CHEMISTRY December 9 2014 PERIODIC TRENDS SCIENCE STARTER
CHEMISTRY December 9, 2014 PERIODIC TRENDS

SCIENCE STARTER • Do the Science Starter • You have 3 minutes to complete the task • You are silent and seated

QUESTION 1 • FLUORINE – PERIOD – GROUP NAME – ATOMIC NUMBER – VALENCE ELECTRONS – # OF ENERGY LEVELS

QUESTION 2 • WHAT IS THE GROUP NAME FOR GROUP 1?

QUESTION 3 • WHAT IS THE NAME OF THE ELEMENT LOCATED AT PERIOD 5 GROUP 17?

ATOMIC RADIUS - period • You may work with a partner to do the first 3 questions. • You may refer to Table S and Periodic Table to help you • You have 2 minutes to answer Questions 1 -3

PART 1 Q 1 -3 Symb Atomic ol Atomic Number Radius Li 3 130. pm Be 4 99 pm B 5 84 pm C 6 75 pm • Q 1: • Q 2: • Q 3:

ATOMIC RADIUS DEFINITION • Atomic Radius is an ESTIMATE of the size of the atom or the distance from the NUCLEUS to the edge of the atom.

PART 1 Q 5 • Going across a period, electrons are added to the outermost energy level. At the same time protons are being added to the nucleus. As a result, the increased concentration of protons in the nucleus creates a higher effective nuclear charge. Thus, a stronger force of attraction pulls the electrons closer to the nucleus resulting in a smaller atomic radius.

ATOMIC RADIUS - Group • You may work with a partner to do questions 6 -8 • You may refer to Table S and Periodic Table to help you • You have 2 minutes to answer Questions 6 -8
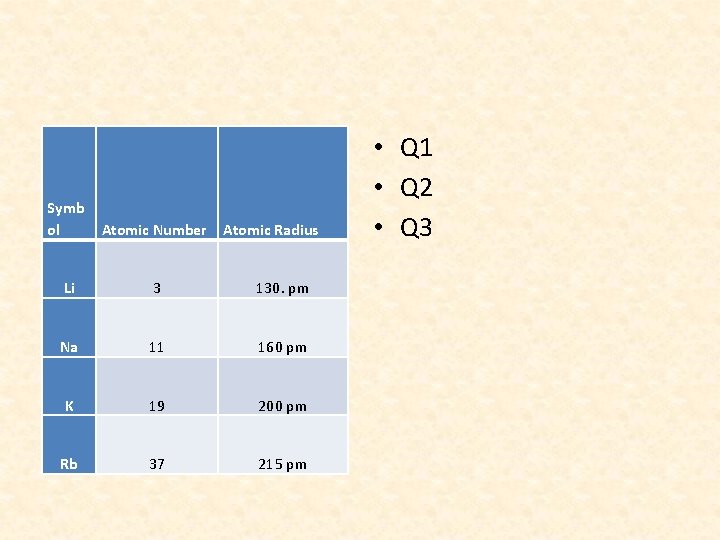
Symb ol Atomic Number Li 3 130. pm Na 11 160 pm K 19 200 pm Rb 37 215 pm Atomic Radius • Q 1 • Q 2 • Q 3

QUESTIONS 8 • Going down a group, the number of energy levels increases. Each subsequent energy level is further from the nucleus. Thus, the atomic radius increases as the group and energy levels increase.

FIRST IONIZATION ENERGY - Period • You may work with a partner to do questions 1 -2 • You may refer to Table S and Periodic Table to help you • You have 1 minute to answer Questions 1 -2
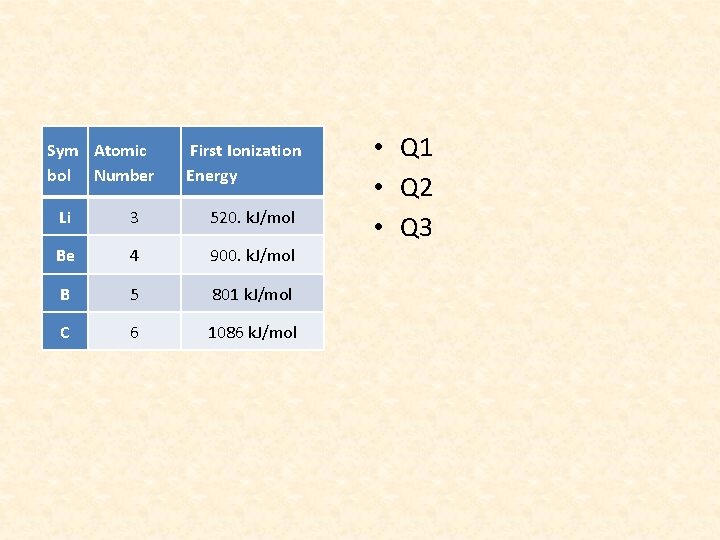
Sym Atomic bol Number First Ionization Energy Li 3 520. k. J/mol Be 4 900. k. J/mol B 5 801 k. J/mol C 6 1086 k. J/mol • Q 1 • Q 2 • Q 3

QUESTION 3 • First Ionization Energy is the energy required to remove the most loosely bound (outermost) electrons from an atom

QUESTION 4 • Going across a period, the atomic radius decreases making the atom smaller thereby making the outer electrons (or valence electrons) closer to the nucleus and more strongly attracted to the center. Therefore, it becomes more difficult to remove the outermost electron

FIRST IONIZATION ENERGY - GROUP • You may work with a partner to do questions 1 -3 • You may refer to Table S and Periodic Table to help you • You have 2 minutes to answer Questions 1 -3

Sym Atomic bol Number First Ionization Energy Li 3 520. k. J/mol Na 11 496 k. J/mol K 19 419 k. J/mol Rb 37 403 k. J/mol • Q 1 • Q 2 • Q 3

QUESTION 4 • Going down a group, the number of energy levels increases. Thus, the valence electrons are further away from the nucleus making it easier to remove the valence electrons.

ELECTRONEGATIVITY- period • You may work with a partner to do questions 5 -6 • You may refer to Table S and Periodic Table to help you • You have 2 minutes to answer Questions 5 -6
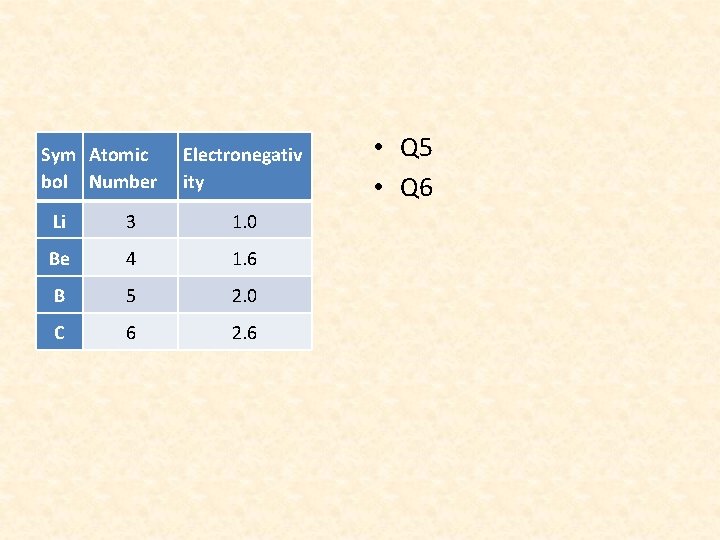
Sym Atomic bol Number Electronegativ ity Li 3 1. 0 Be 4 1. 6 B 5 2. 0 C 6 2. 6 • Q 5 • Q 6

QUESTION 7 • Electronegativity measures an atom’s tendency to attract and form bonds with ELECTRONS

QUESTION 8 • Going across a period, the number of valence electrons increases thus, making the electronegativity increasesas the atomic number increases.

ELECTRONEGATIVITY-groups • You may work with a partner to do questions 5 -6 • You may refer to Table S and Periodic Table to help you • You have 2 minutes to answer Questions 5 -6
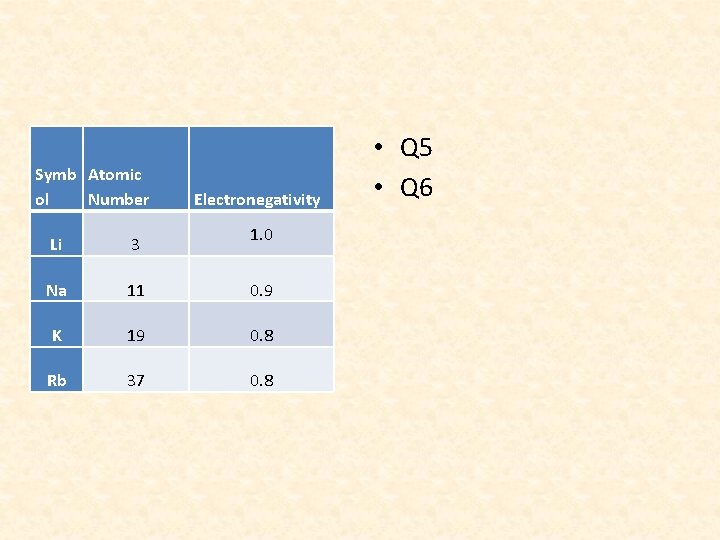
Symb Atomic ol Number Electronegativity 1. 0 Li 3 Na 11 0. 9 K 19 0. 8 Rb 37 0. 8 • Q 5 • Q 6

QUESTION 7 • Based on the table above, as the atomic number increases, the number of energy levels increases. Thus, the valence electrons are located FURTHER away from the nucleus. As a result, the ELECTRONEGATIVITY DECREASES

QUESTION 8 • Going down a group, the number of energy levels increases. Thus, the valence electrons are further away from the nucleus resulting in a decreased electronegativity.

METALLIC – Q 1 • Metallic characteristic of an element refers to how readily an atom can LOSE an electron.

METALLIC – Q 2 • Going across a period from right to left, the metallic characteristic INCREASES because the attraction between the valence electrons and the nucleus is WEAKER. • (MAKING IT EASIER TO LOSE THE ELECTRONS)
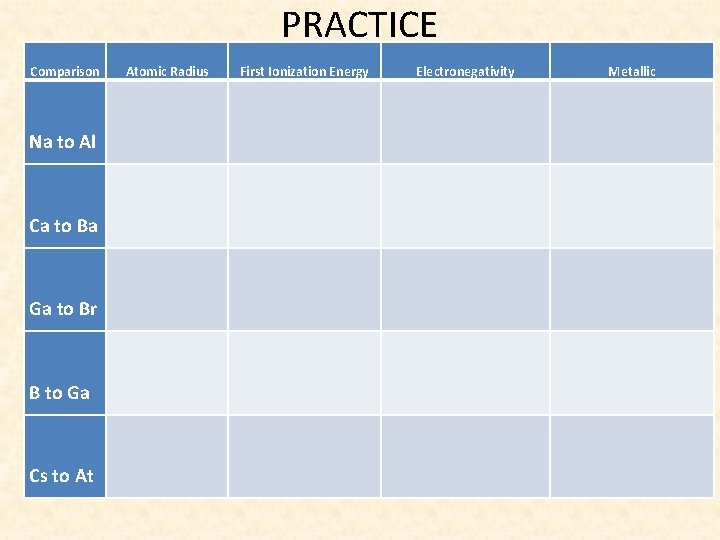
PRACTICE Comparison Atomic Radius First Ionization Energy Electronegativity Metallic Na to Al Ca to Ba Ga to Br B to Ga Cs to At

WRITTEN ASSIGNMENT • IN A CHEMICAL REACTION, WHICH ELEMENT (K OR Br) loses its valence electron first? – State a claim, provide evidence from the periodic table and/or Table S and explain your evidence.
- Slides: 32