CHEMISTRY Do Now Obj IWBAT define and write

CHEMISTRY

Do Now Obj: IWBAT define and write an isotope and determine an element’s mass based on isotopic abundance. 1. What are three subatomic particles? What are their charges? 2. Which subatomic particles live in the nucleus of an atom? 3. How big are electrons (relative to a proton or a neutron)? 4. What number do you get when you multiply all of the numbers on a telephone's number pad? 5 minutes End

Agenda • Do Now • Notes • Activity • Notes • Practice Problems! • More Practice! • Exit Ticket

Notes • Important Equations: 1. Protons: # protons = Atomic # 2. Neutrons: # neutrons = Atomic Mass – Atomic # 3. Electrons: # electrons = # protons

Colors • Protons = Blue • Neutrons = Green • Electrons = Orange

Activity • Today we’ll be using M&Ms as substitutes for subatomic particles • Each color of M&M indicates a different subatomic particle • There are 10 different “atoms” in a bag labeled “A” through “J” • You’ll have roughly 3 minutes with each different “atom” (bag of M&Ms)

Notes • Isotopes – Atoms of an element with different numbers of neutrons • They are identified by their atomic mass • Hint: This is why the atomic mass has numbers after the decimal—it’s an average • Go back to your data from the activity. Put a star * next to all of the isotopes!

Isotope Mass – A Quick Note for the HW • Atomic mass is found using the masses of an atom’s isotopes: • % isotope 1 x mass isotope 1 + % isotope 2 x mass isotope 2 + … • Ex: Gallium has two naturally occurring isotopes. The mass of gallium-69 is 68. 9256 amu and it is 60. 108% abundant. The mass of gallium-71 is 70. 9247 amu and it is 39. 892% abundant. • Mass = 0. 60108 x 68. 9256 amu + 0. 39892 x 70. 9247 amu = 69. 723 amu

But…. How Do We Write Them? ! • There are two ways to write isotopes: 1. The element’s name followed by the atomic mass • Ex: Chromium-58 2. The atomic symbol with the mass number • Ex:
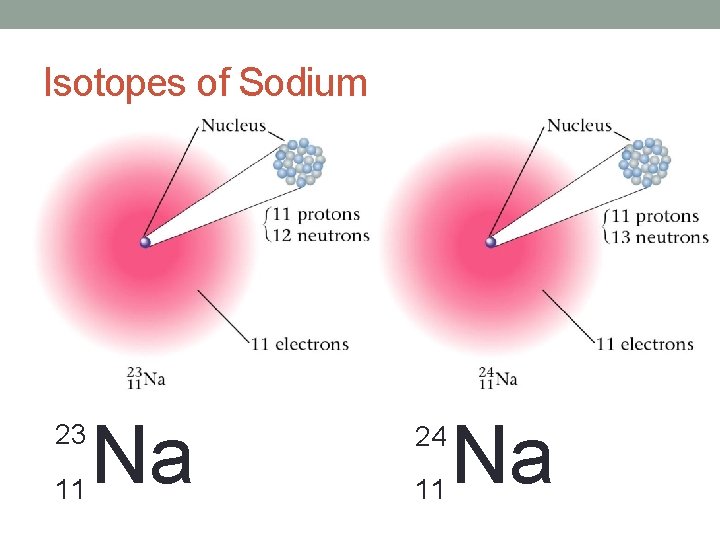
Isotopes of Sodium 23 11 Na 24 11 Na

Example Problem 1 • How many protons, neutrons, and electrons are in: 31 15 Protons: _____ 16 Neutrons: _____ 15 Electrons: _____

Example Problem • Write the isotope symbol for the chemical element that has 6 protons and 8 neutrons (write the symbol both ways) 14 6 C Carbon-14
Practice Problems! • Working quietly by yourself or with your neighbor, fill in the periodic table handout • You have 10 minutes • Raise your hand for Mr. Mannion’s help!

Exit Ticket • In your science journal, answer the following questions: 1. What is an isotope? 2. What are the two different ways you can write an isotope? 3. How do we distinguish between two different isotopes?
- Slides: 14