Chemistry Introduction to Matter 1 1 Describing Matter

Chemistry : Introduction to Matter 1. 1 Describing Matter 10/26/2021 Chem-100 1

matter n Anything that has mass and takes up space 10/26/2021 Chem-100 n Hold up something that is an example of matter. 2

Properties of Matter Chemistry - the study of the properties of matter & how matter changes Substance n a single kind of matter that is pure n Always has a specific make-up & specific set of properties. Write 3 examples of substances. 10/26/2021 Chem-100 3

What are the 2 kinds of properties every form of matter has? Physical Properties & Chemical Properties 10/26/2021 Chem-100 4

Physical Properties n A characteristic of a pure substance that can be observed w/out changing it into another substance Ex: water freezing (solid), water (liquid), water vapor (gas) n Hardness, texture, color, ability to dissolve, luster, conductivity, magnetism, flexibility n Physical properties can be used to classify matter. n 10/26/2021 Chem-100 5

Chemical Properties n A characteristic of a pure substance that describes its ability to change into different substances To do this you must try to change it into another substance. n Flammability, oxidation, tarnishing n Chemical properties be used to classify matter. n 10/26/2021 Chem-100 6

Chemical Properties Flammability When wood burns, what element does it combine with? is a chemical property of Oxygen! wood. 10/26/2021 Chem-100 7

Chemical Properties Flammability What new substances are produced? is a Carbon chemical property of dioxide wood. & ash 10/26/2021 Chem-100 8

Chemical Properties Oxidation is a chemical property of iron. 10/26/2021 When iron rusts, what element does it combine with? Oxygen! Chem-100 9

Chemical Properties Oxidation is a chemical property of iron. 10/26/2021 What new substance is produced? Rust Iron oxide! Chem-100 10
Element – pure substance that cannot be broken down into any other substance 10/26/2021 Chem-100 11
How can you tell one element from another? 10/26/2021 Chem-100 12

How can you tell one element from another? By their chemical & physical properties! 10/26/2021 Chem-100 13

Atoms The basic particle from which all elements are made. n building blocks of matter n Can combine w/ other atoms through chemical bonds Molecules – groups of 2 or more atoms held together by chemical bonds 10/26/2021 Chem-100 14
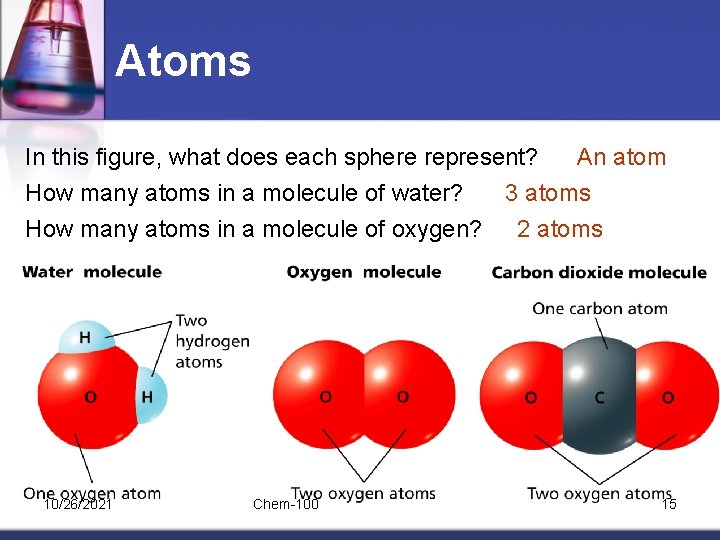
Atoms In this figure, what does each sphere represent? An atom How many atoms in a molecule of water? 3 atoms How many atoms in a molecule of oxygen? 2 atoms 10/26/2021 Chem-100 15
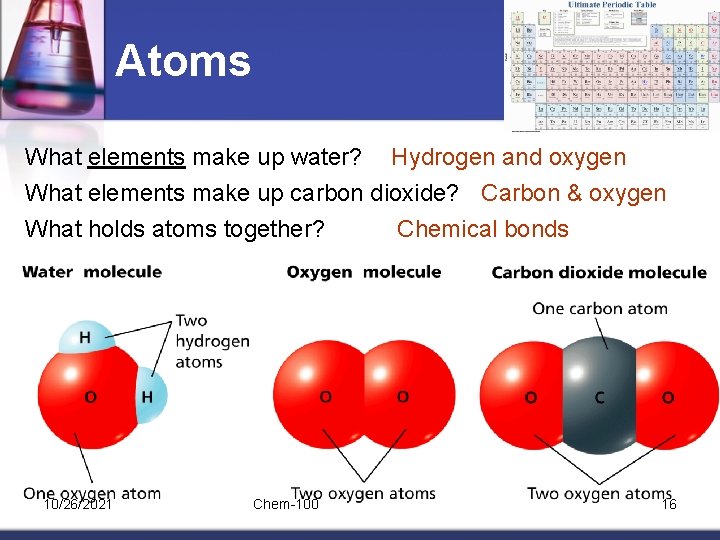
Atoms What elements make up water? Hydrogen and oxygen What elements make up carbon dioxide? Carbon & oxygen What holds atoms together? Chemical bonds 10/26/2021 Chem-100 16

Compounds A pure substance made of 2 or more elements chemically bonded together n When elements combine, they form compounds w/ different properties than their original properties. What is the ratio of atoms in these compounds? CO 2 1 to 2 10/26/2021 CO 1 to 1 Chem-100 H 2 O 2 to 1 17
100 Greatest Discoveries Let’s review oxygen, atoms & molecules Click here for this 8 minute video. 10/26/2021 Chem-100 18

Mixtures 2 or more substances together in the same place but not chemically combined n Each substance keeps its original properties 10/26/2021 Chem-100 19

Heterogeneous mixture Easily separated n You can see all the different parts n n Which of these pictures shows a heterogeneous mixture? 10/26/2021 Chem-100 20

Homogeneous mixture n Mixed so evenly you cannot see all the different parts n Which of these pictures shows a homogeneous mixture? 10/26/2021 Chem-100 21

10/26/2021 Chem-100 brass heterogeneous homogeneous air salad homogeneous Sugar water homogeneous Heterogeneous OR Homogeneous? 22

Separating Mixtures Easy to do because each component keeps its own properties n Separate through magnetic attraction, filtration, distillation or evaporation (see fig. 10) 10/26/2021 Chem-100 23

And now back to Ms. Frizzle’s class… 10/26/2021 Chem-100 Click picture for the 5 minute continuation of the Magic School Bus video. 24
- Slides: 24