Chemistry Fourth Edition Julia Burdge Lecture Power Points

Chemistry Fourth Edition Julia Burdge Lecture Power. Points Chapter 3 Stoichiometry: Ratios of Combination Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education.

3. 1 Molecular and Formula Masses Using atomic masses from the periodic table and a molecular formula, we can determine the molecular mass, which is the mass in atomic mass units (amu) of an individual molecule. molecular mass of H 2 O = 2(atomic mass of H) + atomic mass of O = 2(1. 008 amu) + 16. 00 amu = 18. 02 amu Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 2

3. 1 Molecular and Formula Masses Although an ionic compound does not have a molecular mass, we can use its empirical formula to determine its formula mass. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 3

SAMPLE PROBLEM 3. 1 Calculate the molecular mass or the formula mass, as appropriate, for each of the following compounds: (a) propane, (C 3 H 8) (b) lithium hydroxide, (Li. OH) (c) barium acetate, [Ba(C 2 H 3 O 2)2] Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 4

3. 2 Percent Composition of Compounds A list of the percent by mass of each element in a compound is known as the compound’s percent composition by mass. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 5

SAMPLE PROBLEM 3. 2 Calculate the percent composition by mass of lithium carbonate (Li 2 CO 3). Setup The formula mass of Li 2 CO 3 is 2(6. 941 amu) + 12. 01 amu + 3(16. 00 amu) = 73. 89 amu Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 6

3. 3 Chemical Equations Interpreting and Writing Chemical Equations A chemical reaction, as described in the third hypothesis of Dalton’s atomic theory, is the rearrangement of atoms in a sample of matter. Examples include the rusting of iron and the explosive combination of hydrogen and oxygen gases to produce water. A chemical equation uses chemical symbols to denote what occurs in a chemical reaction. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 7

3. 3 Chemical Equations Interpreting and Writing Chemical Equations “Ammonia and hydrogen chloride react to produce ammonium chloride. ” “Calcium carbonate reacts to produce calcium oxide and carbon dioxide. ” Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 8

3. 3 Chemical Equations Interpreting and Writing Chemical Equations Each chemical species that appears to the left of the arrow is called a reactant. Reactants are those substances that are consumed in the course of a chemical reaction. Each species that appears to the right of the arrow is called a product. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 9

3. 3 Chemical Equations Interpreting and Writing Chemical Equations Indicating physical state: Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 10

3. 3 Chemical Equations Interpreting and Writing Chemical Equations Allotropes: Physical Processes Symbols above a reaction arrow indicate a process is occuring. Δ means heat is being applied. H 2 O means a substance is being dissolved in water. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 11
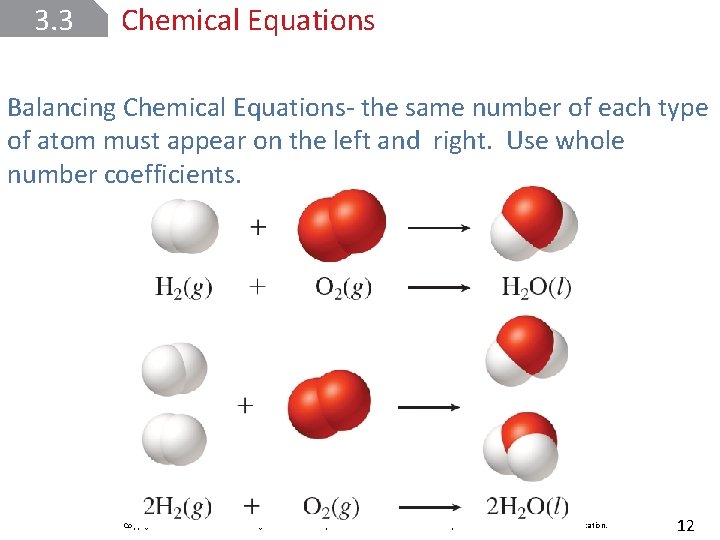
3. 3 Chemical Equations Balancing Chemical Equations- the same number of each type of atom must appear on the left and right. Use whole number coefficients. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 12

3. 3 Chemical Equations Balancing a chemical equation requires something of a trialand-error approach. In general, it will facilitate the balancing process if you do the following: 1. Change the coefficients of compounds (e. g. , CO 2) before changing the coefficients of elements (e. g. , O 2). Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 13

3. 3 Chemical Equations Balancing Chemical Equations 2. Treat polyatomic ions that appear on both sides of the equation (e. g. , CO 32–) as units, rather than counting their constituent atoms individually. 3. Count atoms and/or polyatomic ions carefully, and track their numbers each time you change a coefficient. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 14

3. 3 Chemical Equations Balancing Chemical Equations Generally, following this order make the process easier: • • • 1) polyatomic ions (if any) 2) metals 3) non-metals 4) hydrogen 5) oxygen Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 15
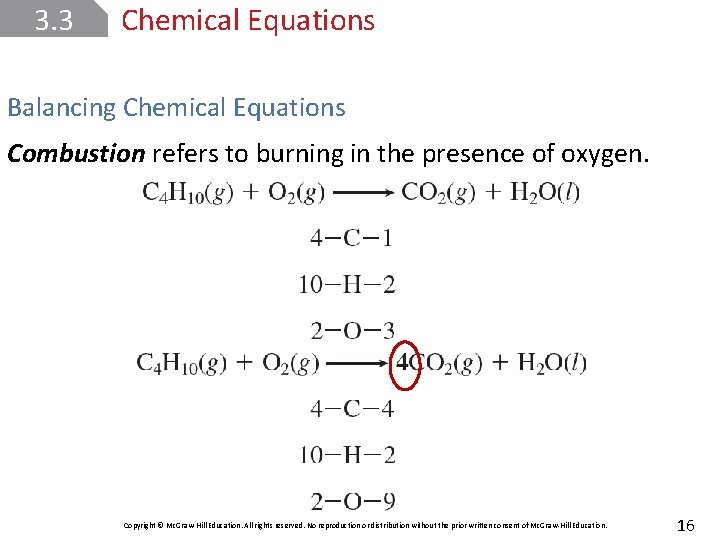
3. 3 Chemical Equations Balancing Chemical Equations Combustion refers to burning in the presence of oxygen. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 16

3. 3 Chemical Equations Balancing Chemical Equations Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 17

3. 3 Chemical Equations Balancing Chemical Equations Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 18

SAMPLE PROBLEM 3. 3 Write and balance the chemical equation for the aqueous reaction of barium hydroxide and perchloric acid to produce aqueous barium perchlorate and water. Setup Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 19
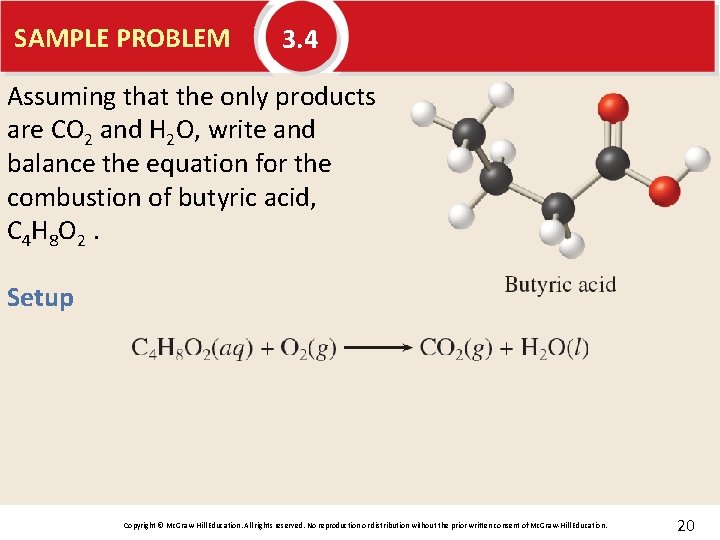
SAMPLE PROBLEM 3. 4 Assuming that the only products are CO 2 and H 2 O, write and balance the equation for the combustion of butyric acid, C 4 H 8 O 2. Setup Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 20

3. 4 The Mole and Molar Masses The Mole People use a variety of counting groups to conveniently indicate the number of objects in some set: 1 “pair” objects 2 objects 1 “dozen” objects 12 objects 1 “gross” objects 144 objects 1 “million” objects 1, 000 objects 1 “trillion” objects 1012 objects 1 “mole” objects 6. 023 × 1023 objects Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 21

3. 4 The Mole and Molar Masses The Mole A “mole” is a counting group , defined as the number of atoms in exactly 12 g of carbon-12. This number of atoms in 12 g of carbon-12 is known as Avogadro’s Number (NA): NA = 6. 0221418 × 1023 objects If you order one dozen doughnuts, you are asking for 12 doughnuts. If you order one mole of doughnuts, you are asking for 6. 022 × 1023 doughnuts! Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 22

3. 4 The Mole and Molar Masses The Mole Conversion factors for moles of objects and number of objects: 1 mol objects = 6. 022 × 1023 objects Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 23

SAMPLE PROBLEM 3. 5 A typical human body contains roughly 30 moles of calcium. Determine (a) the number of Ca atoms in 30. 00 moles of calcium and (b) the number of moles of calcium in a sample containing 1. 00 × 1020 Ca atoms. Setup Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 24

3. 4 The Mole and Molar Masses Determining Molar Mass Chemists determine how many moles there are of a substance by measuring its mass (usually in grams). The molar mass of the substance is then used to convert from grams to moles. The molar mass (M) of a substance is the mass (in grams) of 1 mole of the substance. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 25

3. 4 The Mole and Molar Masses Determining Molar Mass By definition, the mass of a mole of carbon-12 is exactly 12 g. Note that the molar mass of carbon is numerically equal to its atomic mass. Likewise, the atomic mass of calcium is 40. 08 amu and its molar mass is 40. 08 g, the atomic mass of sodium is 22. 99 amu and its molar mass is 22. 99 g, etc. Molar mass (in grams) = atomic mass (in amu). Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 26

3. 4 The Mole and Molar Masses Determining Molar Mass In effect, there is 1 mole of atomic mass units in 1 gram. So, the molar mass (in grams) of any compound is numerically equal to its molecular or formula mass (in amu). Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 27

3. 4 The Mole and Molar Masses Interconverting Mass, Moles, and Numbers of Particles Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 28

SAMPLE PROBLEM 3. 6 Determine (a) the number of moles of C in 10. 00 g of naturally occurring carbon and (b) the mass of 0. 905 mole of sodium chloride. Setup The molar mass of carbon is 12. 01 g/mol. The molar mass of a compound is numerically equal to its formula mass. The molar mass of sodium chloride (Na. Cl) is 58. 44 g/mol. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 29

SAMPLE PROBLEM 3. 7 (a) Determine the number of water molecules and the numbers of H and O atoms in 3. 26 g of water. (b) Determine the mass of 7. 92 × 1019 carbon dioxide molecules. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 30

3. 4 The Mole and Molar Masses Empirical Formula from Percent Composition With the concepts of the mole and molar mass, we can now use the experimentally determined percent composition to determine the empirical formula of a compound. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 31

SAMPLE PROBLEM 3. 8 Determine the empirical formula of a compound that is 30. 45 percent nitrogen and 69. 55 percent oxygen by mass. Strategy Assume a 100 -g sample so that the mass percentages of nitrogen and oxygen given in the problem statement correspond to the masses of N and O in the compound. Then, using the appropriate molar masses, convert the grams of each element to moles. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 32

3. 5 Combustion Analysis Determination of Empirical Formula Knowing the mass of each element contained in a sample of a substance enables us to determine the empirical formula of the substance. One common, practical use of this ability is the experimental determination of empirical formula by combustion analysis. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 33

3. 5 Combustion Analysis Determination of Empirical Formula Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 34

3. 5 Combustion Analysis Determination of Empirical Formula When a compound such as glucose is burned in a combustion analysis apparatus, carbon dioxide (CO 2) and water (H 2 O) are produced. Because only oxygen gas is added to the reaction, the carbon and hydrogen present in the products must have come from the glucose. The oxygen in the products may have come from the glucose, but it may also have come from the added oxygen. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 35

3. 5 Combustion Analysis Determination of Empirical Formula Suppose that in one such experiment the combustion of 18. 8 g of glucose produced 27. 6 g of CO 2 and 11. 3 g of H 2 O. We can calculate the mass of carbon and hydrogen in the original 18. 8 -g sample of glucose as follows: Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 36

3. 5 Combustion Analysis Determination of Empirical Formula Thus, 18. 8 g of glucose contains 7. 53 g of carbon and 1. 26 g of hydrogen. The remaining mass [18. 8 g – (7. 53 g + 1. 26 g) = 10. 01 g] is oxygen. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 37

3. 5 Combustion Analysis Determination of Empirical Formula Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 38

3. 5 Combustion Analysis Determination of Empirical Formula Now we can write a formula indicating the relative numbers of atoms in the compound C 1 H 2 O 1 or CH 2 O. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 39

3. 5 Combustion Analysis Determination of Molecular Formula The molar mass of glucose is about 180 g. The empirical-formula mass of CH 2 O is about 30 g [12. 01 g + 2(1. 008 g) + 16. 00 g]. To determine the molecular formula, first divide the molar mass by the empirical-formula mass: 180 g/30 g = 6. This tells us that there are six empirical-formula units per molecule in glucose. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 40

3. 5 Combustion Analysis Determination of Molecular Formula Multiplying each subscript by 6 (recall that when none is shown, the subscript is understood to be a 1) gives the molecular formula, C 6 H 12 O 6. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 41

SAMPLE PROBLEM 3. 9 Combustion of a 5. 50 -g sample of benzene (a hydrocarbon containing only carbon and hydrogen) produces 18. 59 g CO 2 and 3. 81 g H 2 O. Determine the empirical formula and the molecular formula of benzene, given that its molar mass is approximately 78 g/mol. Setup The necessary molar masses are CO 2, 44. 01 g/mol; H 2 O, 18. 02 g/mol; C, 12. 01 g/mol; H, 1. 008 g/mol; and O, 16. 00 g/mol. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 42
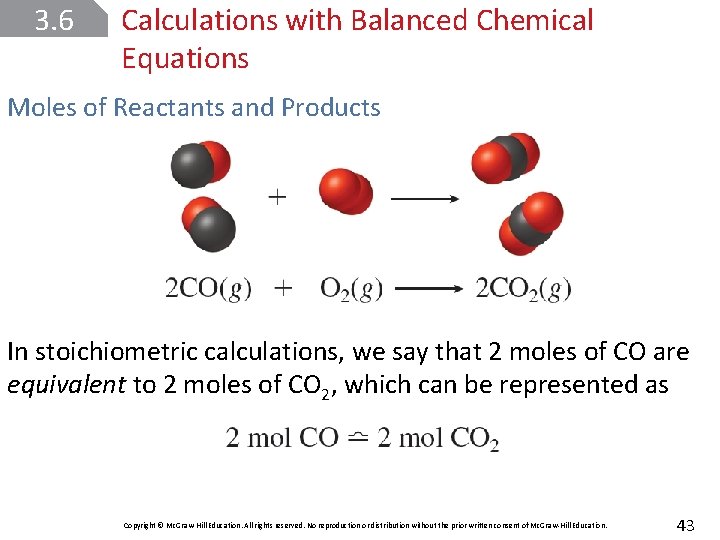
3. 6 Calculations with Balanced Chemical Equations Moles of Reactants and Products In stoichiometric calculations, we say that 2 moles of CO are equivalent to 2 moles of CO 2, which can be represented as Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 43

3. 6 Calculations with Balanced Chemical Equations Moles of Reactants and Products Stoichiometric conversion factors: Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 44

3. 6 Calculations with Balanced Chemical Equations Moles of Reactants and Products Consider the complete reaction of 3. 82 moles of CO to form CO 2. We can determine the stoichiometric amount of O 2 (how many moles of O 2 are needed to react with 3. 82 moles of CO): Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 45

SAMPLE PROBLEM 3. 10 Urea can be synthesized in the laboratory by the combination of ammonia and carbon dioxide according to the equation (a) Calculate the amount of urea that will be produced by the complete reaction of 5. 25 moles of ammonia. (b) Determine the stoichiometric amount of carbon dioxide required to react with 5. 25 moles of ammonia. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 46

3. 6 Calculations with Balanced Chemical Equations Mass of Reactants and Products Balanced chemical equations give us the relative amounts of reactants and products in terms of moles. However, because we measure reactants and products in the laboratory by weighing them, most often such calculations start with mass rather than the number of moles. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 47

SAMPLE PROBLEM 3. 11 Dinitrogen monoxide (N 2 O) (laughing gas; commonly known as nitrous oxide) is commonly used as an anesthetic in dentistry. It is manufactured by heating ammonium nitrate. The balanced equation is (a) Calculate the mass of ammonium nitrate that must be heated in order to produce 10. 0 g of nitrous oxide. (b) Determine the corresponding mass of water produced in the reaction. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 48

3. 7 Limiting Reactants Determining the Limiting Reactant The reactant used up first in a reaction is called the limiting reactant, because the amount of this reactant limits the amount of product that can form. When all the limiting reactant has been consumed, no more product can be formed. Excess reactants are those present in quantities greater than necessary to react with the quantity of the limiting reactant. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 49
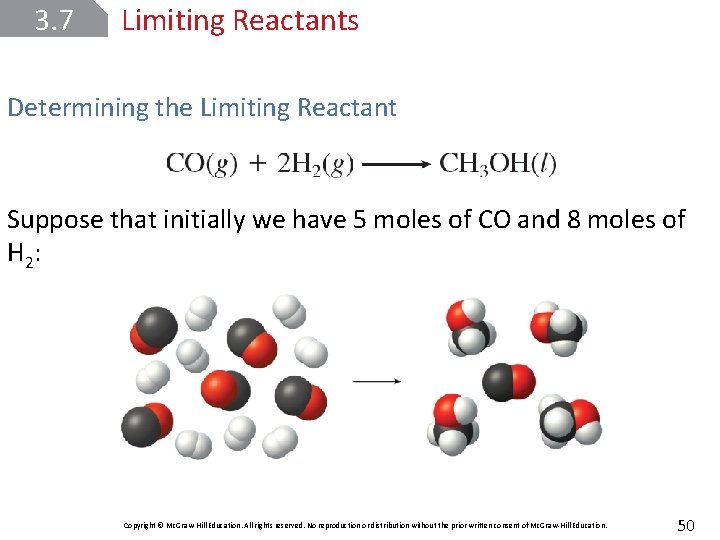
3. 7 Limiting Reactants Determining the Limiting Reactant Suppose that initially we have 5 moles of CO and 8 moles of H 2 : Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 50

3. 7 Limiting Reactants Determining the Limiting Reactant Because there are only 8 moles of H 2 available, there is insufficient H 2 to react with all the CO. Therefore, H 2 is the limiting reactant and CO is the excess reactant. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 51

3. 7 Limiting Reactants Determining the Limiting Reactant To determine how much CO will be left over when the reaction is complete, we must first calculate the amount of CO that will react with all 8 moles of H 2: Thus, there will be 4 moles of CO consumed and 1 mole (5 mol – 4 mol) left over. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 52

SAMPLE PROBLEM 3. 12 Alka-Seltzer tablets contain aspirin, sodium bicarbonate, and citric acid. When they come into contact with water, the sodium bicarbonate (Na. HCO 3) and citric acid (H 3 C 6 H 5 O 7) react to form carbon dioxide gas, among other products. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 53

SAMPLE PROBLEM 3. 12 An Alka-Seltzer tablet contains 1. 700 g of sodium bicarbonate and 1. 000 g of citric acid. Determine, for a single tablet dissolved in water, (a) which ingredient is the limiting reactant, (b) what mass of the excess reactant is left over when the reaction is complete, and (c) what mass of CO 2 forms. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 54

3. 7 Limiting Reactants Reaction Yield When you use stoichiometry to calculate the amount of product formed in a reaction, you are calculating theoretical yield of the reaction. The theoretical yield is the amount of product that forms (in grams) when all the limiting reactant reacts to form the desired product. It is the maximum obtainable yield, predicted by the balanced equation. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 55

3. 7 Limiting Reactants Reaction Yield In practice, the actual yield—the amount of product actually obtained (in grams) from a reaction—is almost always less than theoretical yield. The percent yield tells what percentage the actual yield is of theoretical yield. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 56

SAMPLE PROBLEM 3. 13 Aspirin, acetylsalicylic acid (C 9 H 8 O 4), is produced by the reaction of salicylic acid (C 7 H 6 O 3) and acetic anhydride (C 4 H 6 O 3) according to the following equation: In a certain aspirin synthesis, 104. 8 g of salicylic acid and 110. 9 g of acetic anhydride are combined. Calculate the percent yield of the reaction if 105. 6 g of aspirin are produced. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 57

3. 7 Limiting Reactants Types of Chemical Reactions Combination. A reaction in which two or more reactants combine to form a single product is known as a combination reaction. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 58

3. 7 Limiting Reactants Types of Chemical Reactions Decomposition. A reaction in which a single reactant breaks down into two or more products is known as a decomposition reaction. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 59

3. 7 Limiting Reactants Types of Chemical Reactions Combustion. A combustion reaction is one in which a substance burns in the presence of oxygen. Combustion of a compound that contains C and H (or C, H, and O) produces carbon dioxide gas and water. By convention, we will consider the water produced in a combustion reaction to be liquid water. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 60

SAMPLE PROBLEM 3. 14 Determine whether each of the following equations represents a combination reaction, a decomposition reaction, or a combustion reaction: Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 61
- Slides: 61