Chemistry in cosmetics Cosmetics In Greek kosmetik tekhn












- Slides: 36

Chemistry in cosmetics Cosmetics : In Greek (kosmetikē tekhnē), "technique of dress and ornament" Make-up: Any substances or products used to enhance the appearance. Common cosmetics: Lipstick, mascara, eyeshadow, foundation, rouge, skin cleansers and skin lotions, shampoo, hairstyling products (gel, hair spray, etc. ) and perfume

Chemistry in cosmetics § Creams: Creams are semi-solid emulsions of water and oil or o/w. Uses: • Protect the skin • Retention of moisture • Cleansing • Emollient

Chemistry in cosmetics § Creams: • Composition: 1 - Vegetable oil or fat. 2 - Waxes such as bees wax. 3 - Lanolin (from sheep’s wool). 4 - Perfume. 5 - emulsifier. 6 - water.

§ Perfumes: Composition: 1 - Odoriferous substances: organic compounds with characteristic pleasant odors (some is synthetic) 2 - Vehicle: Solvents (usually ethanol and water mixture) 3 - Fixtive: A substance used to reduce the evaporation rate and improve stability. • Benzoin • Glycerol

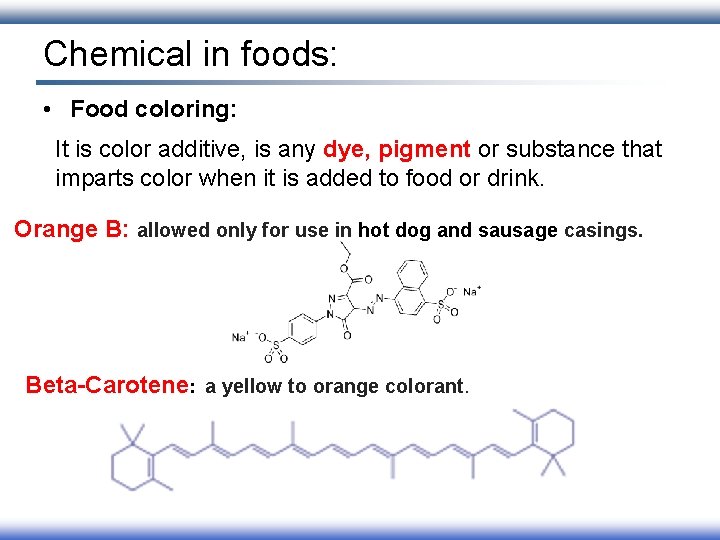
Chemical in foods: • Food coloring: It is color additive, is any dye, pigment or substance that imparts color when it is added to food or drink. Orange B: allowed only for use in hot dog and sausage casings. Beta-Carotene: a yellow to orange colorant.

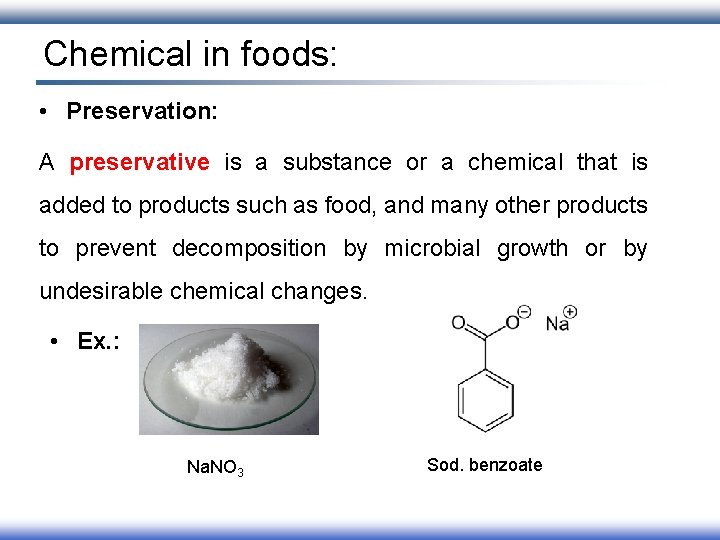
Chemical in foods: • Preservation: A preservative is a substance or a chemical that is added to products such as food, and many other products to prevent decomposition by microbial growth or by undesirable chemical changes. • Ex. : Na. NO 3 Sod. benzoate

Chemical in foods: • Antioxidant: An antioxidant is a molecule that inhibits the oxidation of other molecules. Oxidation is a chemical reaction that can produce free radicals, leading to chain reactions that may damage cells. • Ex. : • Vitamin A , C and E Dibutylhydroxytoluene (BHT)

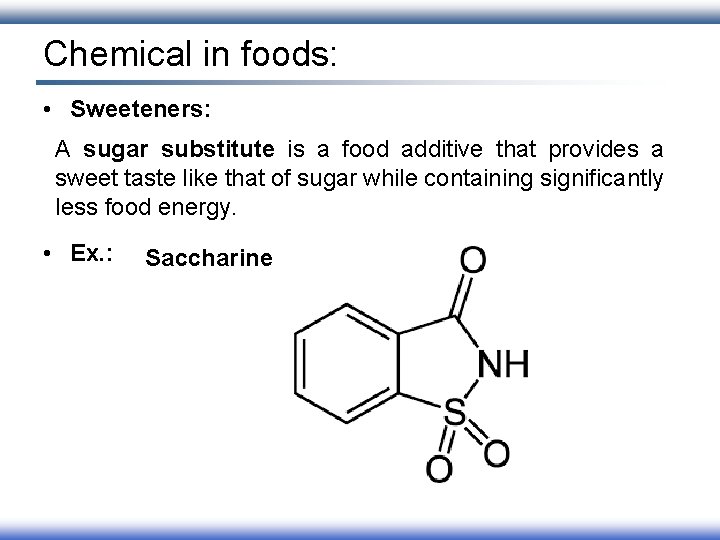
Chemical in foods: • Sweeteners: A sugar substitute is a food additive that provides a sweet taste like that of sugar while containing significantly less food energy. • Ex. : Saccharine

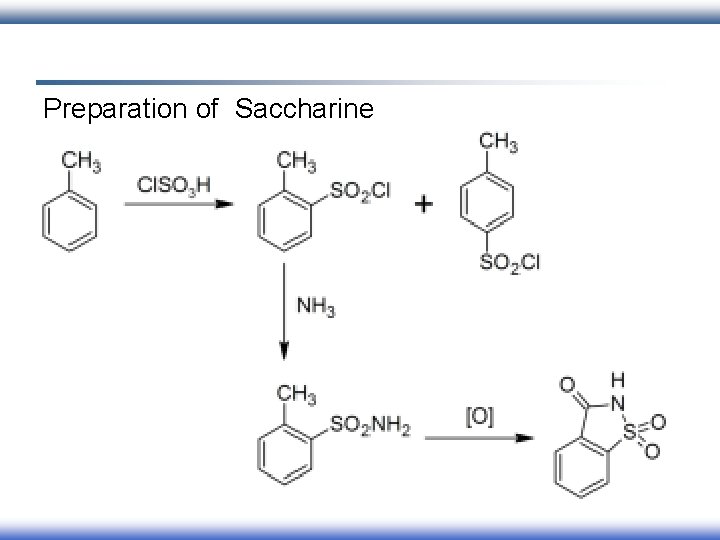
Preparation of Saccharine

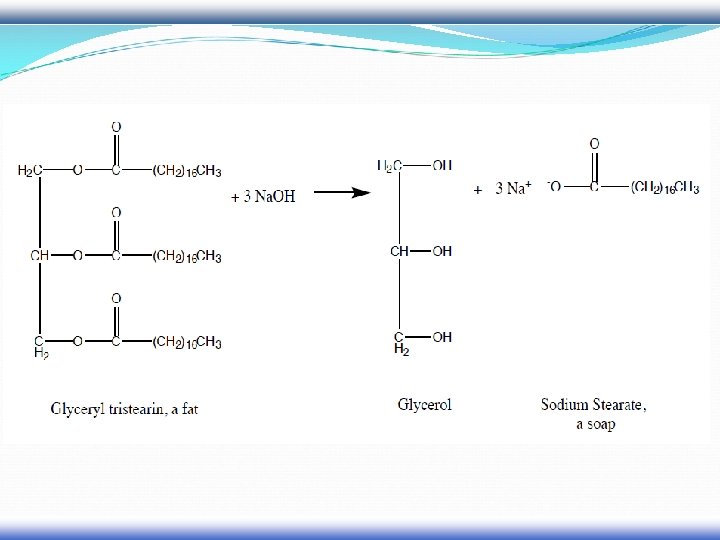
Soap and Detergents

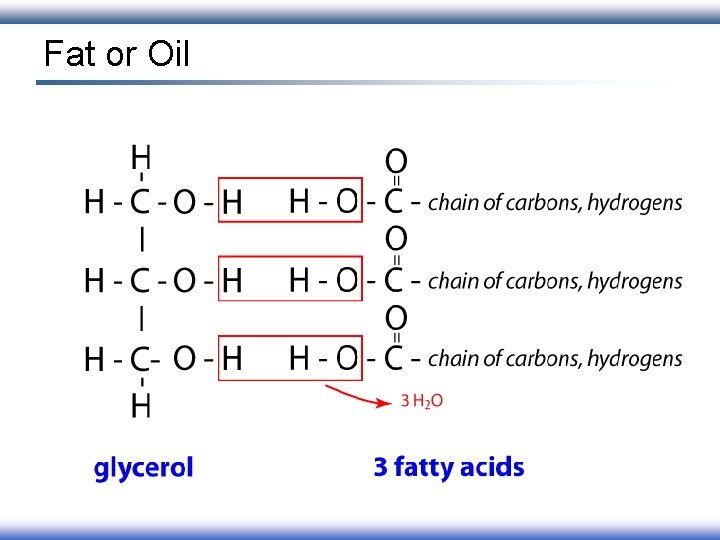
Fat or Oil

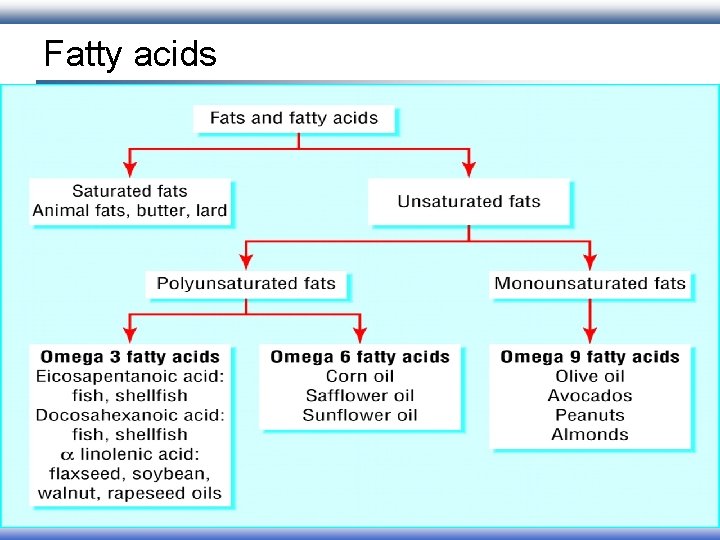
Fatty acids

Fat or Oil

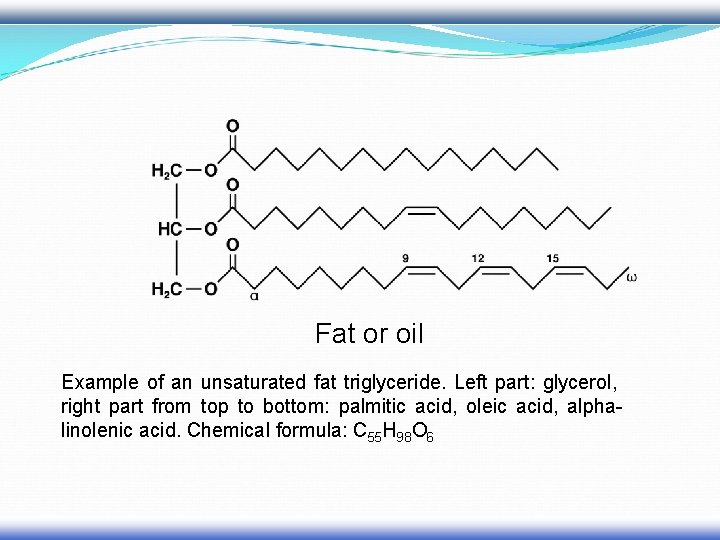
Fat or oil Example of an unsaturated fat triglyceride. Left part: glycerol, right part from top to bottom: palmitic acid, oleic acid, alphalinolenic acid. Chemical formula: C 55 H 98 O 6


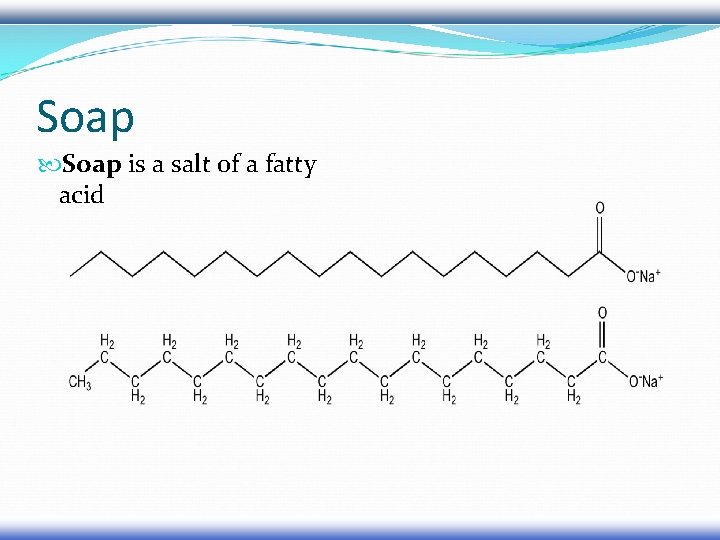
Soap is a salt of a fatty acid

Soaps are less effective in hard water, which is water that contains a significant concentration of Mg 2+ and Ca 2+ ions. These ions form precipitates with soap molecules, and this precipitate is often seen as a gray line on a bathtub or sink and is often called “soap scum”.

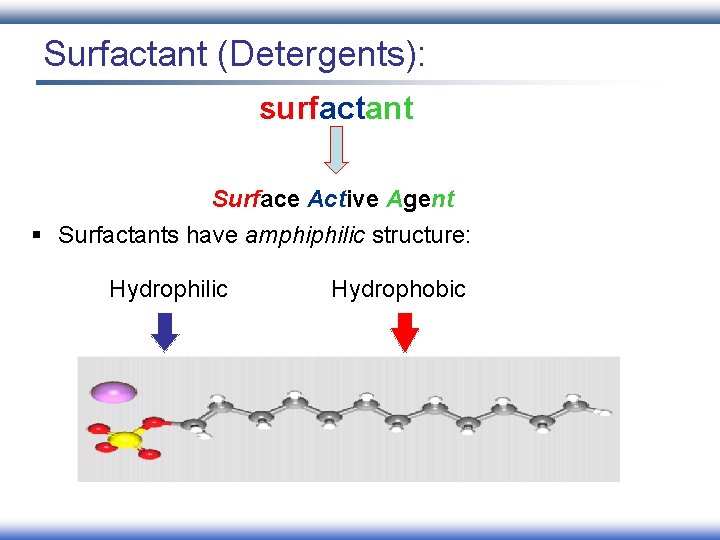
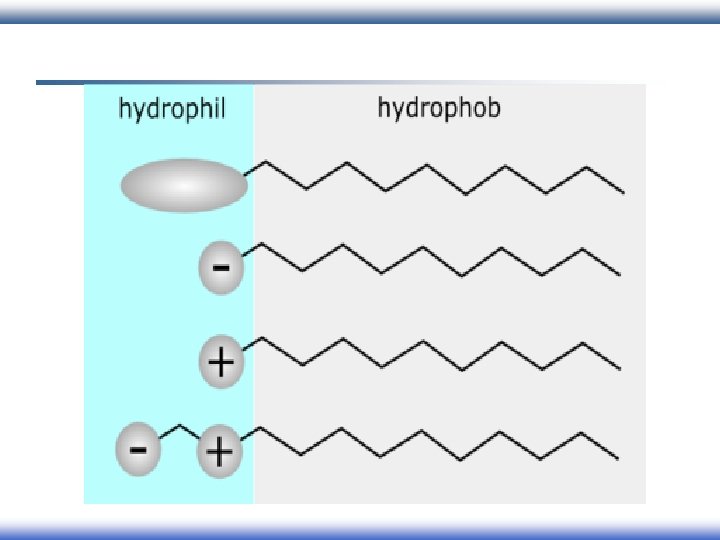
Surfactant (Detergents): surfactant Surface Active Agent § Surfactants have amphiphilic structure: Hydrophilic Hydrophobic

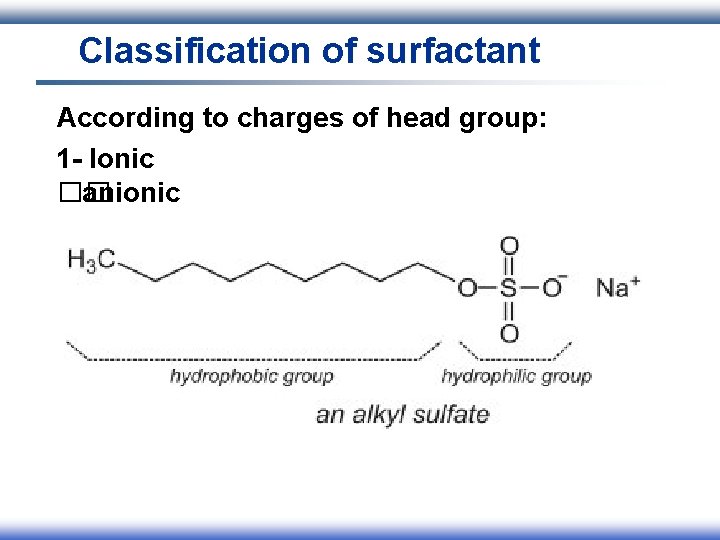
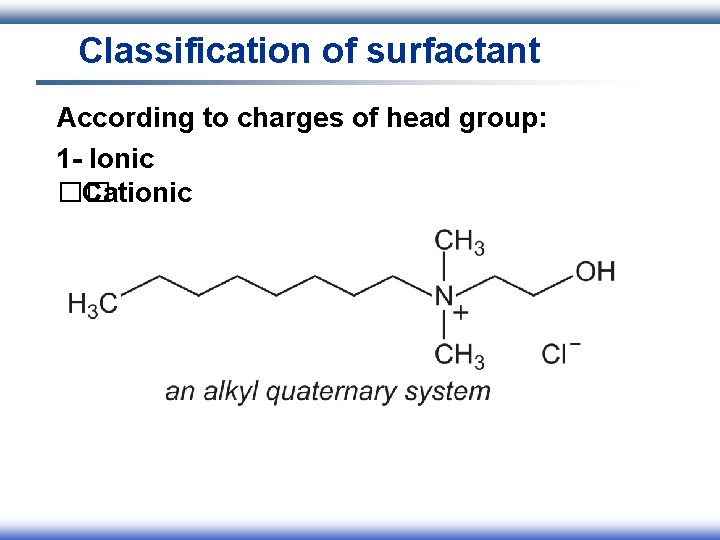
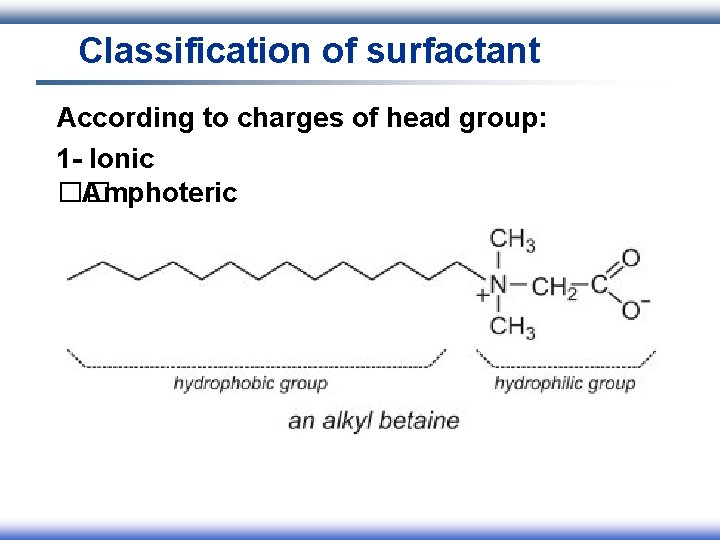
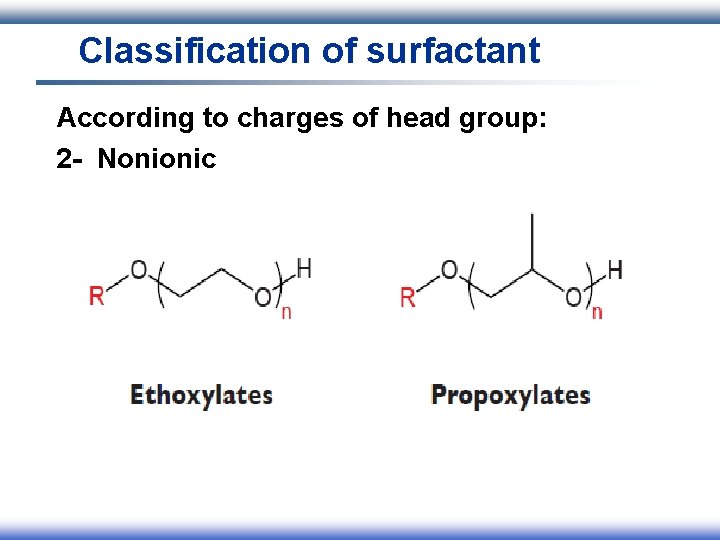
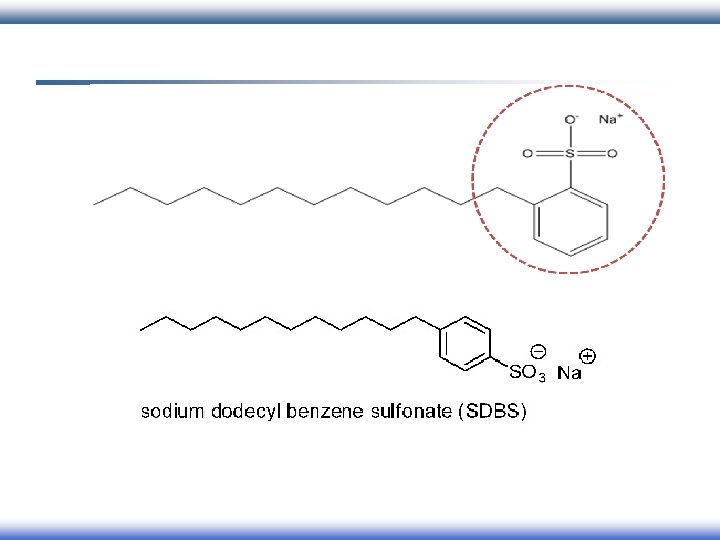
Classification of surfactant According to charges of head group: 1 - Ionic I. Anionic II. Cationic III. Amphoteric 2 - Nonionic

Classification of surfactant According to charges of head group: 1 - Ionic �� anionic

Classification of surfactant According to charges of head group: 1 - Ionic �� Cationic

Classification of surfactant According to charges of head group: 1 - Ionic �� Amphoteric

Classification of surfactant According to charges of head group: 2 - Nonionic




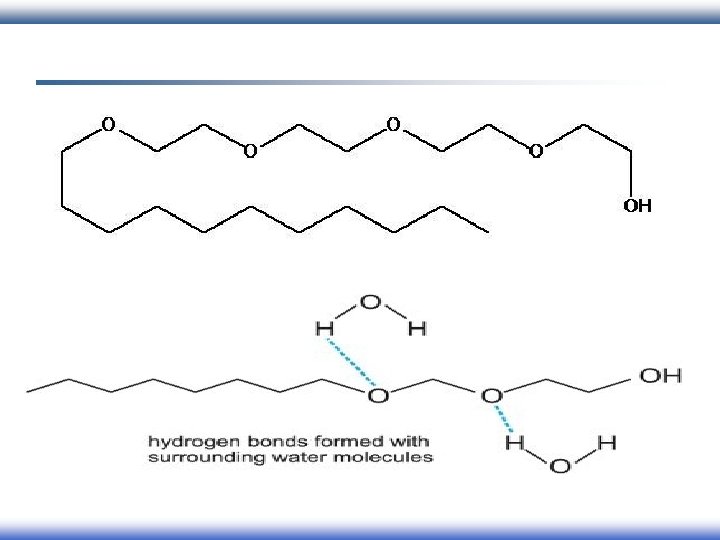
Behavior of surfactants: § When a molecule with amphiphilic structure is dissolved in aqueous medium, the hydrophobic group distorts the structure of the water. § As a result of this distortion, some of the surfactant molecules are expelled to the surfaces of the system with their hydrophobic groups oriented to minimize contact with the water molecules. Nonpolar tail Polar head

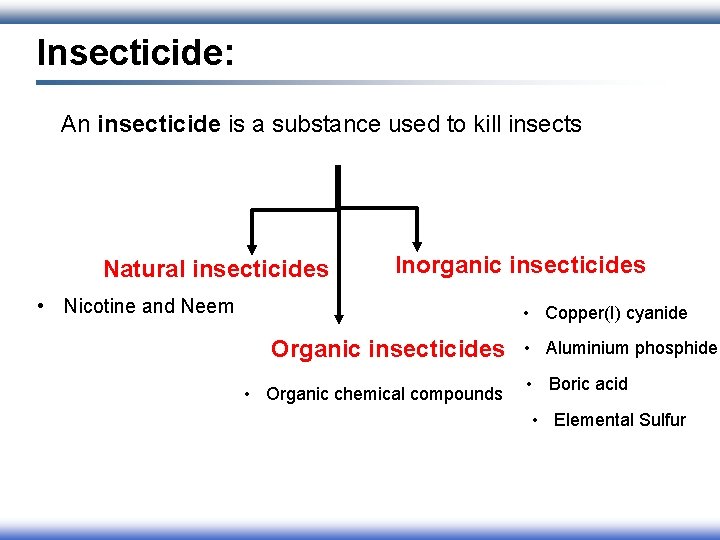
Insecticide: An insecticide is a substance used to kill insects Natural insecticides Inorganic insecticides • Nicotine and Neem • Copper(I) cyanide Organic insecticides • Organic chemical compounds • Aluminium phosphide • Boric acid • Elemental Sulfur

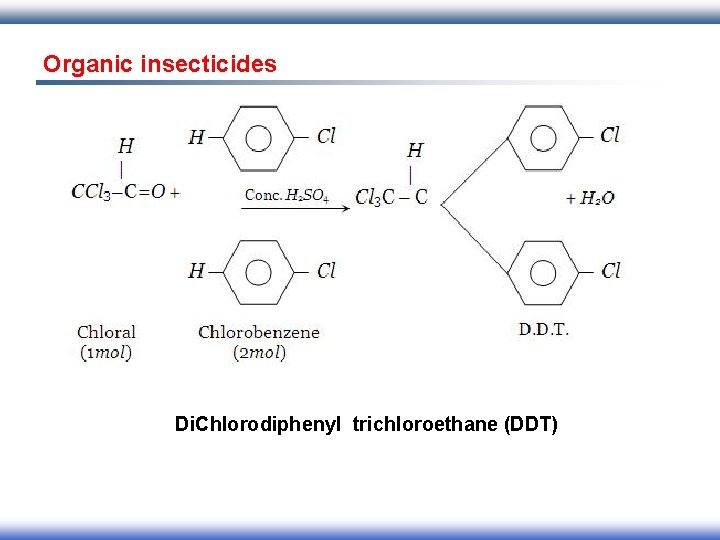
Organic insecticides Di. Chlorodiphenyl trichloroethane (DDT)

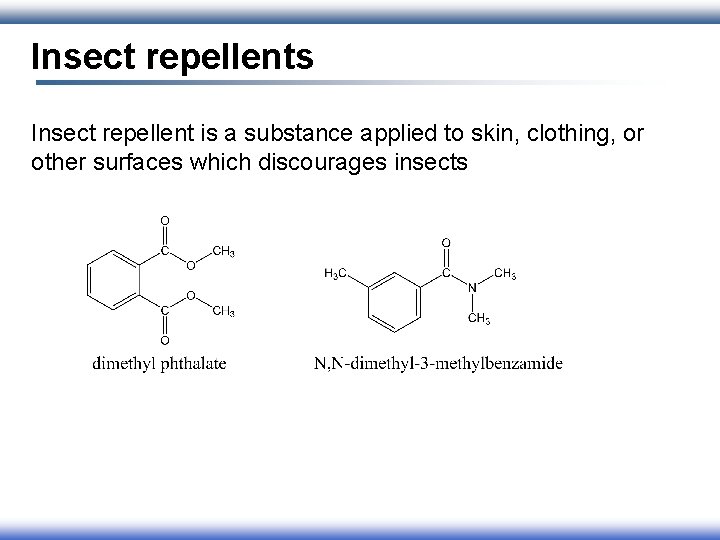
Insect repellents Insect repellent is a substance applied to skin, clothing, or other surfaces which discourages insects

Drugs 1. Analgesics and antipyretics • Analgesic drugs relieve pain. • Antipyretics are substances that reduce fever.

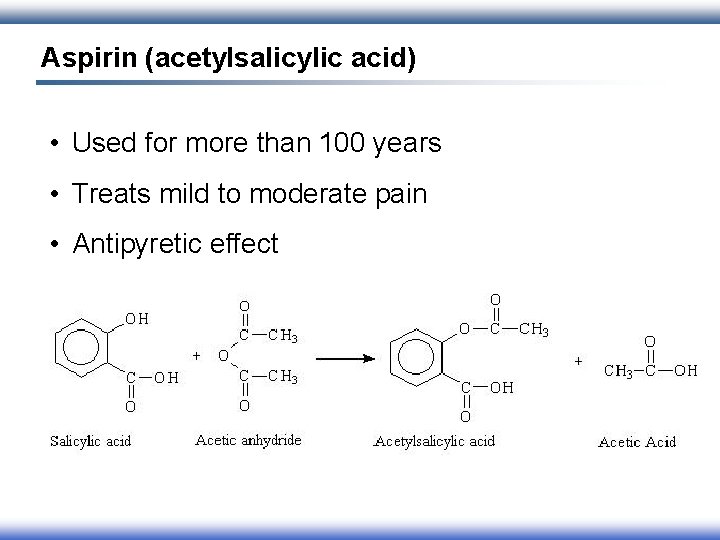
Aspirin (acetylsalicylic acid) • Used for more than 100 years • Treats mild to moderate pain • Antipyretic effect

Side Effects • Gastrointestinal irritation and bleeding • Increases bleeding time • Tinnitus • Children: Reye’s syndrome

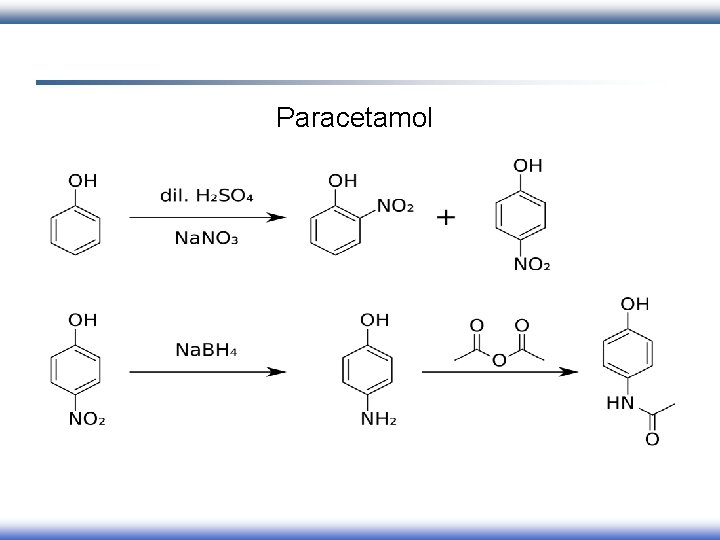
Paracetamol

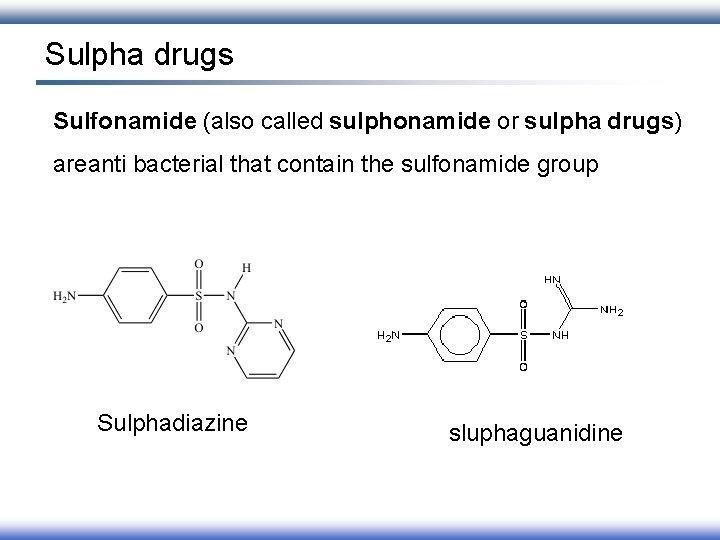
Sulpha drugs Sulfonamide (also called sulphonamide or sulpha drugs) areanti bacterial that contain the sulfonamide group Sulphadiazine sluphaguanidine

Antibiotics Penicillin • Discovered in 1928 by Alexander Fleming. • Penicillin antibiotics were among the first medications • Widely used