Chemistry Fourth Edition Julia Burdge Lecture Power Points

Chemistry Fourth Edition Julia Burdge Lecture Power. Points Chapter 2 Atoms, Molecules, and Ions Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education.

2. 1 The Atomic Theory Dalton’s Atomic Theory 1. Elements are composed of extremely small particles called atoms. All atoms of a given element are identical, having the same size, mass, and chemical properties. The atoms of one element are different from the atoms of all other elements. 2. Compounds are composed of atoms of more than one element. In any given compound, the same types of atoms are always present in the same relative numbers. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 2

2. 1 The Atomic Theory Dalton’s Atomic Theory 3. A chemical reaction rearranges atoms in chemical compounds; it does not create or destroy them. This is known as the “law of conservation of mass”. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 3
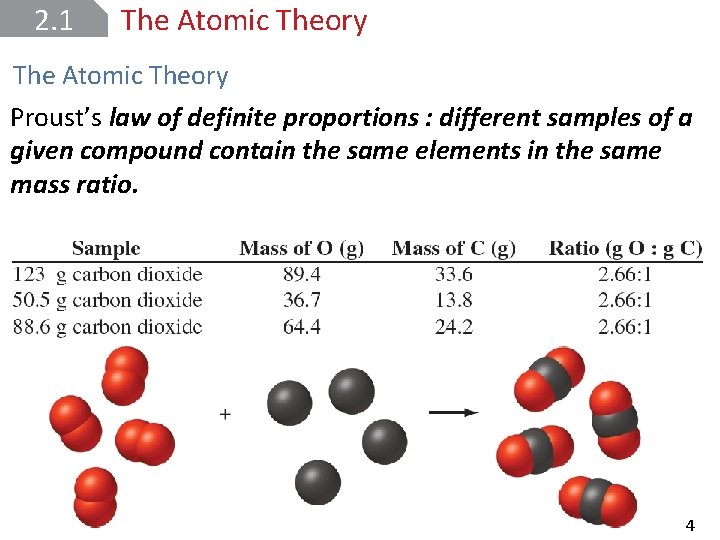
2. 1 The Atomic Theory Proust’s law of definite proportions : different samples of a given compound contain the same elements in the same mass ratio. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 4
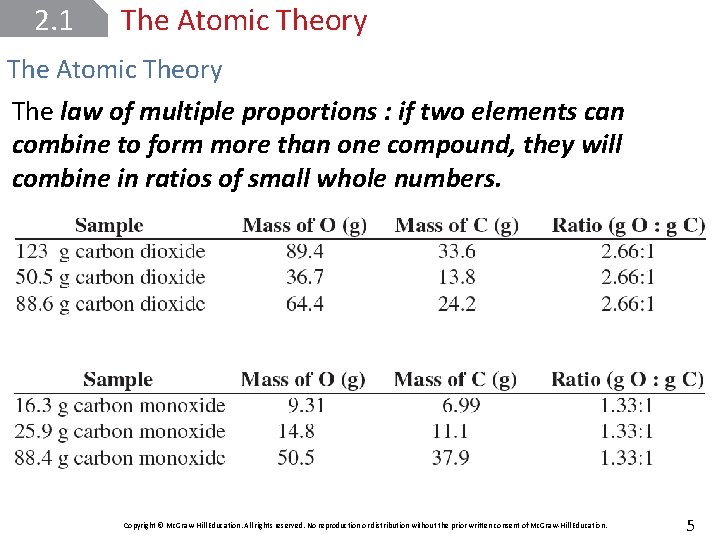
2. 1 The Atomic Theory The law of multiple proportions : if two elements can combine to form more than one compound, they will combine in ratios of small whole numbers. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 5
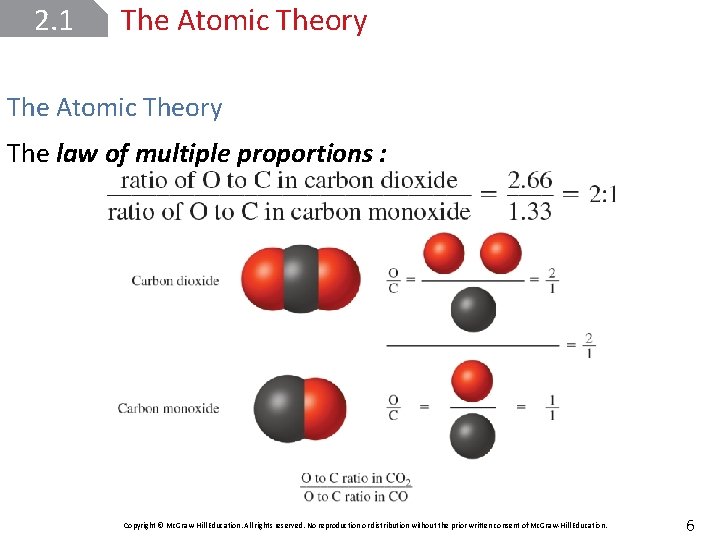
2. 1 The Atomic Theory The law of multiple proportions : Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 6

2. 2 The Structure of the Atom Discovery of the Electron The use of a cathode ray tube led to the discovery of the electron. Because the cathode rays are deflected by magnetic and electric fields, they must be streams of charged particles. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 7

2. 2 The Structure of the Atom Discovery of the Electron Because the cathode ray is deflected away from the negative pole of a bar magnet, and toward the positive pole, it must consist of negatively charged particles. We call these electrons. Using this apparatus, J. J. Thompson was able to calculate the ratio of an electron’s charge to its mass; 1. 76 x 108 C/g, where C stands for coulomb, or A*s. © The Mc. Graw-Hill Companies, Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction orphotographer distribution without the prior written consent of Mc. Graw-Hill Education. Inc. /Charles D. Winters, 8
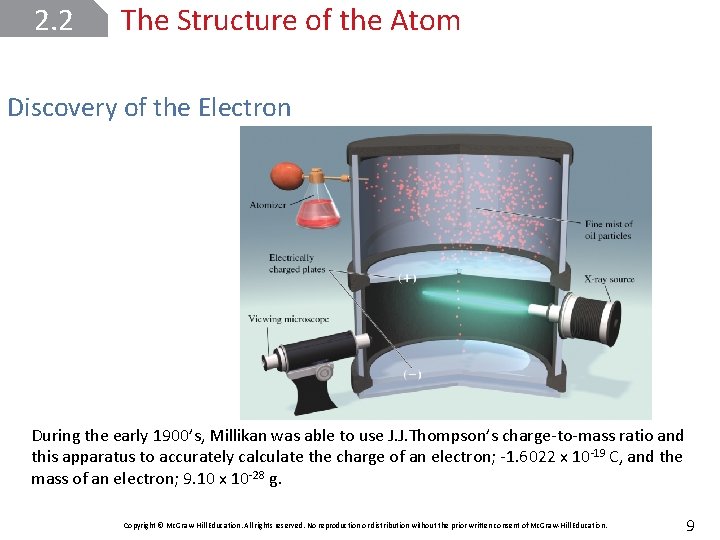
2. 2 The Structure of the Atom Discovery of the Electron During the early 1900’s, Millikan was able to use J. J. Thompson’s charge-to-mass ratio and this apparatus to accurately calculate the charge of an electron; -1. 6022 x 10 -19 C, and the mass of an electron; 9. 10 x 10 -28 g. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 9

2. 2 The Structure of the Atom Discovery of the Electron J. J. Thomson: Charge-to-mass ratio of the electron Millikan: Charge of the electron Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 10

2. 2 The Structure of the Atom The Proton and the Nucleus Rutherford’s Experiment Because scientists knew that atoms contained electrons, but were electrically neutral, they must contain some source of positive charge. Rutherford decided to experiment with atoms to see where this positive charge was located. He shot alpha particles (+) through thin sheets of gold foil. Most went straight through, but some were deflected by large angles, and some bounced straight back. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 11

2. 2 The Structure of the Atom Nuclear Model of the Atom The atom’s positive charges, Rutherford proposed, were all concentrated in the nucleus, which is an extremely dense central core within the atom. The positively charged particles in the nucleus are called protons. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 12
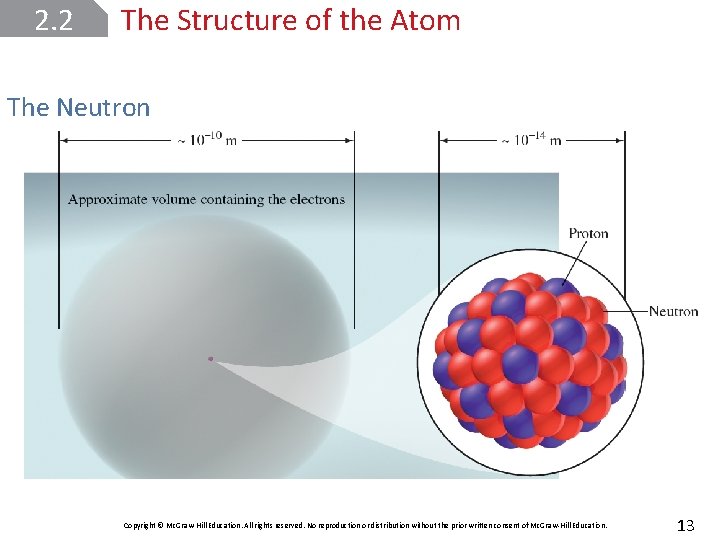
2. 2 The Structure of the Atom The Neutron Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 13
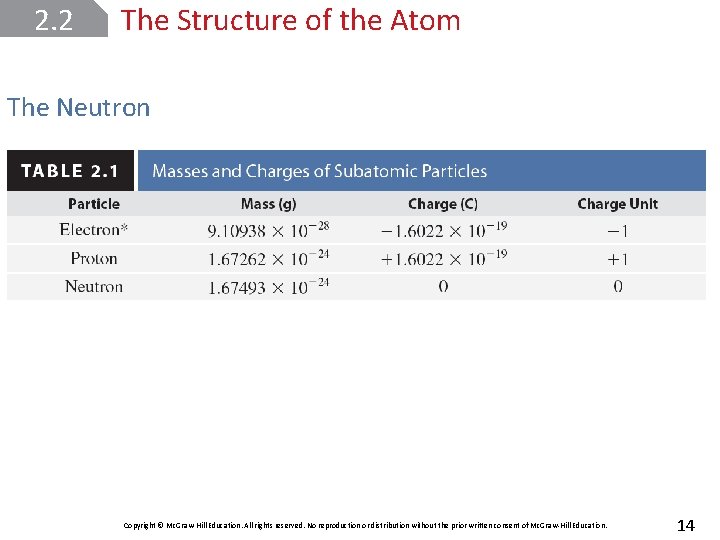
2. 2 The Structure of the Atom The Neutron Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 14

2. 3 Atomic Number, Mass Number, and Isotopes The atomic number (Z) is the number of protons in the nucleus of each atom of an element. The mass number (A) is the total number of neutrons and protons present in the nucleus of an atom of an element. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 15
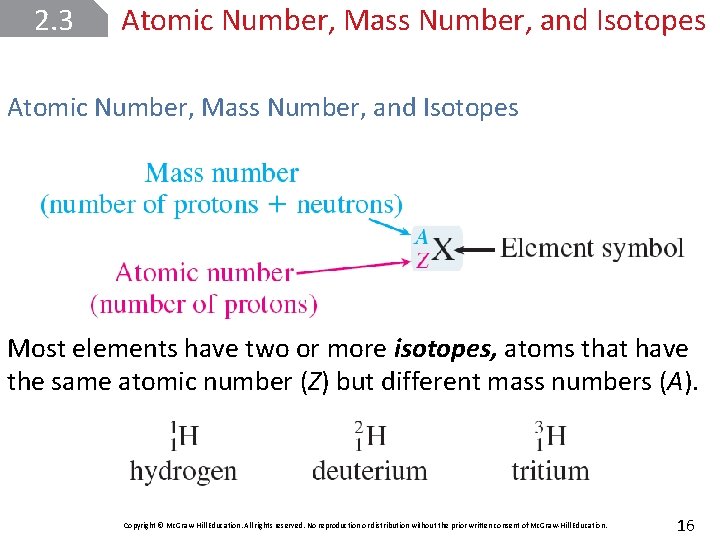
2. 3 Atomic Number, Mass Number, and Isotopes Most elements have two or more isotopes, atoms that have the same atomic number (Z) but different mass numbers (A). Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 16

2. 3 Atomic Number, Mass Number, and Isotopes 235 – 92 = 143 neutrons 238 – 92 = 146 neutrons Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 17

SAMPLE PROBLEM 2. 2 Determine the numbers of protons, neutrons, and electrons in each of the following species: Setup Number of protons = Z, number of neutrons = A – Z, and number of electrons = number of protons. Recall that the 14 in carbon-14 is the mass number. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 18

SAMPLE PROBLEM 2. 2 Solution (a) The atomic number is 17, so there are 17 protons. The mass number is 35, so the number of neutrons is 35 – 17 = 18. The number of electrons equals the number of protons, so there are 17 electrons. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 19

SAMPLE PROBLEM 2. 2 Solution (b) Again, the atomic number is 17, so there are 17 protons. The mass number is 37, so the number of neutrons is 37 – 17 = 20. The number of electrons equals the number of protons, so there are 17 electrons, too. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 20

SAMPLE PROBLEM 2. 2 Solution (c) The atomic number of K (potassium) is 19, so there are 19 protons. The mass number is 41, so there are 41 – 19 = 22 neutrons. There are 19 electrons. (d) Carbon-14 can also be represented as 14 C. The atomic number of carbon is 6, so there are 6 protons and 6 electrons. There are 14 – 6 = 8 neutrons. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 21
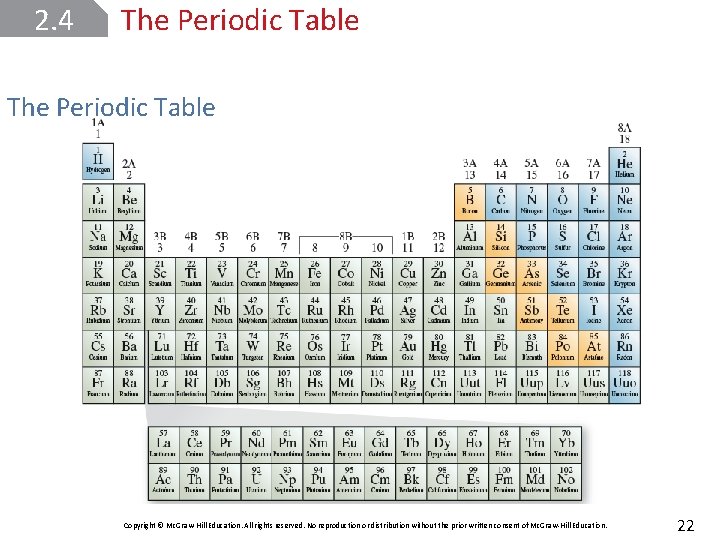
2. 4 The Periodic Table Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 22

2. 4 The Periodic Table Elements are arranged in horizontal rows called periods and in vertical columns called groups or families. Elements in the same group tend to have similar physical and chemical properties. The elements can be categorized as metals, nonmetals, or metalloids. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 23

2. 4 The Periodic Table A metal is a good conductor of heat and electricity, whereas a nonmetal is usually a poor conductor of heat and electricity. A metalloid has properties that are intermediate between those of metals and nonmetals. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 24
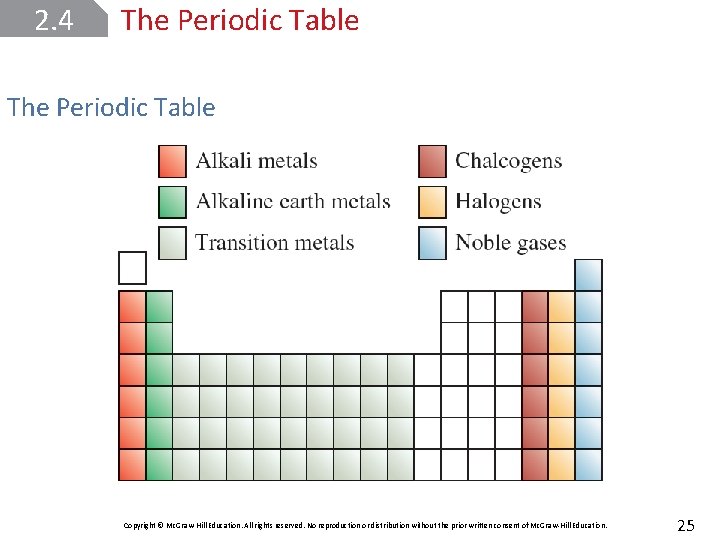
2. 4 The Periodic Table Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 25

2. 5 The Atomic Mass Scale and Average Atomic Mass The Periodic Table According to international agreement, atomic mass is the mass of an atom in atomic mass units. One atomic mass unit (amu) is defined as a mass exactly equal to one-twelfth the mass of one carbon-12 atom. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 26

2. 5 The Atomic Mass Scale and Average Atomic Mass The Periodic Table Experiments have shown that a hydrogen atom (1 H) is only 8. 3985 percent as massive as the carbon-12 atom. Thus, if the mass of one carbon-12 atom is exactly 12 amu, the atomic mass of hydrogen must be 0. 083985 × 12 amu, or 1. 0078 amu. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 27
2. 5 The Atomic Mass Scale and Average Atomic Mass The Periodic Table When you look up the atomic mass of carbon in a table such as the on the inside front cover of the text, you will find that its value is 12. 01 amu, not 12. 00 amu. The difference arises because most naturally occurring elements (including carbon) have more than one isotope. The atomic masses in the periodic table are average atomic masses. The term atomic weight is sometimes used to mean average atomic mass. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 28
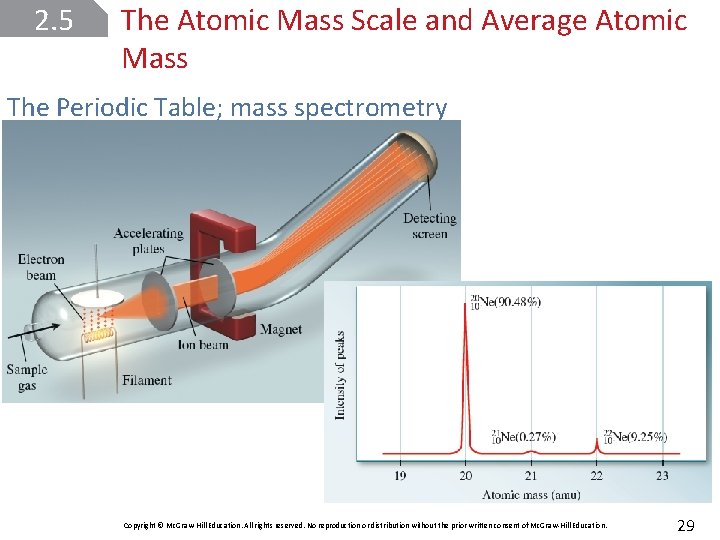
2. 5 The Atomic Mass Scale and Average Atomic Mass The Periodic Table; mass spectrometry Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 29

SAMPLE PROBLEM 2. 3 Oxygen is the most abundant element in both Earth’s crust and the human body. The atomic masses of its three stable isotopes, 168 O (99. 757 percent), 178 O (0. 038 percent), and 188 O (0. 205 percent), are 15. 9949, 16. 9991, and 17. 9992 amu, respectively. Calculate the average atomic mass of oxygen using the relative abundances given in parentheses. Report the result to four significant figures. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 30

SAMPLE PROBLEM 2. 3 Solution Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 31

2. 6 Ions and Ionic Compounds Atomic Ions An atomic ion or monatomic ion is one that consists of just one atom with a positive or negative charge. The loss of one or more electrons from an atom yields a cation, an ion with a net positive charge. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 32

2. 6 Ions and Ionic Compounds Atomic Ions An anion is an ion whose net charge is negative due to an increase in the number of electrons. Sodium chloride (Na. Cl), ordinary table salt, is called an ionic compound because it consists of cations (Na+) and anions (Cl–). Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 33
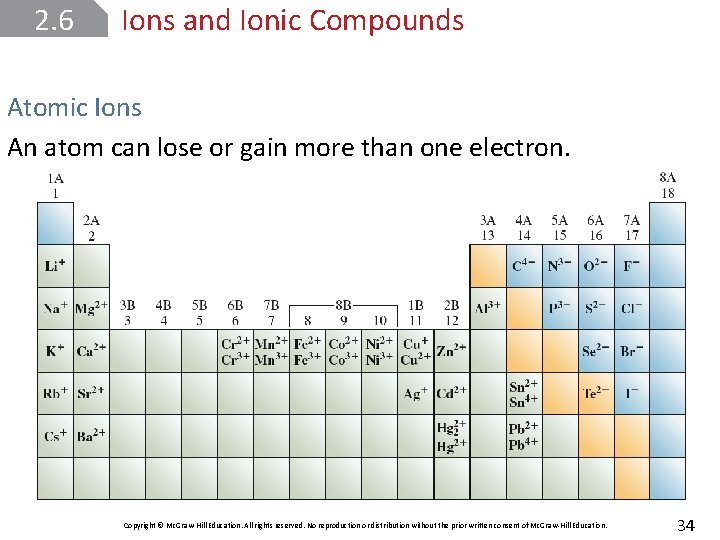
2. 6 Ions and Ionic Compounds Atomic Ions An atom can lose or gain more than one electron. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 34

2. 6 Ions and Ionic Compounds Atomic Ions For metals that can have different cations, designate which with Roman numerals, using the Stock system. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 35

2. 6 Ions and Ionic Compounds Atomic Ions An older nomenclature system – for cations with two possible charges - assigns the ending –ous to the cation with the smaller positive charge and the ending –ic to the cation with the greater positive charge: Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 36

2. 6 Ions and Ionic Compounds Atomic Ions A monatomic anion is named by changing the ending of the element’s name to –ide, and adding the word ion. Thus, the anion of chlorine (Cl–), is called chloride ion. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 37

2. 6 Ions and Ionic Compounds Polyatomic Ions that consist of a combination of two or more atoms are called polyatomic ions; a tightly bound group of atoms that stays together and carries a charge. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 38
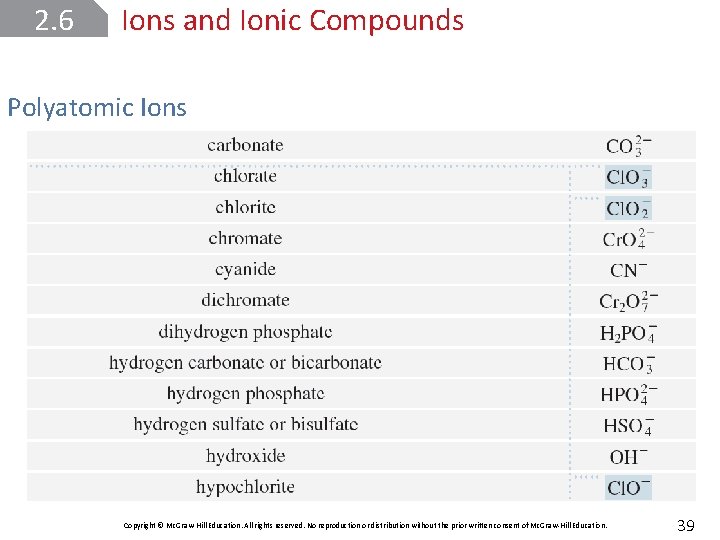
2. 6 Ions and Ionic Compounds Polyatomic Ions Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 39
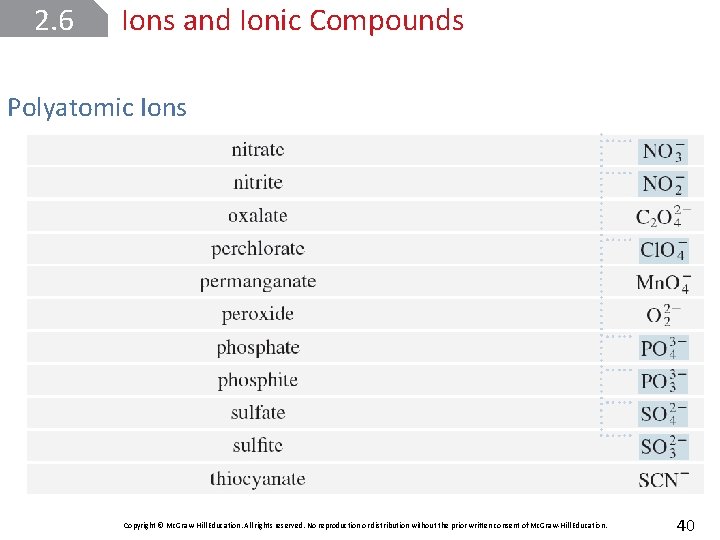
2. 6 Ions and Ionic Compounds Polyatomic Ions Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 40
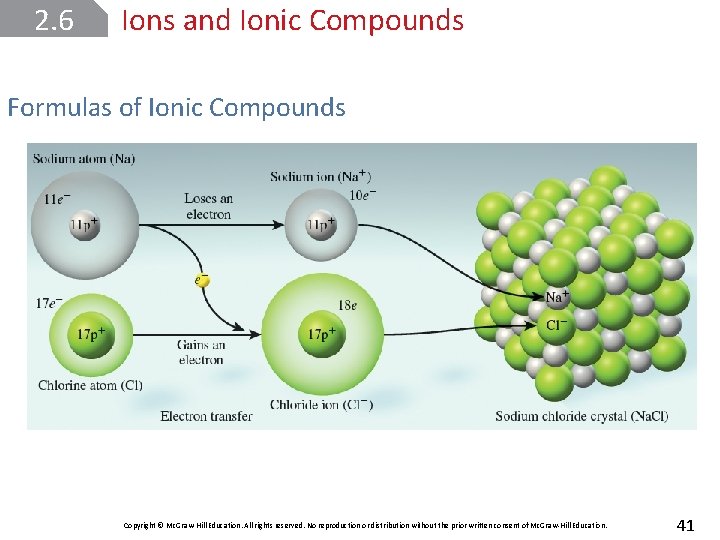
2. 6 Ions and Ionic Compounds Formulas of Ionic Compounds Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 41

2. 6 Ions and Ionic Compounds Formulas of Ionic Compounds Aluminum Oxide Calcium Phosphate Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 42

2. 6 Ions and Ionic Compounds Naming Ionic Compounds Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 43

SAMPLE PROBLEM 2. 4 Name the following ionic compounds: (a) Mg. O (b) Al(OH)3 (c) Fe 2(SO 4)3 Setup (a) Mg 2+ O 2– (b) Al 3+ OH– (c) Since the charge on SO 42– is – 2, the charge on Fe must be +3: Fe 3+ Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 44

SAMPLE PROBLEM 2. 4 Name the following ionic compounds: (a) Mg. O (b) Al(OH)3 (c) Fe 2(SO 4)3 Solution (a) magnesium oxide (b) aluminum hydroxide (c) iron (III) sulfate Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 45

SAMPLE PROBLEM 2. 5 Deduce the formulas of the following ionic compounds: (a) mercury(I) chloride (b) lead(II) chromate (c) potassium hydrogen phosphate Setup (a) Hg 22+ (b) Pb 2+ (c) K+ Cl– Cr. O 42– HPO 42– Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 46

SAMPLE PROBLEM 2. 5 Deduce the formulas of the following ionic compounds: (a) mercury(I) chloride (b) lead(II) chromate (c) potassium hydrogen phosphate Solution (a) Hg 2 Cl 2 (b) Pb. Cr. O 4 (c) K 2 HPO 4 Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 47

2. 6 Ions and Ionic Compounds Oxoanions are polyatomic anions that contain one or more oxygen atoms and one atom (the “central atom”) of another element. Examples include the chlorate (Cl. O 3– ), nitrate (NO 3– ), and sulfate (SO 42–) ions. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 48

2. 6 Ions and Ionic Compounds Oxoanions Starting with the oxoanions whose names end in –ate, we can name these ions as follows: 1. The ion with one more O atom than the –ate ion is called the per. . . ate ion. Thus, Cl. O 3– is the chlorate ion, so Cl. O 4– is the perchlorate ion. 2. The ion with one less O atom than the –ate anion is called the –ite ion. Thus, Cl. O 2– is the chlorite ion. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 49

2. 6 Ions and Ionic Compounds Oxoanions Starting with the oxoanions whose names end in –ate, we can name these ions as follows: 3. The ion with two fewer O atoms than the –ate ion is called the hypo. . . ite ion. Thus, Cl. O– is the hypochlorite ion. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 50

SAMPLE PROBLEM 2. 6 Name the following species: (a) Br. O 4– (b) HCO 3– (c) SO 32 Setup (a) Br. O 3– is the bromate ion. (b) CO 32– is the carbonate ion. (c) SO 42 - is the sulfate ion. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 51

SAMPLE PROBLEM 2. 6 Name the following species: (a) Br. O 4– (b) HCO 3– (c) SO 32 Solution (a) perbromate ion (b) hydrogen carbonate ion (or bicarbonate ion) (c) sulfite ion Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 52

2. 6 Ions and Ionic Compounds Hydrates are compounds that have a specific number of water molecules within their solid structure. © The Mc. Graw-Hill Companies, Inc. /Charles D. Winters, photographer Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 53

2. 7 Molecules and Molecular Compounds Molecules A molecule is a neutral combination of at least two nonmetal atoms in a specific arrangement held together by electrostatic forces known as covalent chemical bonds. The hydrogen molecule, symbolized as H 2, is called a diatomic molecule because it contains two atoms. Other elements that normally exist as diatomic molecules are nitrogen (N 2), oxygen (O 2), and the Group 7 A elements— fluorine (F 2), chlorine (Cl 2), bromine (Br 2), and iodine (I 2). KNOW THESE! Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 54

2. 7 Molecules and Molecular Compounds Molecules In homonuclear diatomic molecules, both atoms in each molecule are of the same element. (These are the 7 diatomic elements. ) A diatomic molecule can also contain atoms of different elements (heteronuclear diatomic molecules). Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 55

2. 7 Molecules and Molecular Compounds Molecular Formulas A chemical formula denotes the composition of the substance. A molecular formula shows the exact number of atoms of each element in a molecule. An allotrope is one of two or more distinct forms of an element. Two of the allotropic forms of the element carbon— diamond and graphite—have dramatically different properties. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 56

2. 7 Molecules and Molecular Compounds Molecular Formulas The structural formula shows not only the elemental composition, but also the general arrangement of atoms within the molecule. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 57
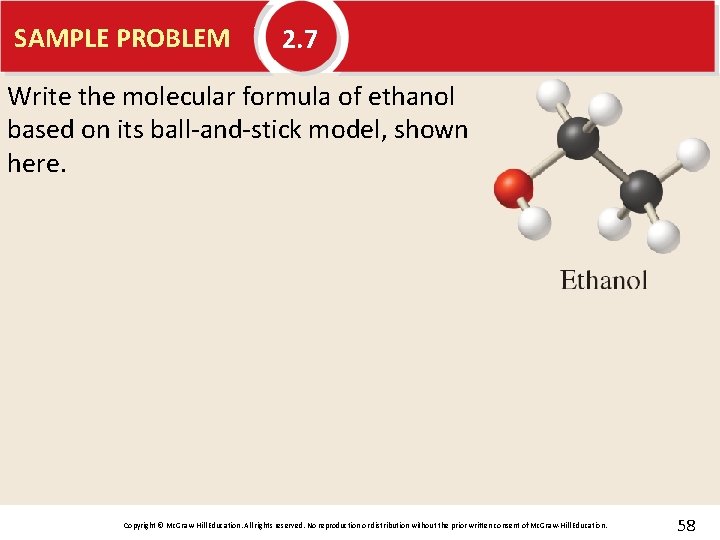
SAMPLE PROBLEM 2. 7 Write the molecular formula of ethanol based on its ball-and-stick model, shown here. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 58
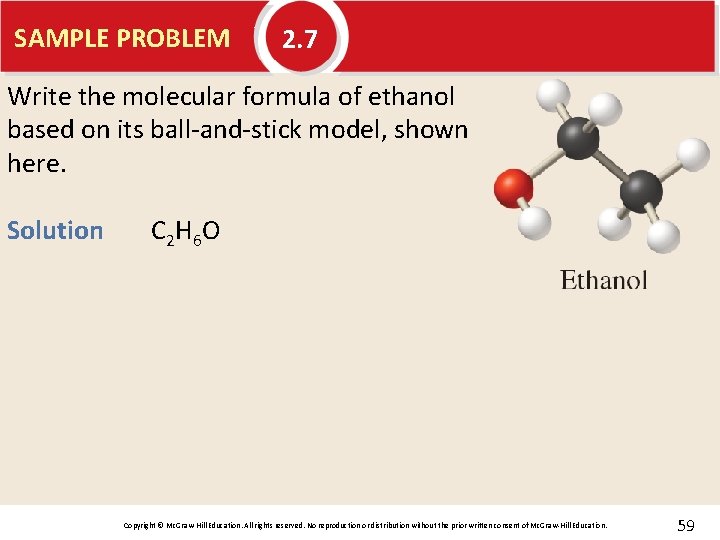
SAMPLE PROBLEM 2. 7 Write the molecular formula of ethanol based on its ball-and-stick model, shown here. Solution C 2 H 6 O Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 59

2. 7 Molecules and Molecular Compounds Naming Molecular Compounds Binary molecular compounds consist of just two different nonmetal elements. To name such a compound, we first name the element that appears first in the formula. We then name the second element, changing the ending of its name to –ide. HCl Si. C hydrogen chloride silicon carbide Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 60
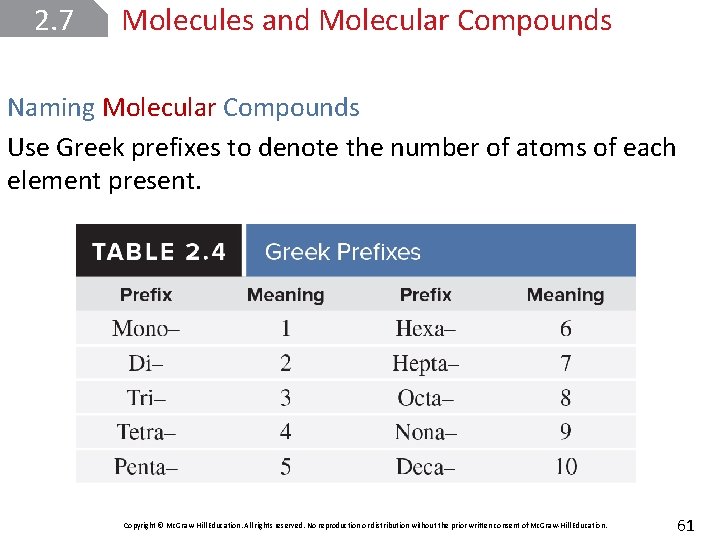
2. 7 Molecules and Molecular Compounds Naming Molecular Compounds Use Greek prefixes to denote the number of atoms of each element present. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 61
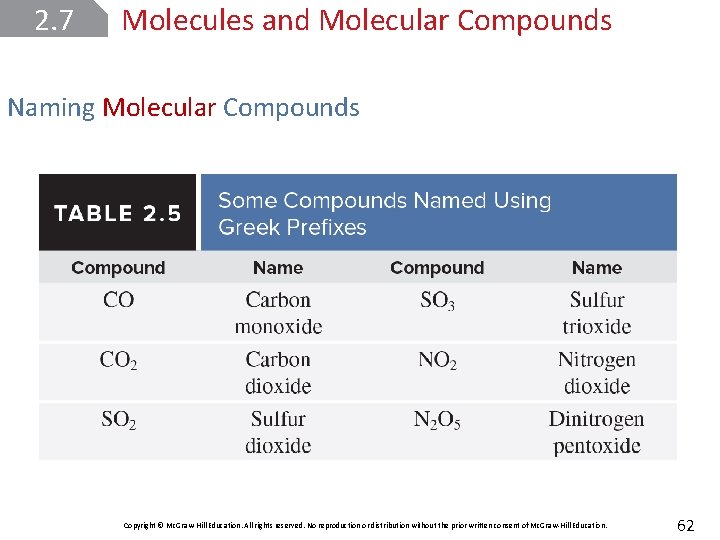
2. 7 Molecules and Molecular Compounds Naming Molecular Compounds Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 62

2. 7 Molecules and Molecular Compounds Naming Molecular Compounds The prefix mono– is generally omitted for the first element. SO 2, for example, is named sulfur dioxide, not monosulfur dioxide. For ease of pronunciation, we usually eliminate the last letter of a prefix that ends in “o” or “a” when naming an oxide. Thus, N 2 O 5 is dinitrogen pentoxide, rather than dinitrogen pentaoxide. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 63

SAMPLE PROBLEM 2. 8 Name the following binary molecular compounds: (a) NF 3 and (b) N 2 O 4. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 64

SAMPLE PROBLEM 2. 8 Name the following binary molecular compounds: (a) NF 3 and (b) N 2 O 4. Solution (a) nitrogen trifluoride and (b) dinitrogen tetroxide Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 65

SAMPLE PROBLEM 2. 9 Write the chemical formulas for the following binary molecular compounds: (a) sulfur tetrafluoride and (b) tetraphosphorus decasulfide. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 66

SAMPLE PROBLEM 2. 9 Write the chemical formulas for the following binary molecular compounds: (a) sulfur tetrafluoride and (b) tetraphosphorus decasulfide. Solution (a) SF 4 and (b) P 4 S 10 Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 67

2. 7 Molecules and Molecular Compounds Naming Molecular Compounds The names of molecular compounds containing hydrogen do not usually conform to the systematic nomenclature guidelines; we use common names. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 68

2. 7 Molecules and Molecular Compounds Simple Acids make up another important class of molecular compounds. One definition of an acid is a substance that produces hydrogen ions (H+) when dissolved in water. Several binary molecular compounds produce hydrogen ions when dissolved in water and are, therefore, acids. In these cases, two different names can be assigned to the same chemical formula. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 69

2. 7 Molecules and Molecular Compounds Simple Acids For example, HCl, hydrogen chloride, is a gaseous compound. HCl(g) When it is dissolved in water, however, we call it hydrochloric acid. HCl(aq) The rules for naming simple acids of this type are as follows: remove the –gen ending from hydrogen (leaving hydro–), change the –ide ending on the second element to –ic, combine the two words, and add the word acid. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 70
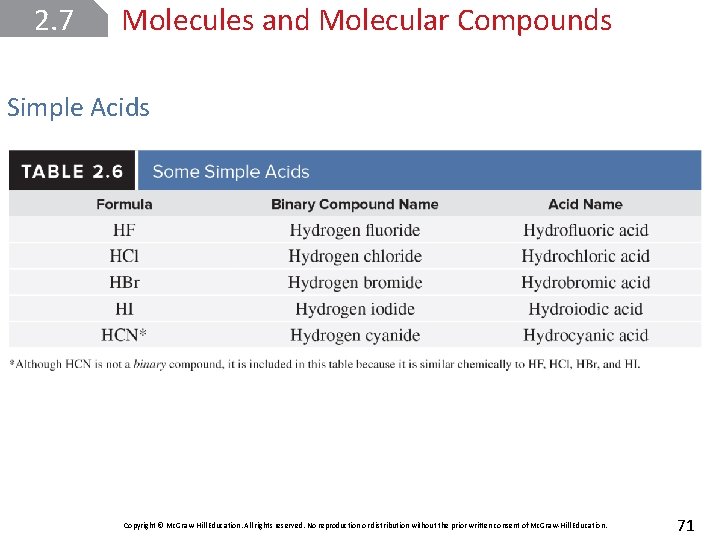
2. 7 Molecules and Molecular Compounds Simple Acids Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 71

2. 7 Molecules and Molecular Compounds Oxoacids An important class of acids known as oxoacids, which ionize to produce hydrogen ions and the corresponding oxoanions. The formula of an oxoacid can be determined by adding enough H+ ions to the corresponding oxoanion to yield a formula with no net charge. For example, the formulas of oxoacids based on the nitrate (NO 3– ) and sulfate (SO 42–) ions are HNO 3 and H 2 SO 4, respectively. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 72

2. 7 Molecules and Molecular Compounds Oxoacids The names of oxoacids are derived from the names of the corresponding oxoanions using the following guidelines: 1. An acid based on an –ate ion is called. . . ic acid. Thus, HCl. O 3 is called chloric acid. 2. An acid based on an –ite ion is called. . . ous acid. Thus, HCl. O 2 is called chlorous acid. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 73

2. 7 Molecules and Molecular Compounds Oxoacids The names of oxoacids are derived from the names of the corresponding oxoanions using the following guidelines: 3. Prefixes in oxoanion names are retained in the names of the corresponding oxoacids. Thus, HCl. O 4 and HCl. O are called perchloric acid and hypochlorous acid, respectively. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 74

2. 7 Molecules and Molecular Compounds Oxoacids Many oxoacids, such as H 2 SO 4 and H 3 PO 4, are polyprotic— meaning that they have more than one ionizable hydrogen atom. In these cases, the names of anions in which one or more (but not all) of the hydrogen ions have been removed must indicate the number of H ions that remain. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 75

SAMPLE PROBLEM 2. 10 Determine the formula of sulfurous acid. Setup The sulfite ion is SO 32–. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 76

SAMPLE PROBLEM 2. 10 Determine the formula of sulfurous acid. Solution The formula of sulfurous acid is H 2 SO 3. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 77

2. 8 Compounds in Review Compounds can be made up of ions and molecules. It is important that you be able to determine whether a compound is ionic or molecular from its chemical formula. By convention, we use Greek prefixes to indicate ratios of combination in molecular compounds, but not in ionic compounds—because knowing the charges on the constituent ions means that ionic compound names without prefixes are unambiguous. Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 78
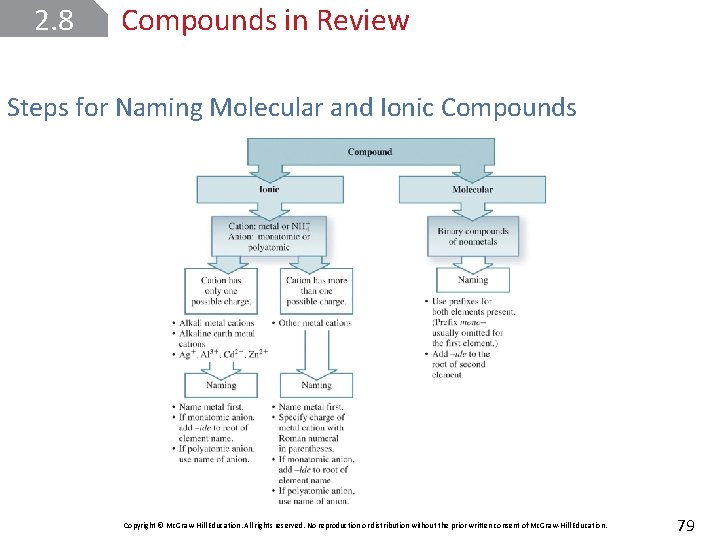
2. 8 Compounds in Review Steps for Naming Molecular and Ionic Compounds Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 79
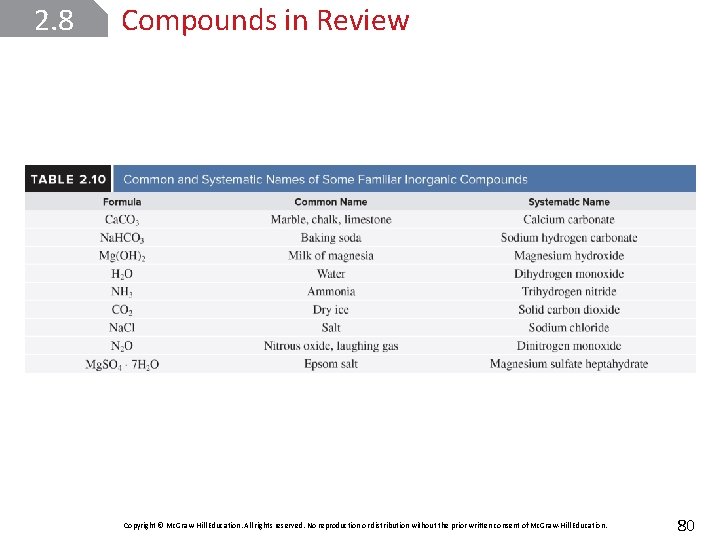
2. 8 Compounds in Review Copyright © Mc. Graw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of Mc. Graw-Hill Education. 80
- Slides: 80