Chemistry for Biologists Chemistry for Biologists Define the

Chemistry for Biologists

Chemistry for Biologists Define the following: • Element • Atom • Proton • Neutron • Electron • Isotope

Chemistry for Biologists • An element is a substance made up of atoms with the same number of protons. Elements are the simplest substances known. They can be metals (e. g. iron, copper, sodium magnesium) or non-metals (e. g. carbon, hydrogen, oxygen, nitrogen). There are just over 100 of them.

Chemistry for Biologists • Atoms are the building blocks of all matter. They consist of three sub-atomic particles: protons, neutrons and electrons.
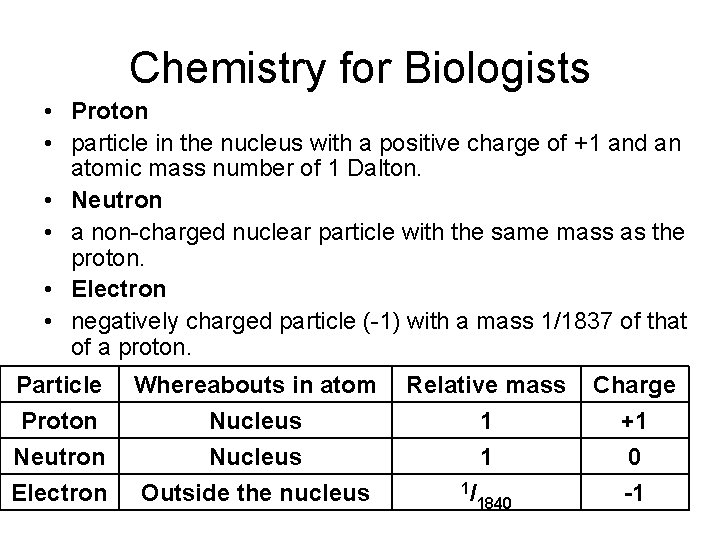
Chemistry for Biologists • Proton • particle in the nucleus with a positive charge of +1 and an atomic mass number of 1 Dalton. • Neutron • a non-charged nuclear particle with the same mass as the proton. • Electron • negatively charged particle (-1) with a mass 1/1837 of that of a proton. Particle Proton Neutron Electron Whereabouts in atom Nucleus Outside the nucleus Relative mass 1 1 1/ 1840 Charge +1 0 -1

• Many elements have different isotopes. Although the numbers of protons and electrons are the same in all atoms of a given element, the number of neutrons may differ.
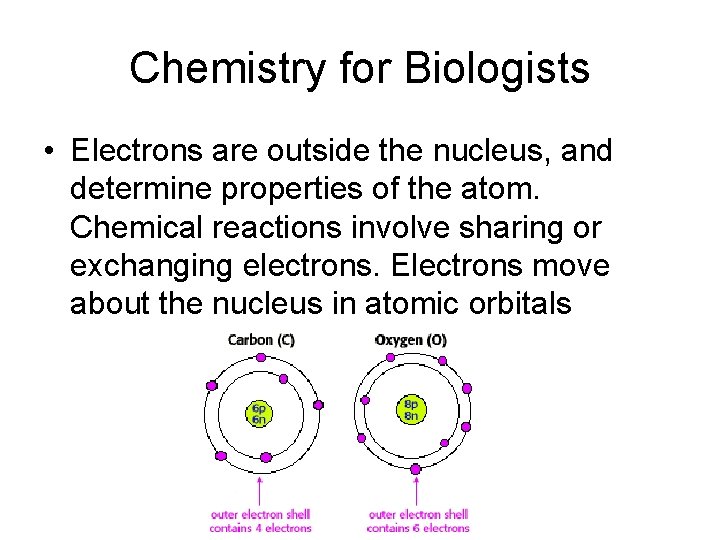
Chemistry for Biologists • Electrons are outside the nucleus, and determine properties of the atom. Chemical reactions involve sharing or exchanging electrons. Electrons move about the nucleus in atomic orbitals

Chemistry for Biologists • A molecule is two or more atoms linked by a chemical bond. • Molecules can contain different types of bonds. What are these?
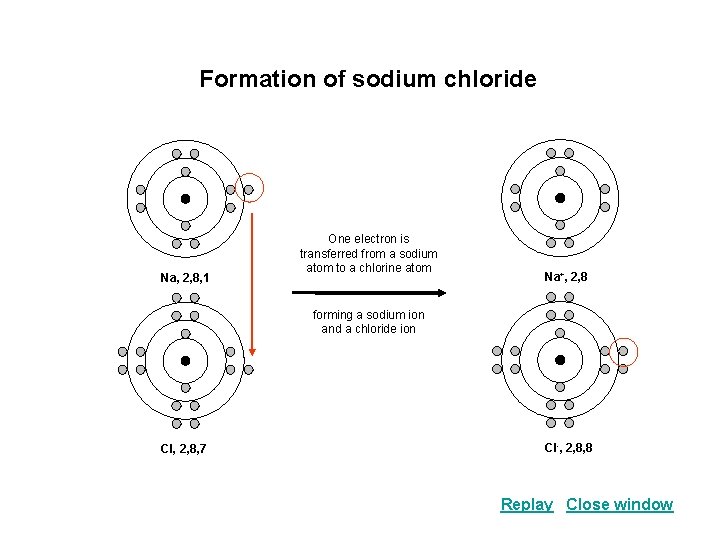
Formation of sodium chloride Na, 2, 8, 1 One electron is transferred from a sodium atom to a chlorine atom Na+, 2, 8 forming a sodium ion and a chloride ion Cl, 2, 8, 7 Cl-, 2, 8, 8 Replay Close window
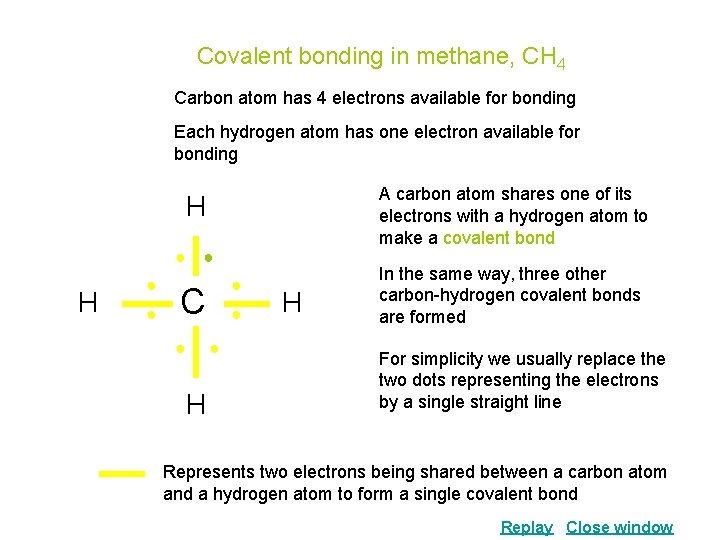
Covalent bonding in methane, CH 4 Carbon atom has 4 electrons available for bonding Each hydrogen atom has one electron available for bonding A carbon atom shares one of its electrons with a hydrogen atom to make a covalent bond H ● ● ● C ● ● ● H H In the same way, three other carbon-hydrogen covalent bonds are formed For simplicity we usually replace the two dots representing the electrons by a single straight line Represents two electrons being shared between a carbon atom and a hydrogen atom to form a single covalent bond Replay Close window
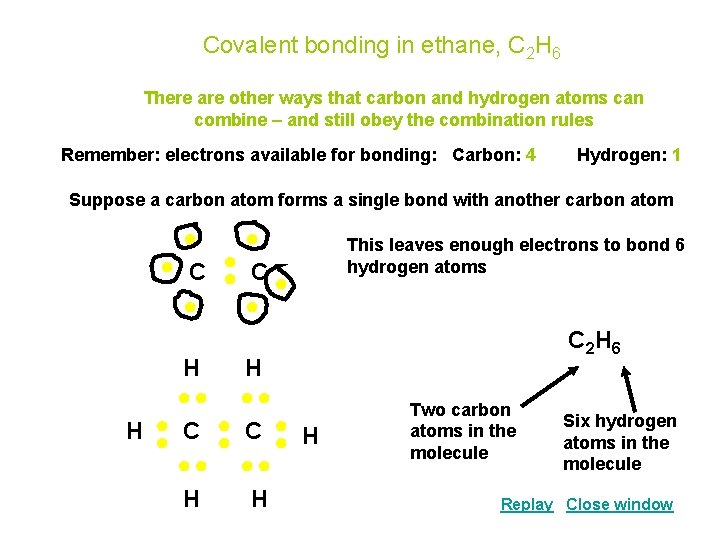
Covalent bonding in ethane, C 2 H 6 There are other ways that carbon and hydrogen atoms can combine – and still obey the combination rules Remember: electrons available for bonding: Carbon: 4 Hydrogen: 1 Suppose a carbon atom forms a single bond with another carbon atom ● ● ● C● ● ● H H ●● ●● ● C ● H H ● C ● ●● ●● H H This leaves enough electrons to bond 6 hydrogen atoms C 2 H 6 Two carbon atoms in the molecule Six hydrogen atoms in the molecule Replay Close window

Chemistry for Biologists Non-covalent bonds and other weak forces are important in biological structures.

Electrostatic bonds (ionic) Result from the electrostatic attraction between two ionized groups of opposite charge, such as carboxyl (-COO-) and amino (-NH 3+). In water, these bonds are very weak. Ionic attractions are important in stabilising the three dimensional shapes of proteins

Hydrogen bonds Result from electrostatic attraction between an electronegative atom (O or N) and a hydrogen atom that is bonded covalently to a second electronegative atom. N-H ----- O=C- -O-H----- O=C-

Van Der Waals These bonds-are short range attractive forces between chemical groups in contact. Caused by slight charge displacements.

• Hydrophobic attractions • These cause non-polar groups such as hydrocarbon chains to associate with each other in an aqueous environment. • Multiple weak bonds or forces can cause strong interactions

Chemistry for Biologists Organic molecules contain carbon Alcohol hydrocarbon with a hydrogen replaced by "OH". A hydroxyl group Acid hydrocarbon with a hydrogen replaced by "COOH". A carboxyl group This disassociates in solution to form COOH -> COO- + H+

Chemistry for Biologists Amine A hydrocarbon with a hydrogen replaced by an amine "NH 2". Basic- accepts protons. NH 2 + H+ -> NH 3+ Phosphate The addition of -PO 4 • Amino Acid A hydrocarbon with amino and carboxyl groups

Chemistry for Biologists Chemical groups found in organic molecules Group name • Aldehyde group CHO • Keto group C=O • Hydroxyl group COH • Carboxyl group COOH • Primary alcohol group
- Slides: 19