Chemistry FIFTH EDITION by Steven S Zumdahl University

Chemistry FIFTH EDITION by Steven S. Zumdahl University of Illinois Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 1

Chemistry FIFTH EDITION Chapter 17 Electrochemistry Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 2
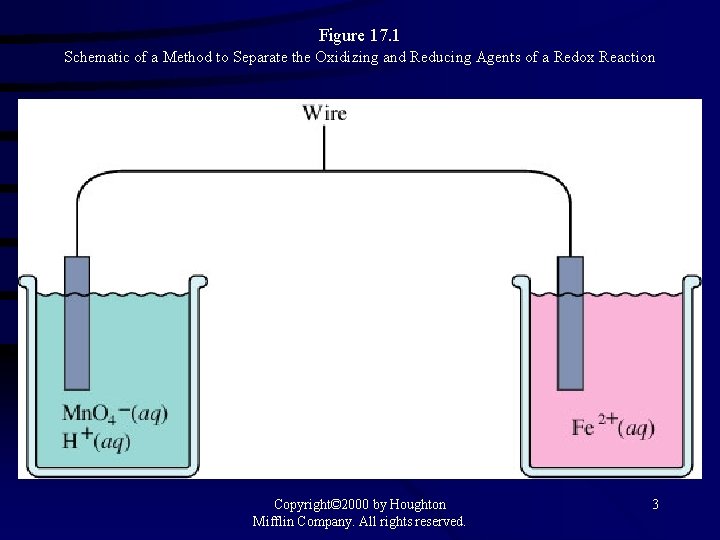
Figure 17. 1 Schematic of a Method to Separate the Oxidizing and Reducing Agents of a Redox Reaction Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 3
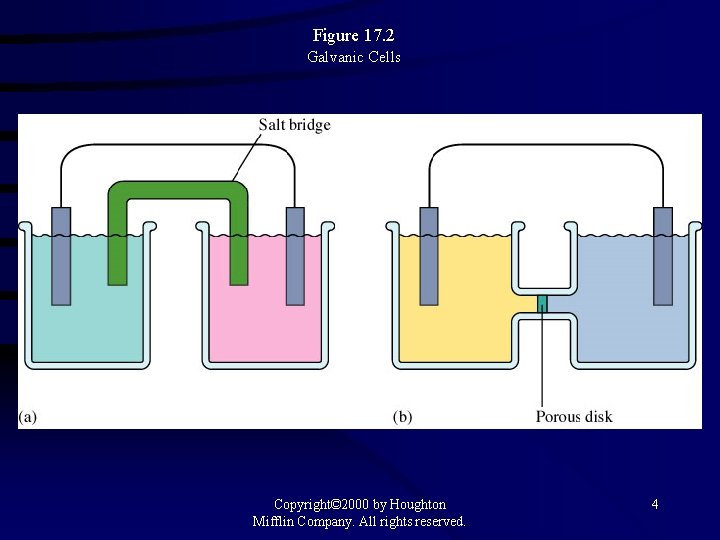
Figure 17. 2 Galvanic Cells Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 4
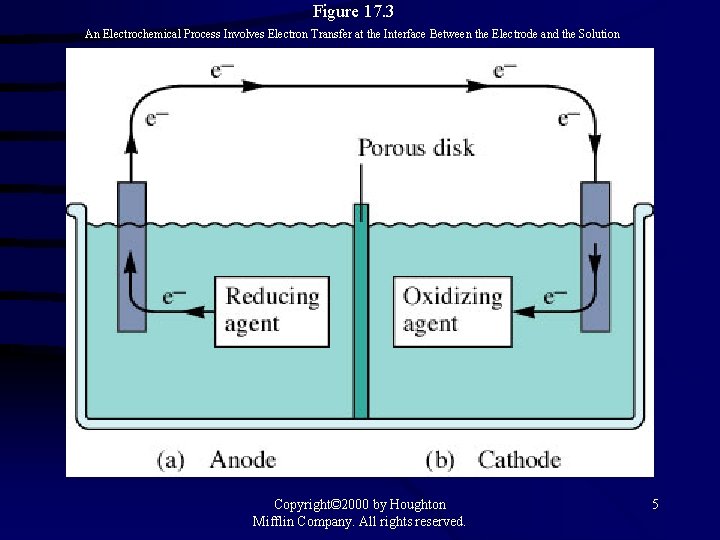
Figure 17. 3 An Electrochemical Process Involves Electron Transfer at the Interface Between the Electrode and the Solution Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 5
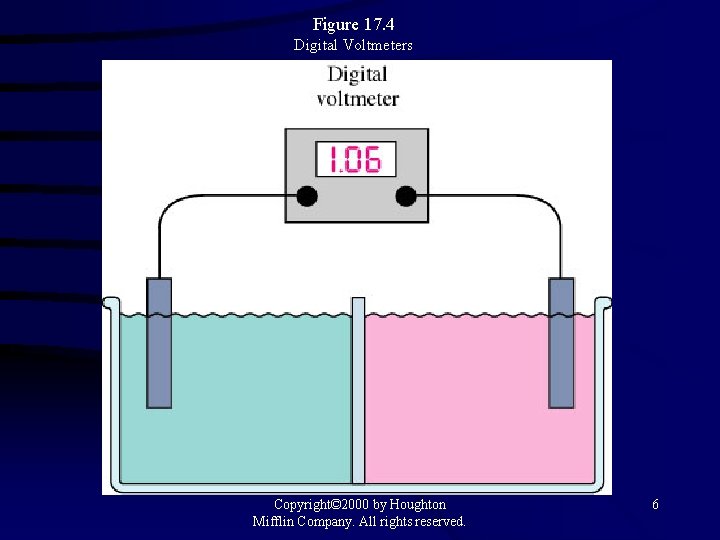
Figure 17. 4 Digital Voltmeters Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 6
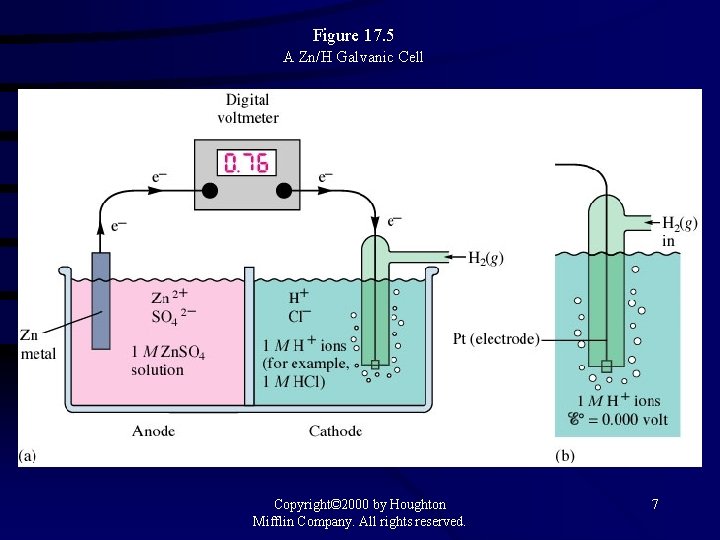
Figure 17. 5 A Zn/H Galvanic Cell Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 7
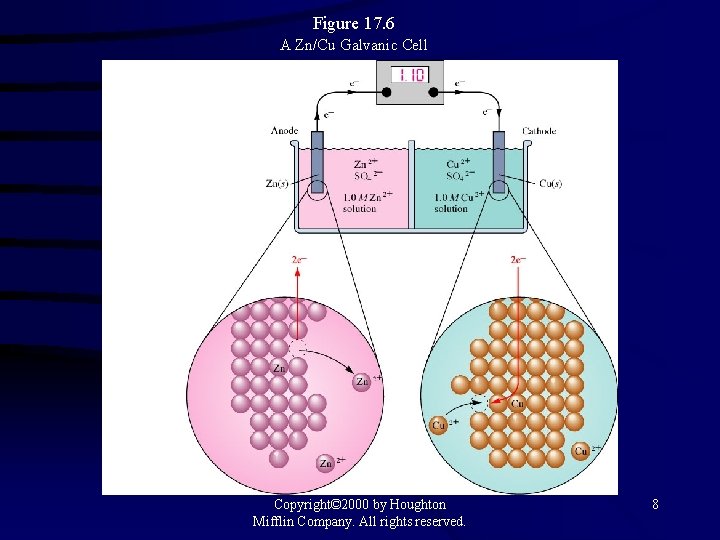
Figure 17. 6 A Zn/Cu Galvanic Cell Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 8
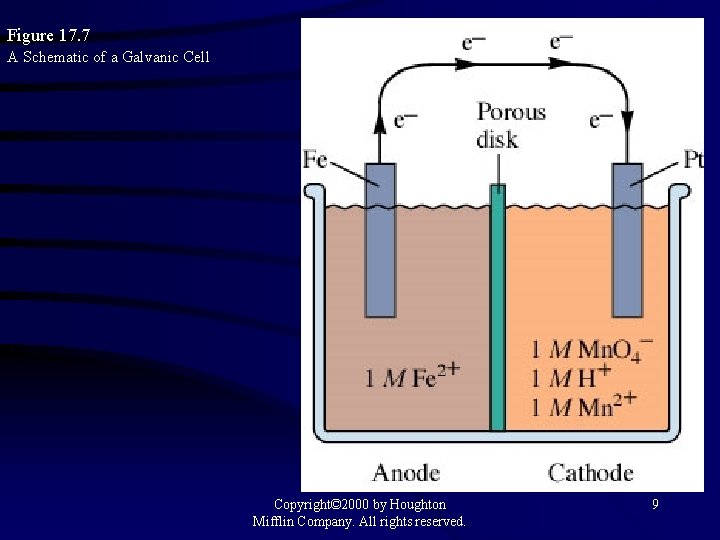
Figure 17. 7 A Schematic of a Galvanic Cell Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 9
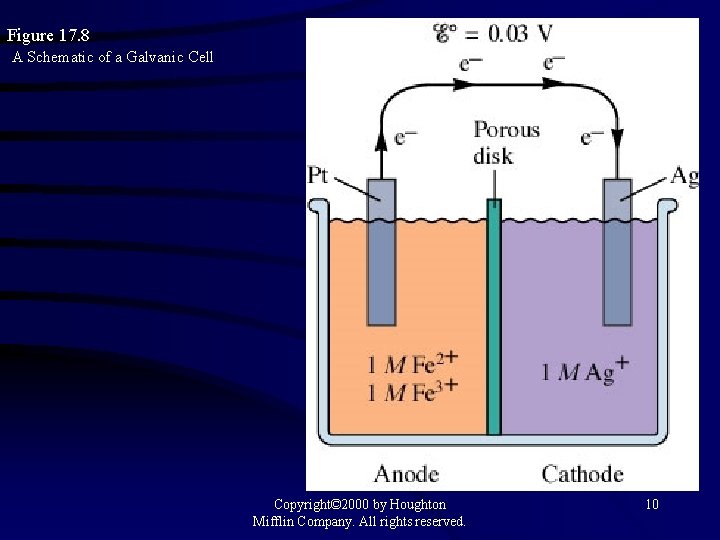
Figure 17. 8 A Schematic of a Galvanic Cell Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 10
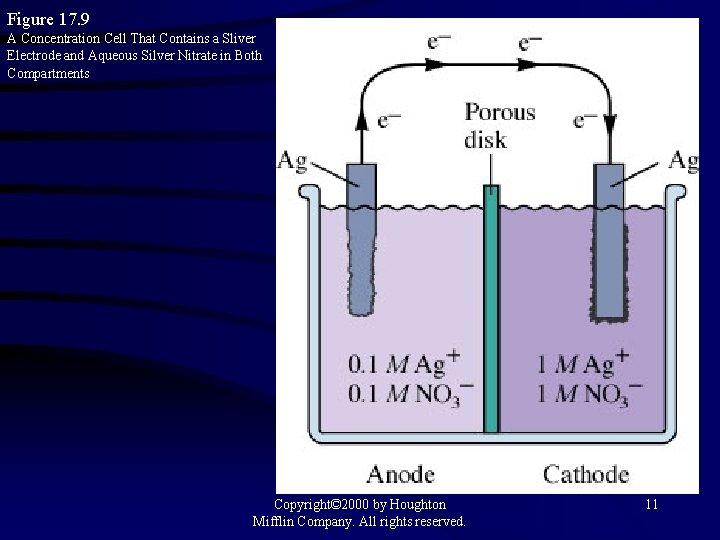
Figure 17. 9 A Concentration Cell That Contains a Sliver Electrode and Aqueous Silver Nitrate in Both Compartments Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 11
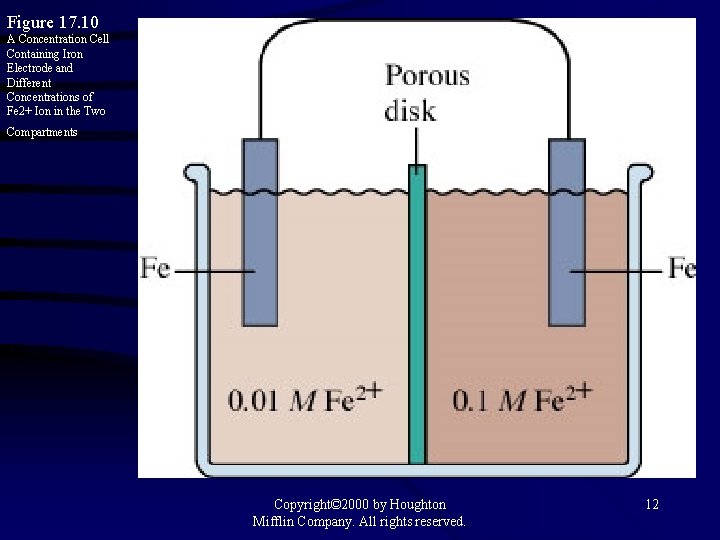
Figure 17. 10 A Concentration Cell Containing Iron Electrode and Different Concentrations of Fe 2+ Ion in the Two Compartments Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 12
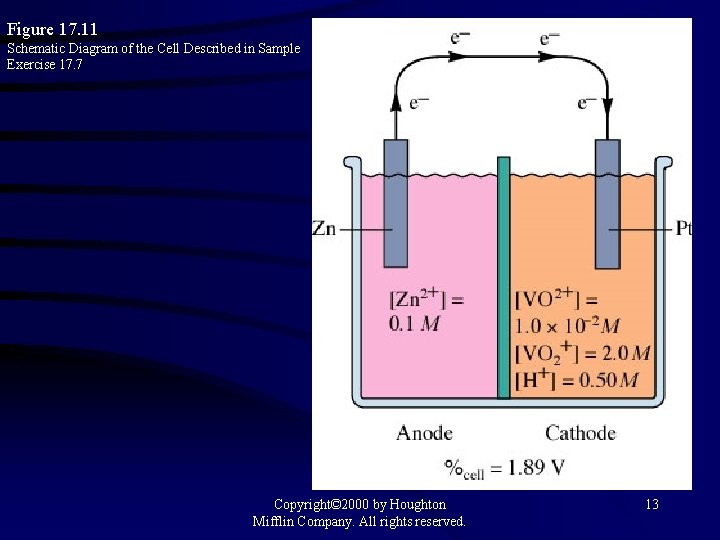
Figure 17. 11 Schematic Diagram of the Cell Described in Sample Exercise 17. 7 Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 13
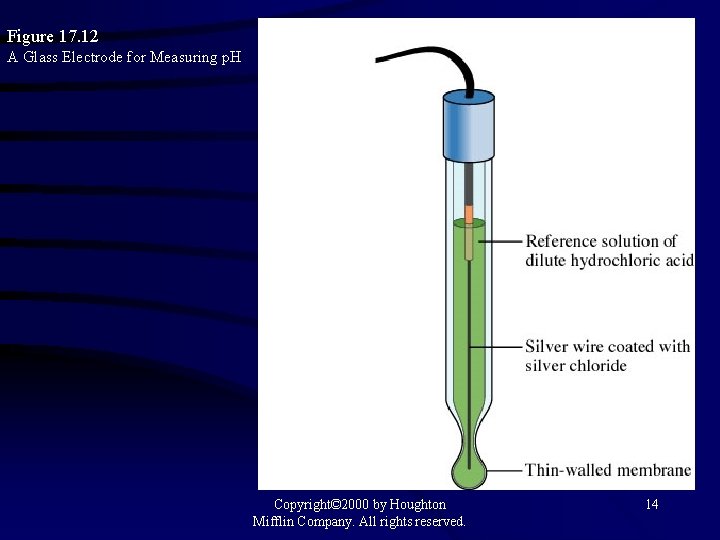
Figure 17. 12 A Glass Electrode for Measuring p. H Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 14
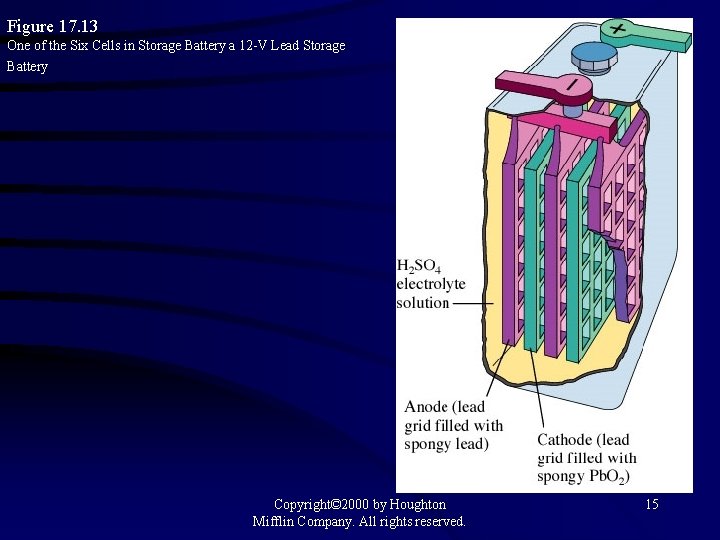
Figure 17. 13 One of the Six Cells in Storage Battery a 12 -V Lead Storage Battery Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 15
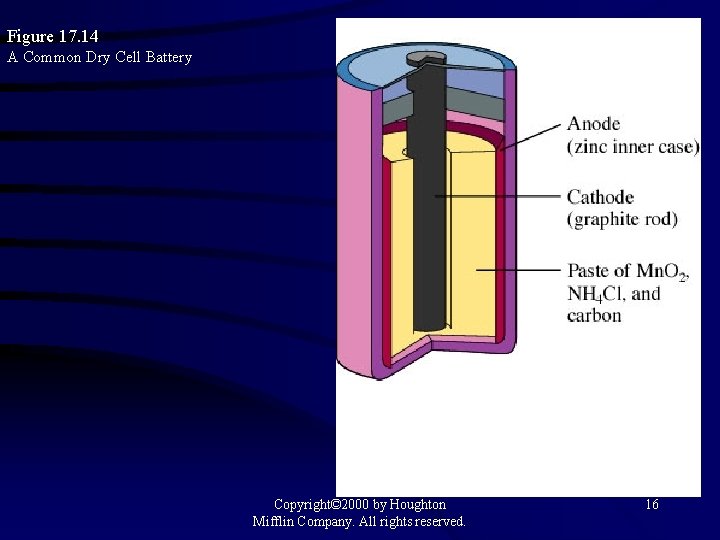
Figure 17. 14 A Common Dry Cell Battery Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 16
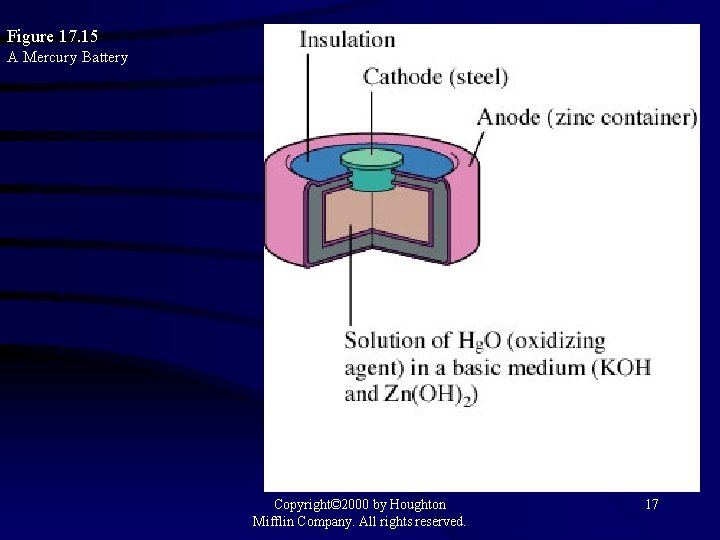
Figure 17. 15 A Mercury Battery Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 17
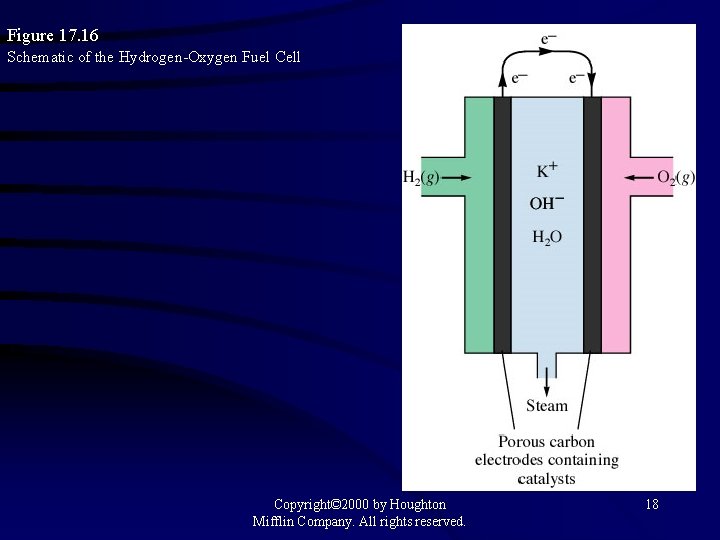
Figure 17. 16 Schematic of the Hydrogen-Oxygen Fuel Cell Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 18
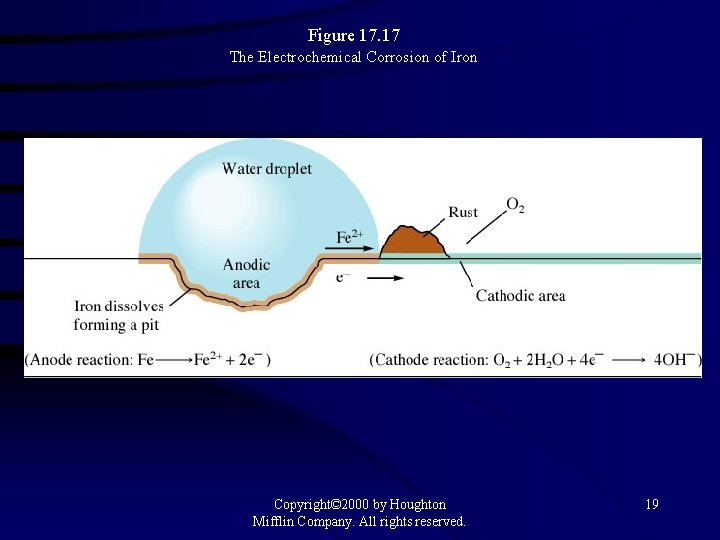
Figure 17. 17 The Electrochemical Corrosion of Iron Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 19
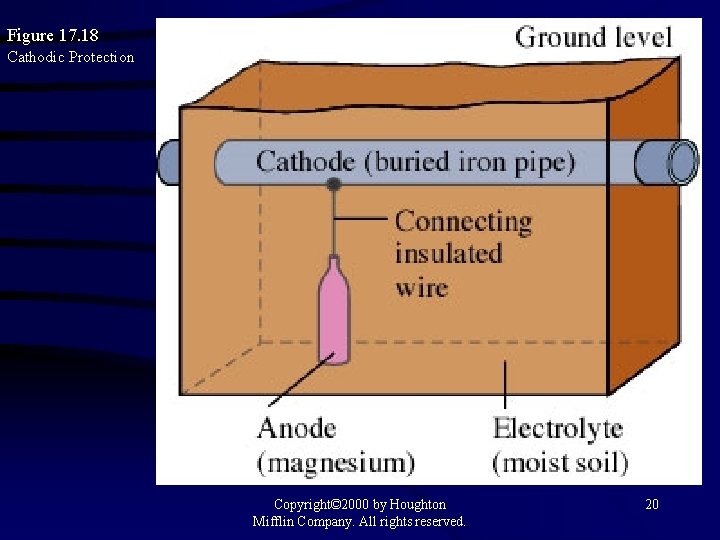
Figure 17. 18 Cathodic Protection Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 20
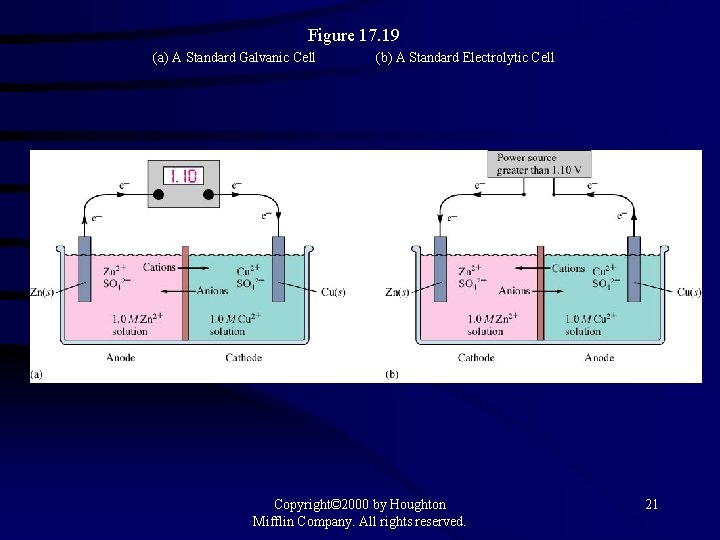
Figure 17. 19 (a) A Standard Galvanic Cell (b) A Standard Electrolytic Cell Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 21
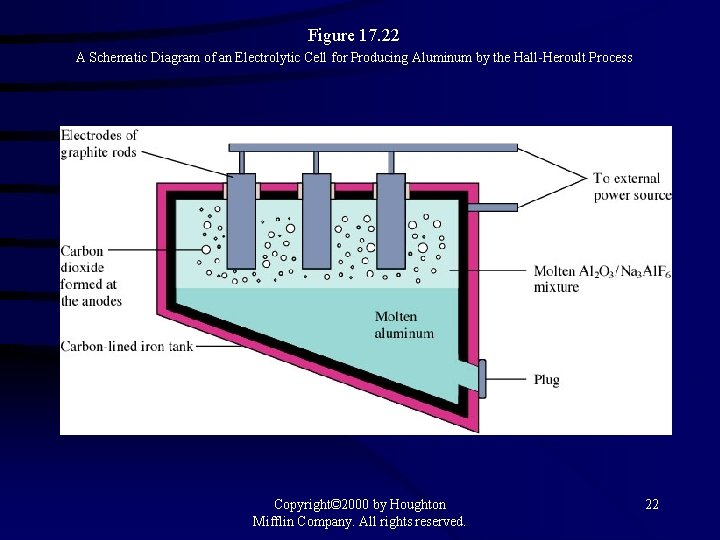
Figure 17. 22 A Schematic Diagram of an Electrolytic Cell for Producing Aluminum by the Hall-Heroult Process Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 22
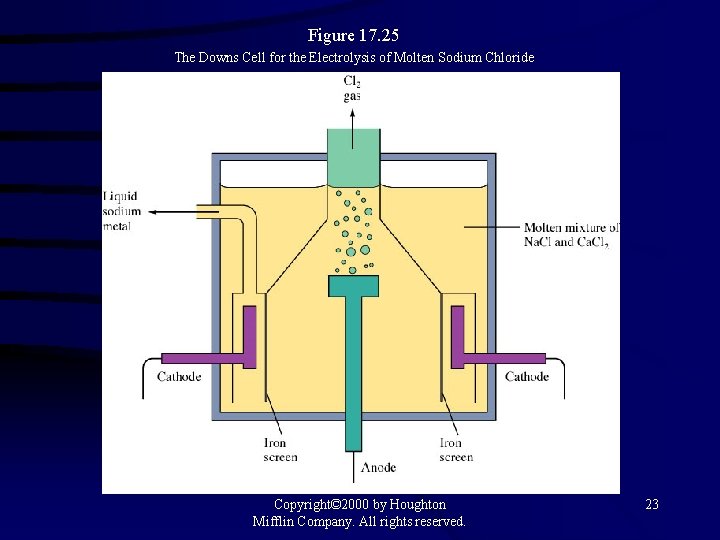
Figure 17. 25 The Downs Cell for the Electrolysis of Molten Sodium Chloride Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 23
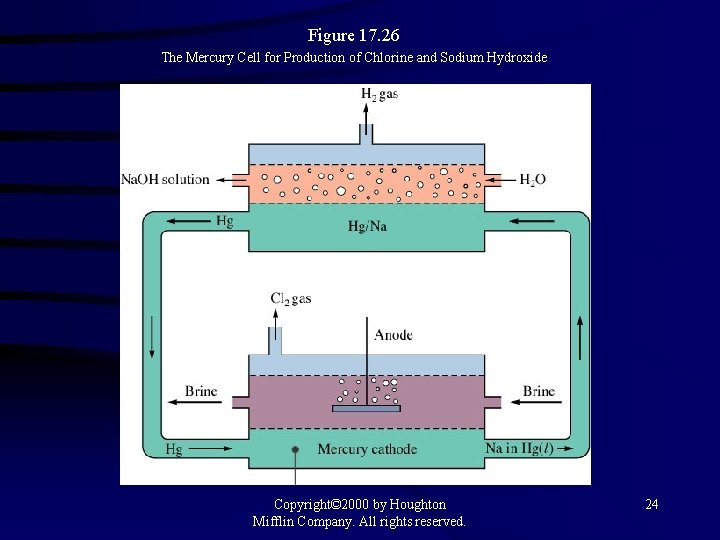
Figure 17. 26 The Mercury Cell for Production of Chlorine and Sodium Hydroxide Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 24
- Slides: 24