NH 3(g) + heat The forward reaction takes place at the same rate as the reverse reaction.
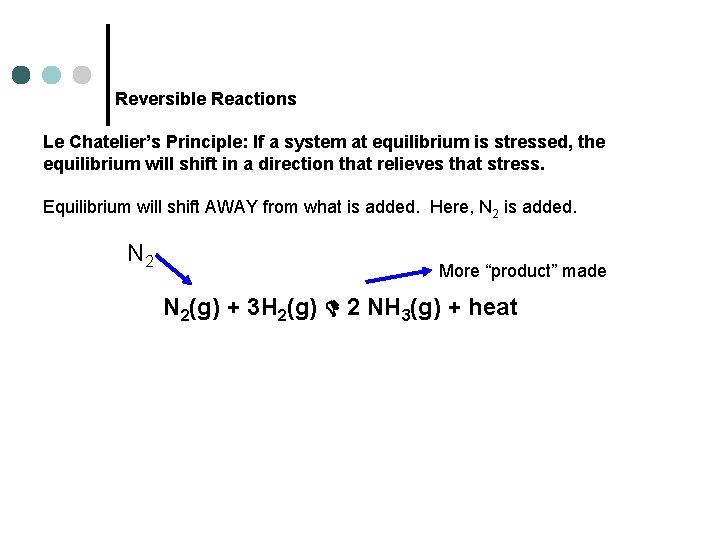
Reversible Reactions Le Chatelier’s Principle: If a system at equilibrium is stressed, the equilibrium will shift in a direction that relieves that stress. Equilibrium will shift AWAY from what is added. Here, N 2 is added. N 2 More “product” made N 2(g) + 3 H 2(g) 2 NH 3(g) + heat

Reversible Reactions Le Chatelier’s Principle: If a system at equilibrium is stressed, the equilibrium will shift in a direction that relieves that stress. Equilibrium will shift AWAY from what is added. Here, NH 3 is added. More “reactants” made NH 3 N 2(g) + 3 H 2(g) 2 NH 3(g) + heat

Reversible Reactions Le Chatelier’s Principle: If a system at equilibrium is stressed, the equilibrium will shift in a direction that relieves that stress. Equilibrium will shift TOWARDS what is removed. Here H 2 is removed. H 2 N 2(g) + 3 H 2(g) 2 NH 3(g) + heat More “reactants” made

Reversible Reactions Le Chatelier’s Principle: If a system at equilibrium is stressed, the equilibrium will shift in a direction that relieves that stress. Equilibrium will shift TOWARDS what is removed. Here heat is removed. heat N 2(g) + 3 H 2(g) 2 NH 3(g) + heat More “product” made
![What is p. H? p. H indicates the hydrogen ion molarity [H+] in a What is p. H? p. H indicates the hydrogen ion molarity [H+] in a](http://slidetodoc.com/presentation_image_h2/bb0fdc885be614d0681bcc055b7bbd70/image-35.jpg)
What is p. H? p. H indicates the hydrogen ion molarity [H+] in a solution p. H = -log[H+] p. OH indicates the hydroxide ion molarity [OH-] in a solution. p. OH = -log[OH-] 3 Example: A 1. 0 x 10 -3 molar solution of HCl would have a p. H of ___ 4 Example: A 1. 0 x 10 -4 molar solution of KOH would have a p. OH of ___ Memorize: p. H + p. OH = 14. 6 Example: A solution with a p. H of 8 will have a p. OH of: ____.
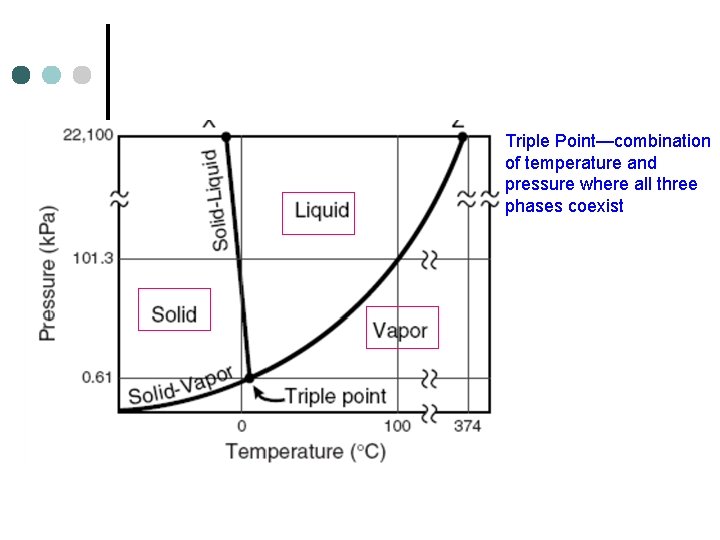
Triple Point—combination of temperature and pressure where all three phases coexist
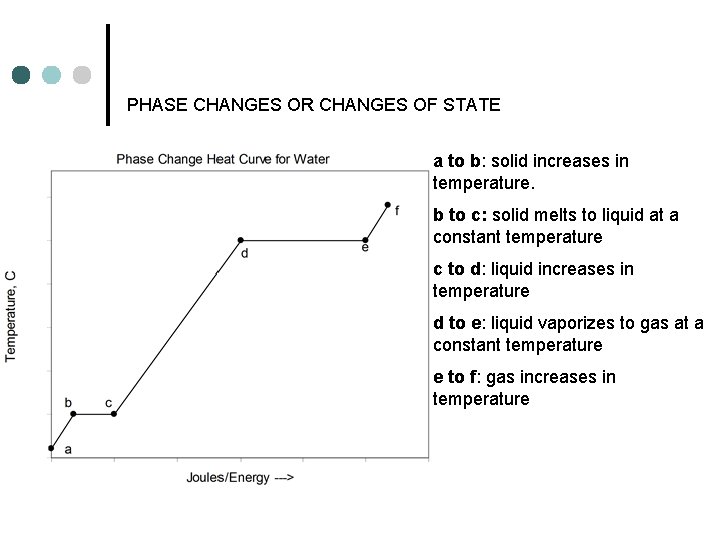
PHASE CHANGES OR CHANGES OF STATE a to b: solid increases in temperature. b to c: solid melts to liquid at a constant temperature c to d: liquid increases in temperature d to e: liquid vaporizes to gas at a constant temperature e to f: gas increases in temperature

GOOD LUCK ON THE SOL
- Slides: 38