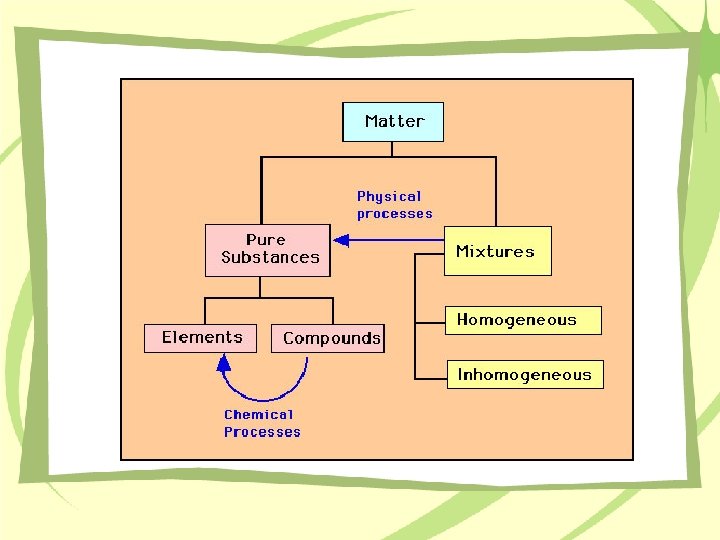
Chemistry Chemistry Overview Inorganic Organic Biochemistry Analytical Matter
Chemistry
Chemistry Overview • • Inorganic Organic Biochemistry Analytical
Matter • Define • Composition – Fundamental Elements – Atomic Particles
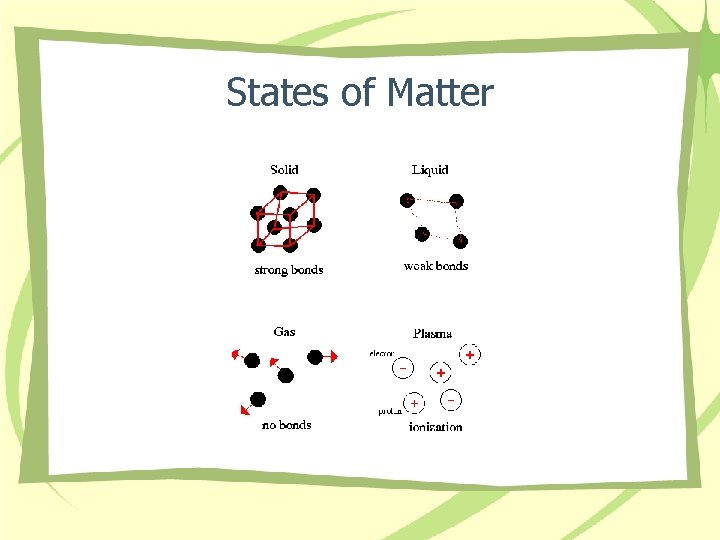
States of Matter
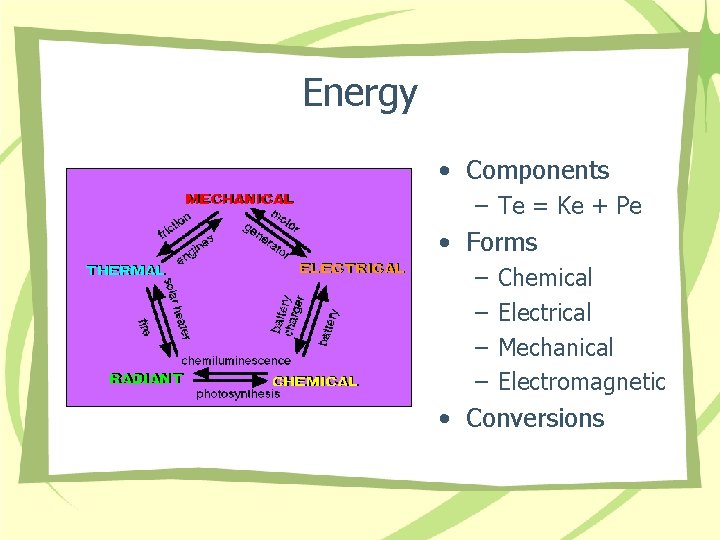
Energy • Components – Te = Ke + Pe • Forms – – Chemical Electrical Mechanical Electromagnetic • Conversions
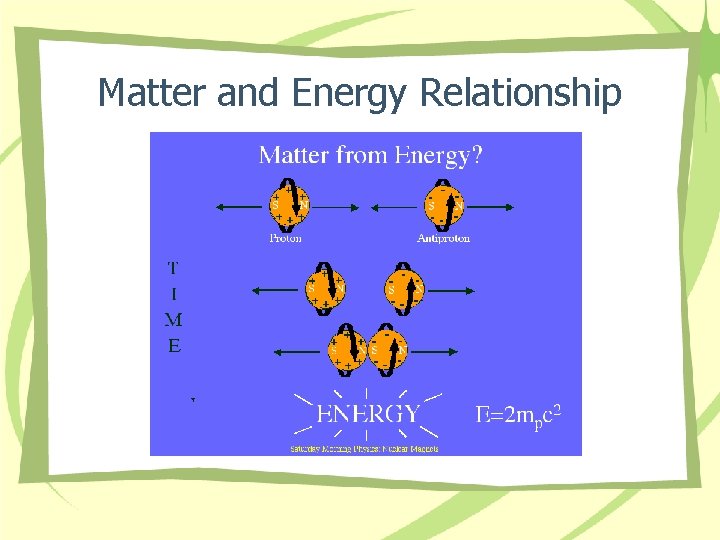
Matter and Energy Relationship
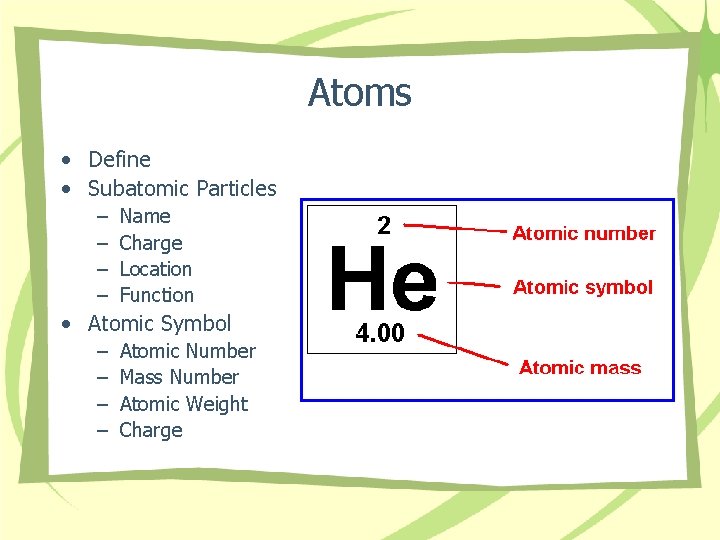
Atoms • Define • Subatomic Particles – – Name Charge Location Function • Atomic Symbol – – Atomic Number Mass Number Atomic Weight Charge
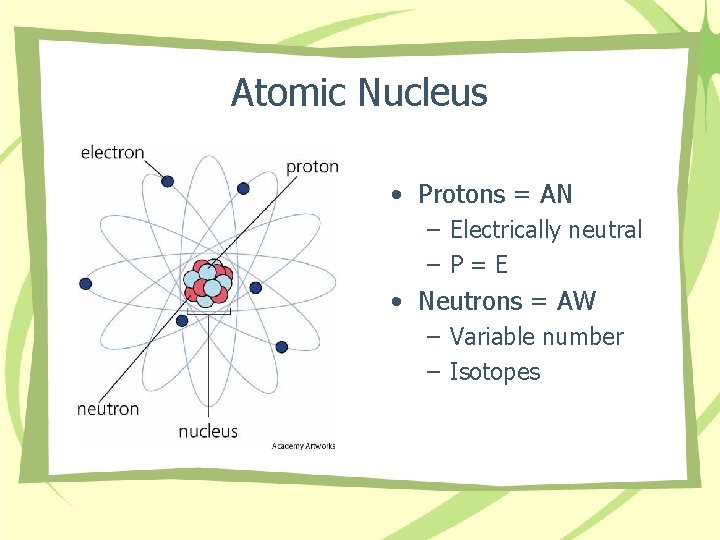
Atomic Nucleus • Protons = AN – Electrically neutral –P=E • Neutrons = AW – Variable number – Isotopes
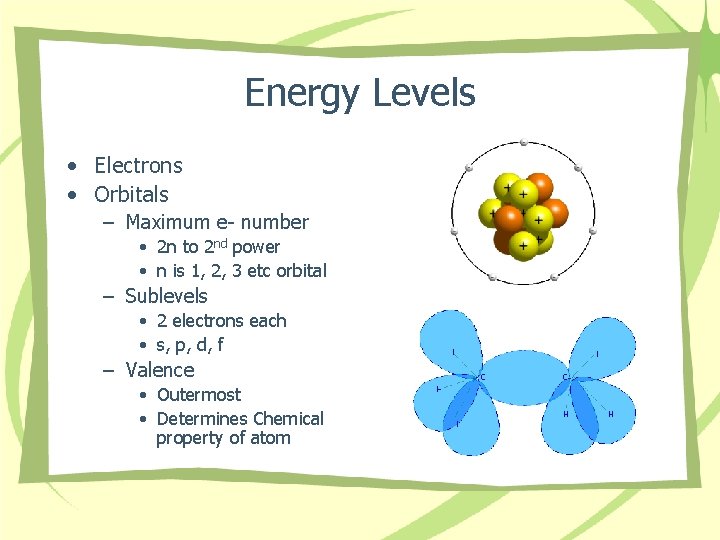
Energy Levels • Electrons • Orbitals – Maximum e- number • 2 n to 2 nd power • n is 1, 2, 3 etc orbital – Sublevels • 2 electrons each • s, p, d, f – Valence • Outermost • Determines Chemical property of atom
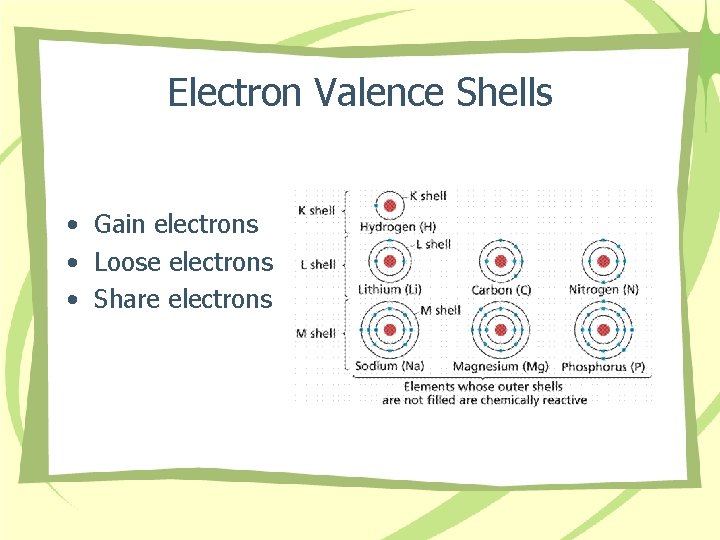
Electron Valence Shells • Gain electrons • Loose electrons • Share electrons
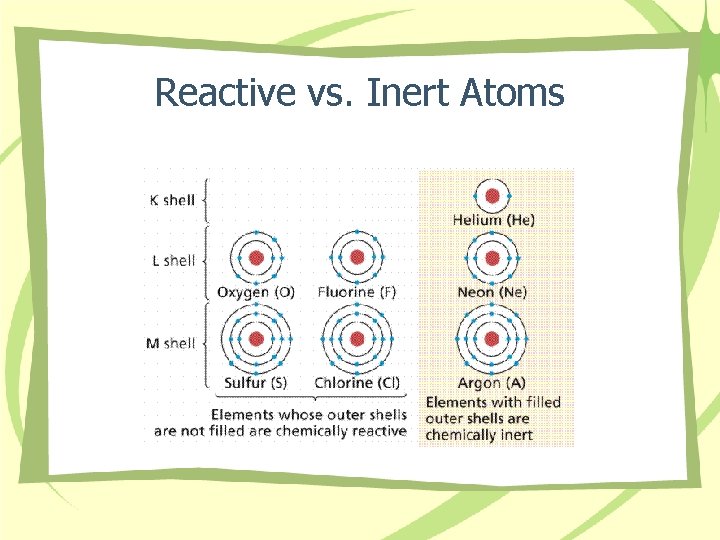
Reactive vs. Inert Atoms
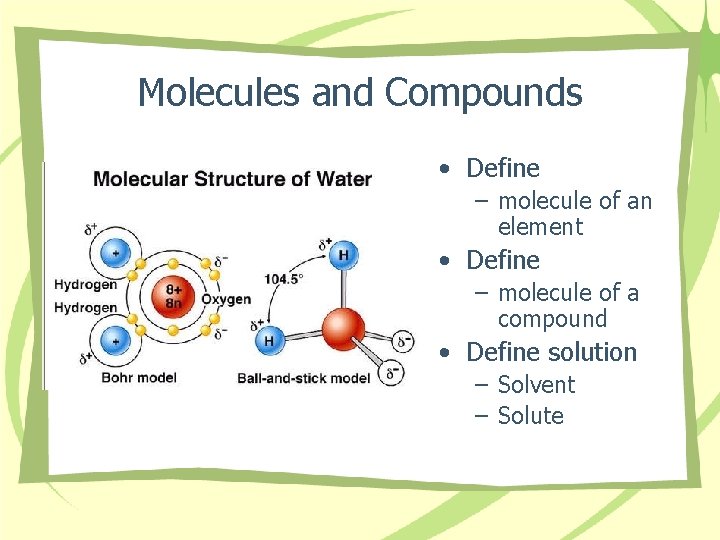
Molecules and Compounds • Define – molecule of an element • Define – molecule of a compound • Define solution – Solvent – Solute
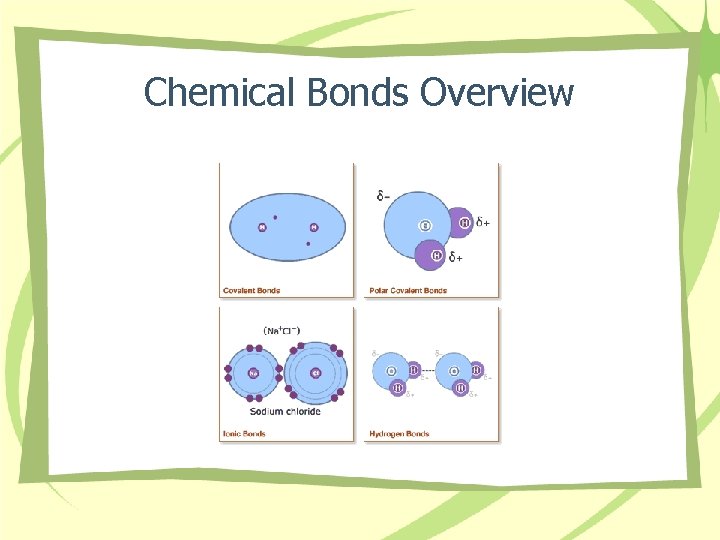
Chemical Bonds Overview
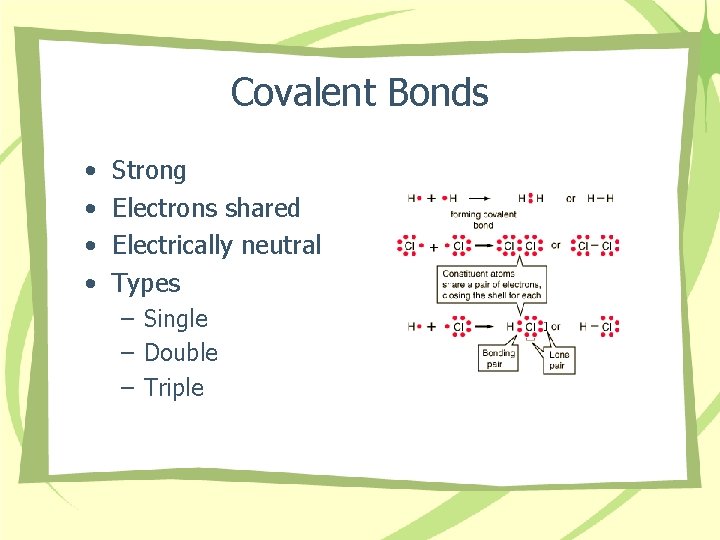
Covalent Bonds • • Strong Electrons shared Electrically neutral Types – Single – Double – Triple
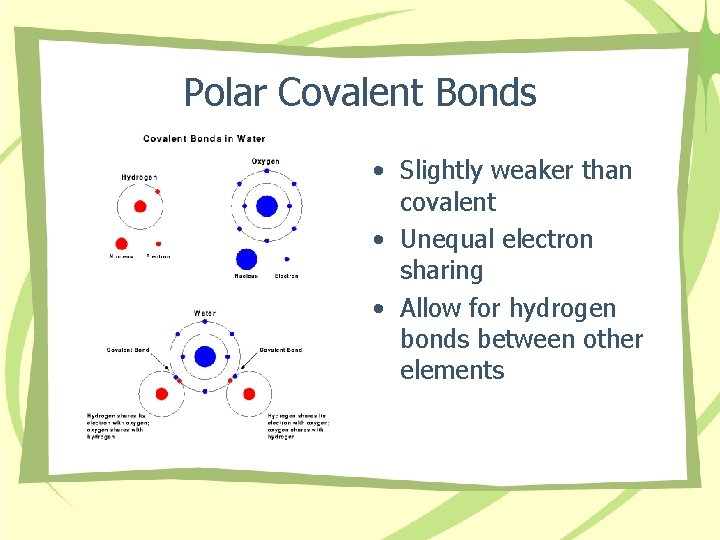
Polar Covalent Bonds • Slightly weaker than covalent • Unequal electron sharing • Allow for hydrogen bonds between other elements
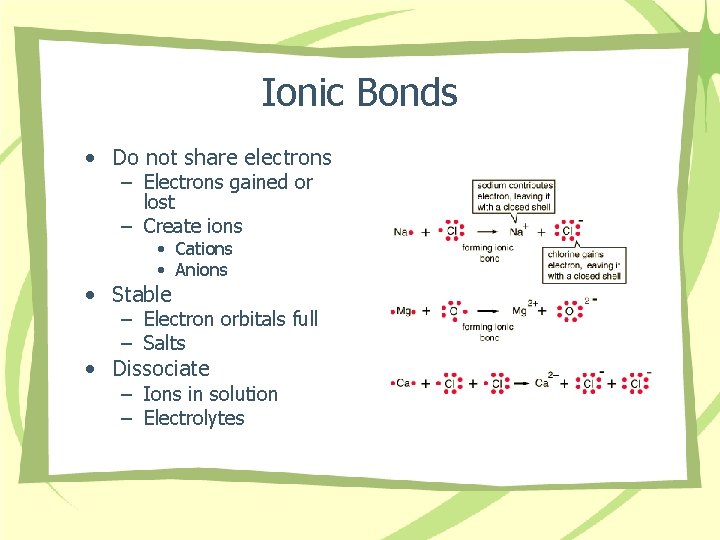
Ionic Bonds • Do not share electrons – Electrons gained or lost – Create ions • Cations • Anions • Stable – Electron orbitals full – Salts • Dissociate – Ions in solution – Electrolytes
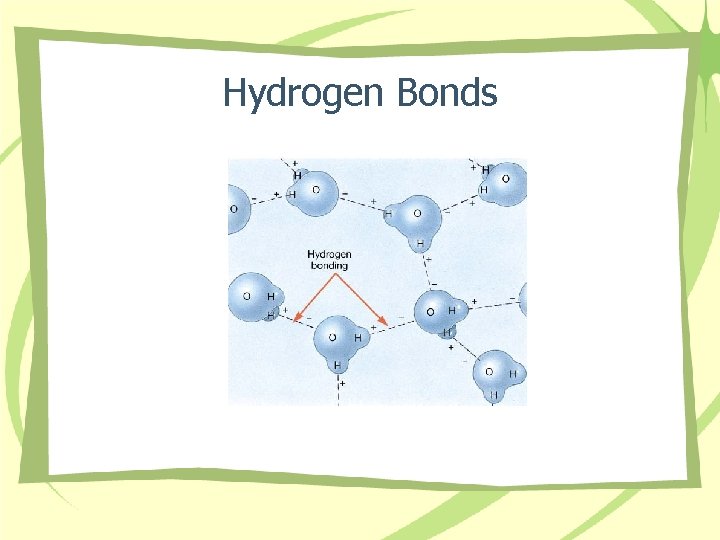
Hydrogen Bonds

Chemical Reactions • Reaction components – Reactants – Products – Direction indicated by arrows • Types – – Decomposition Synthesis Displacement Reversible • Reaction Rates – – – Temperature Particle size Concentration of reactants Enzymes Energy requirements
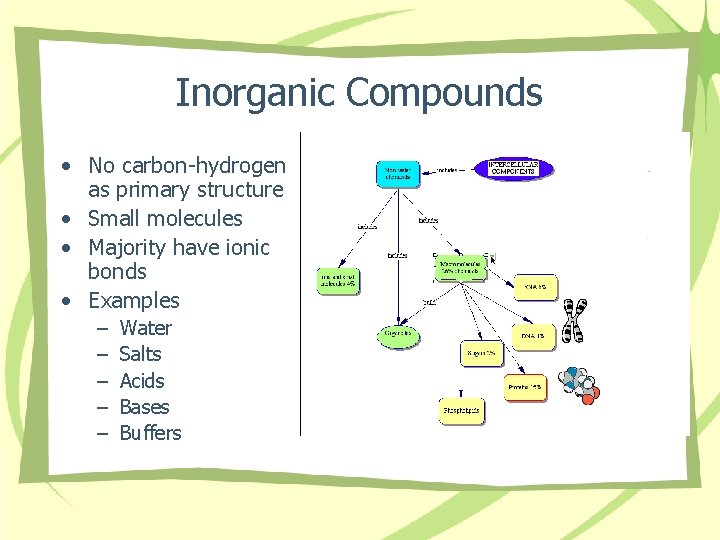
Inorganic Compounds • No carbon-hydrogen as primary structure • Small molecules • Majority have ionic bonds • Examples – – – Water Salts Acids Bases Buffers

Water • 2/3 body weight • 75% of cell volume • Bonds – Polar covalent – Hydrogen • Chemistry – Dehydration synthesis – Hydrolysis • Properties – – – Temperature Solvent Reactant p. H Surface tension Solubility

p. H • • • Define Math equation Scale Normal range Problems – – Acidemia, acidosis Alkalemia, alkalosis Compensated Uncompensated
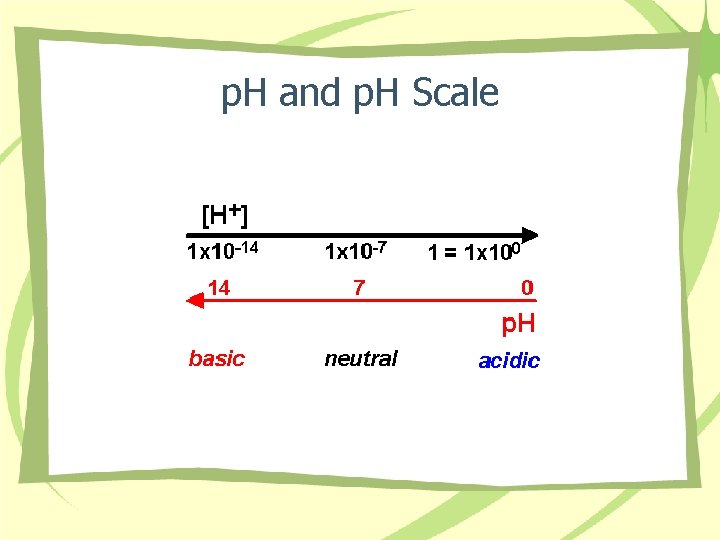
p. H and p. H Scale
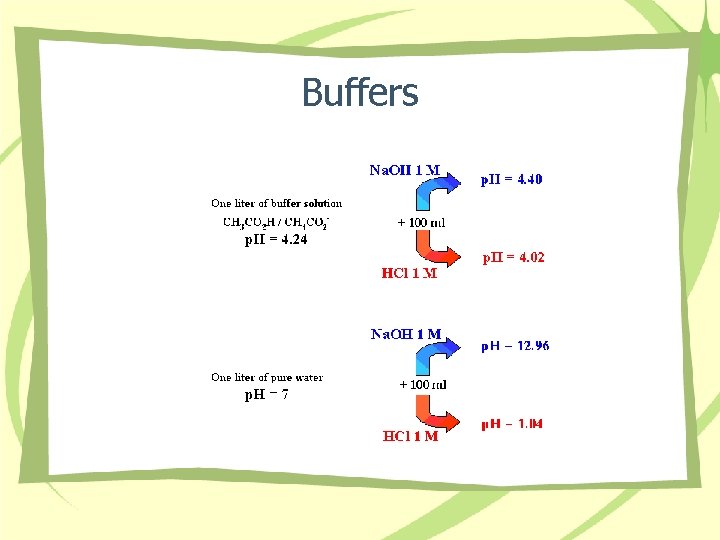
Buffers
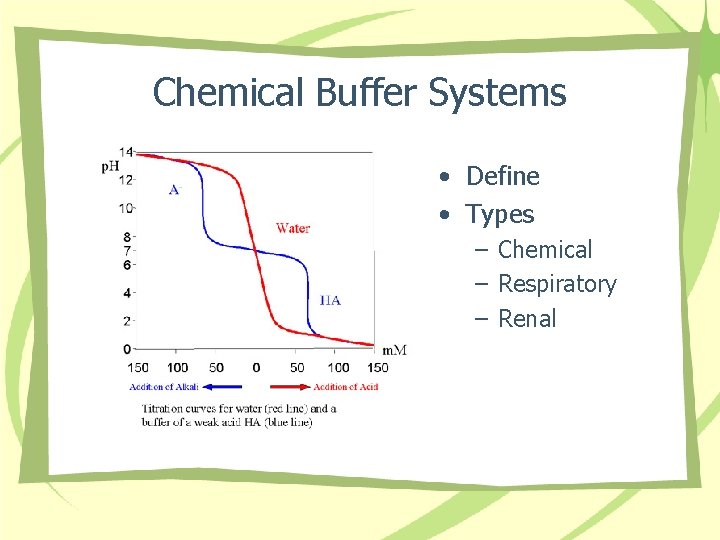
Chemical Buffer Systems • Define • Types – Chemical – Respiratory – Renal
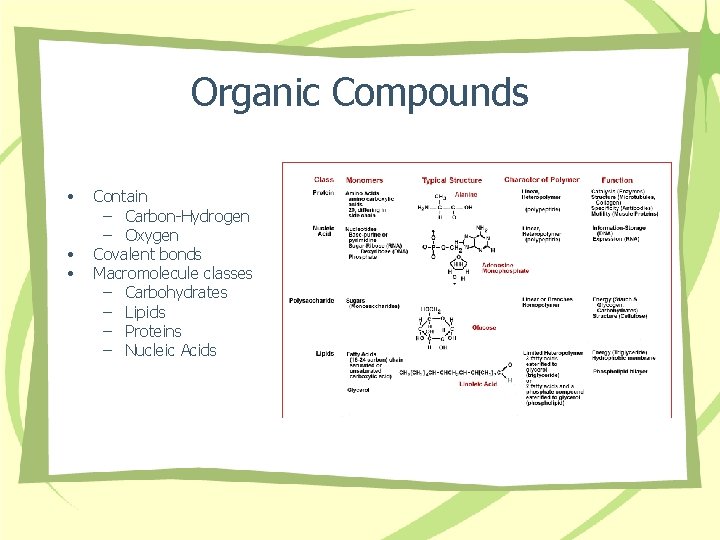
Organic Compounds • • • Contain – Carbon-Hydrogen – Oxygen Covalent bonds Macromolecule classes – Carbohydrates – Lipids – Proteins – Nucleic Acids
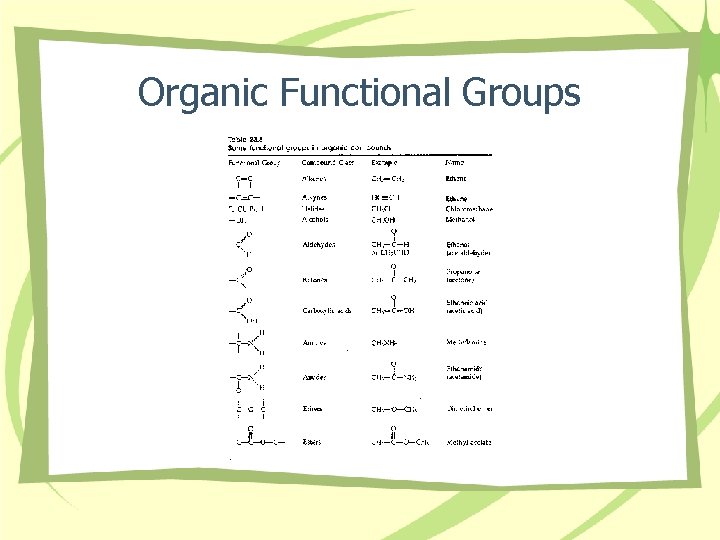
Organic Functional Groups
Carbohydrates • Source • Function • Structure – Formula – Combinations • Mono • Di • Poly-
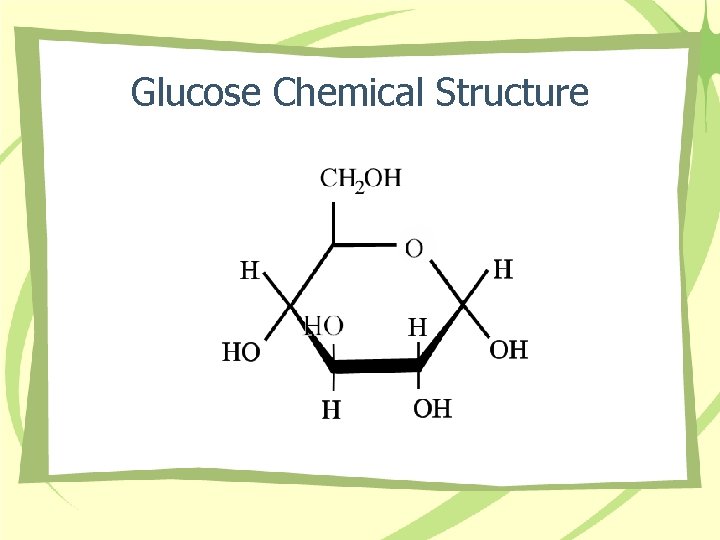
Glucose Chemical Structure
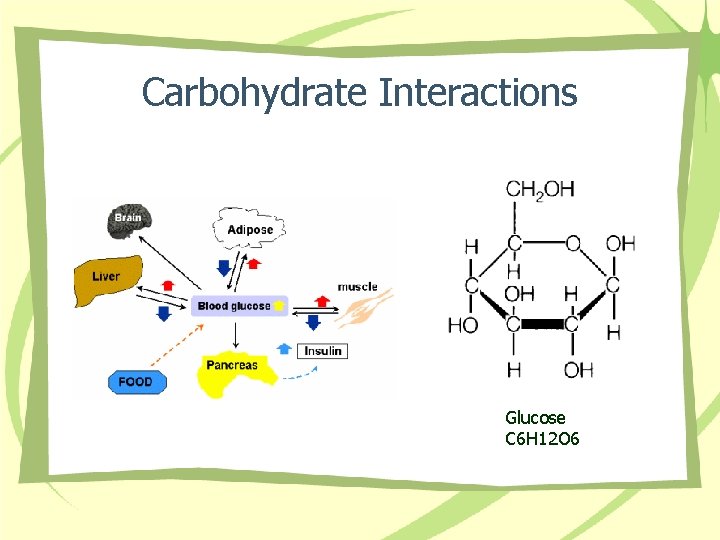
Carbohydrate Interactions Glucose C 6 H 12 O 6
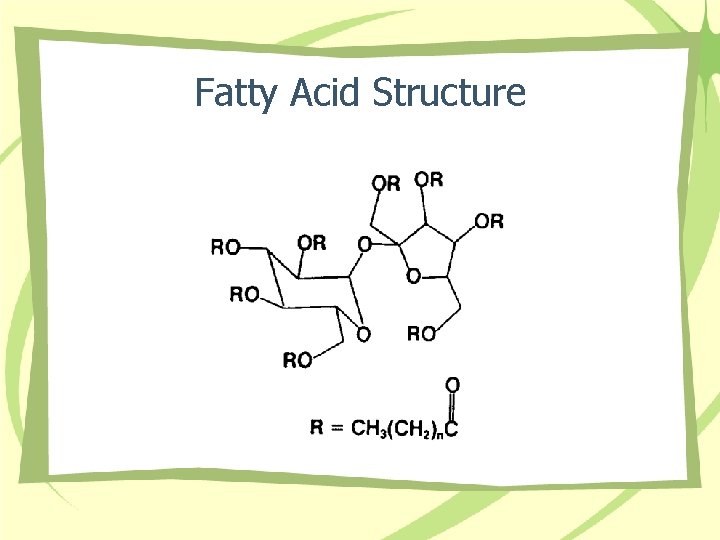
Fatty Acid Structure
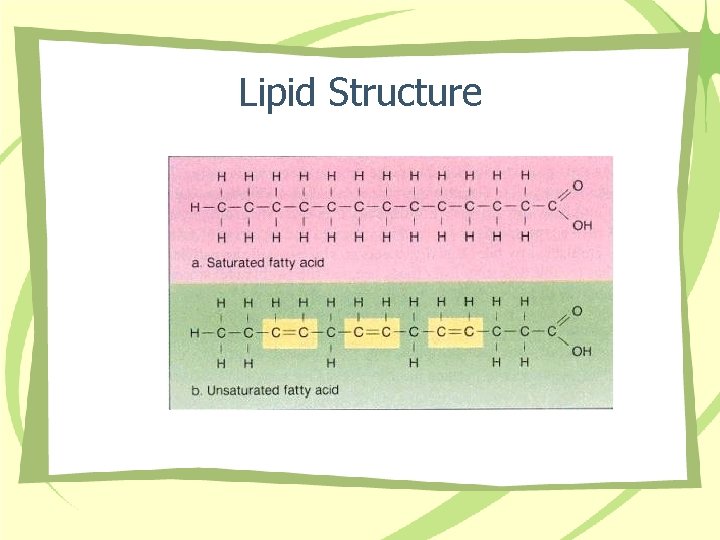
Lipid Structure
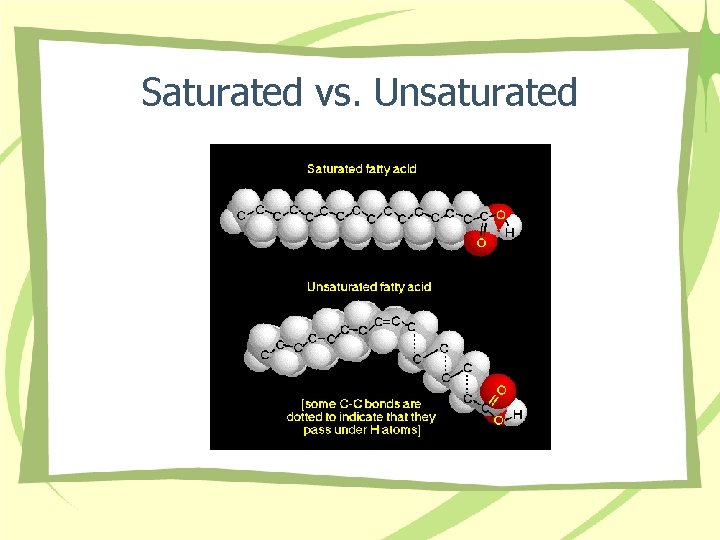
Saturated vs. Unsaturated
Lipids • Basic Structure • Class Types – – – Fatty Acids Glycerides Prostaglandins Steroids Phospholipids Glycolipids • Function
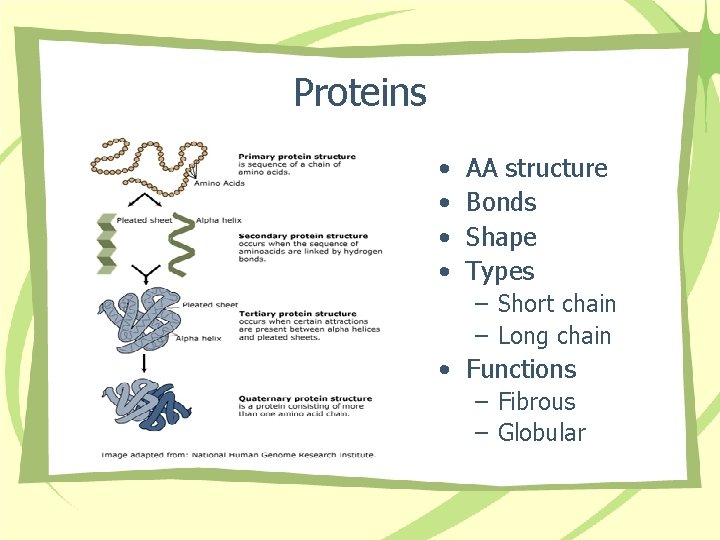
Proteins • • AA structure Bonds Shape Types – Short chain – Long chain • Functions – Fibrous – Globular
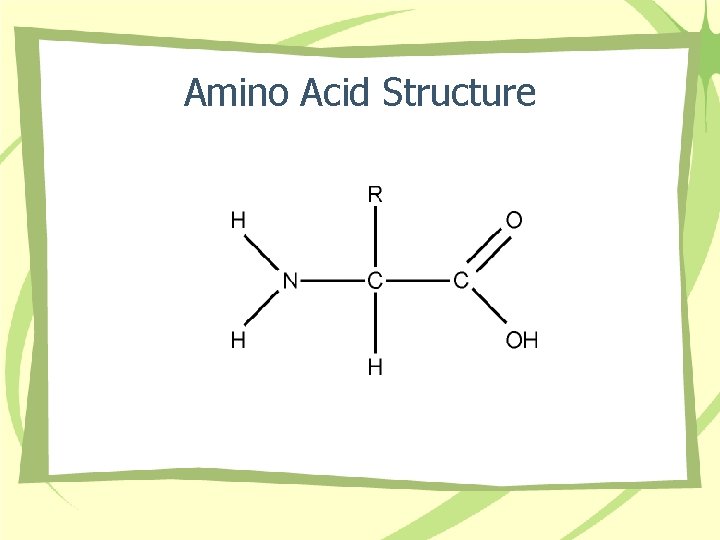
Amino Acid Structure
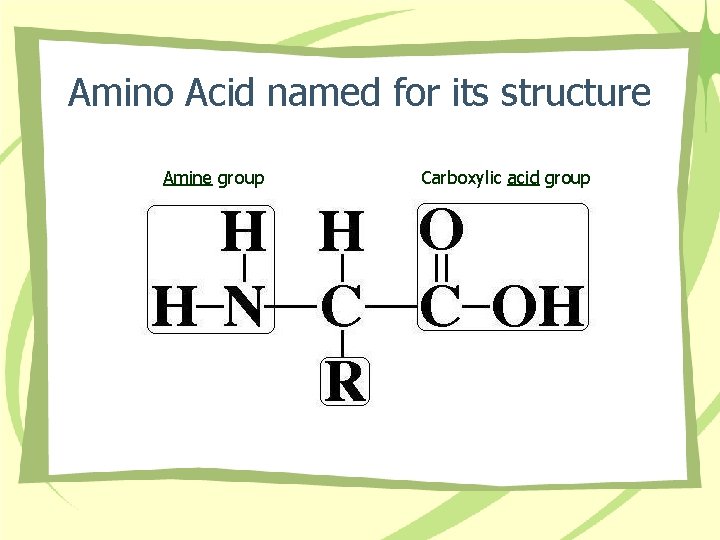
Amino Acid named for its structure Amine group Carboxylic acid group
Amino Acid Groups
Peptide Bonds

Fibrous Proteins • • Structure Support Movement/Contraction External Defense Elastin and collagen
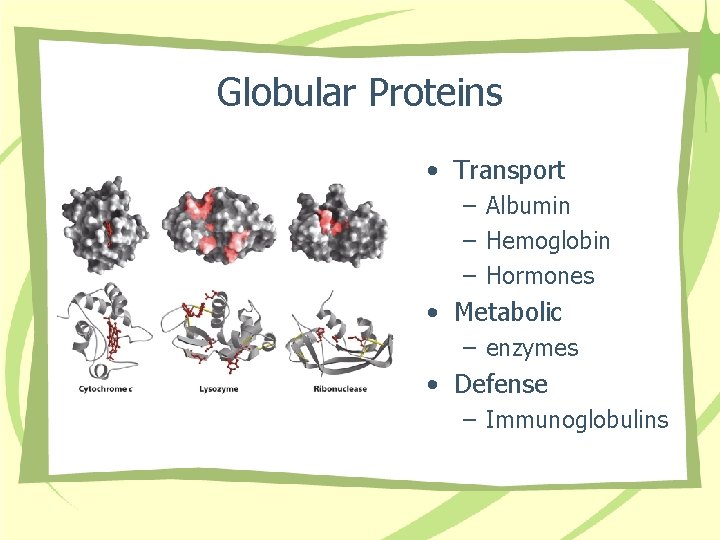
Globular Proteins • Transport – Albumin – Hemoglobin – Hormones • Metabolic – enzymes • Defense – Immunoglobulins
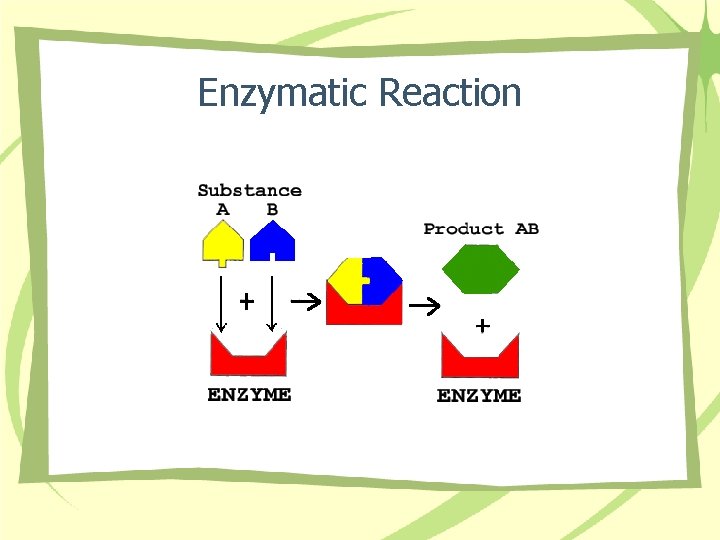
Enzymatic Reaction
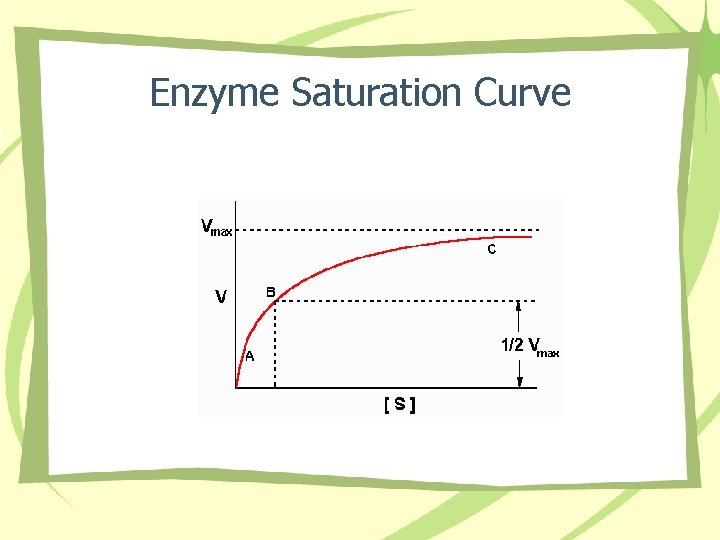
Enzyme Saturation Curve
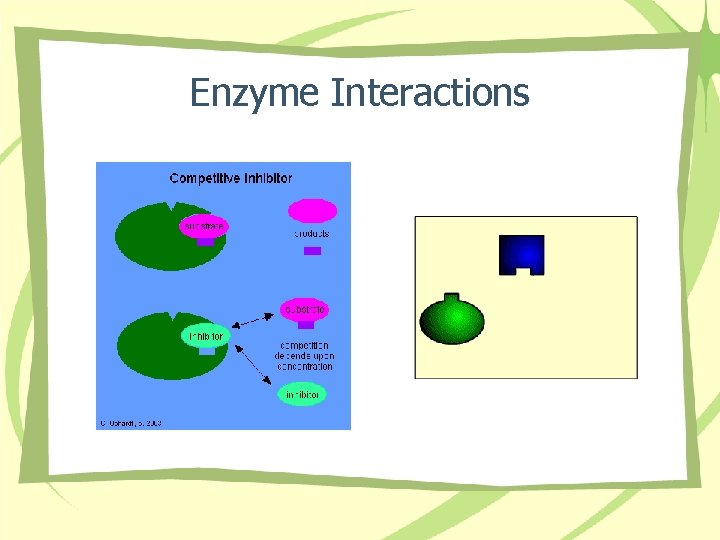
Enzyme Interactions
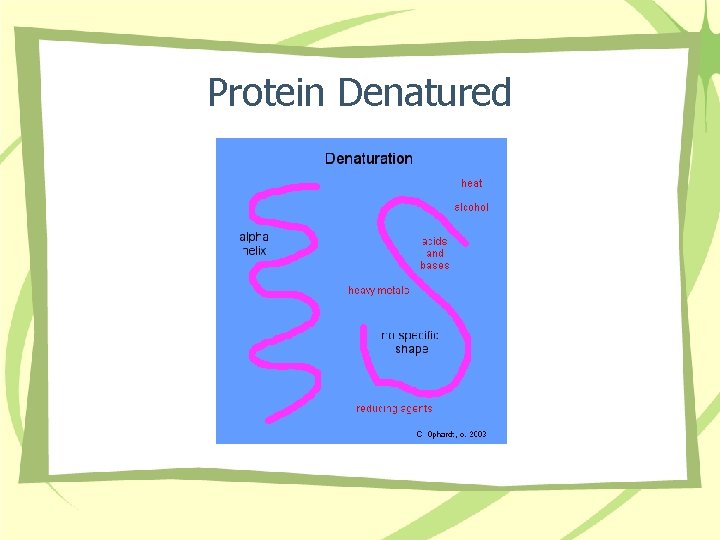
Protein Denatured
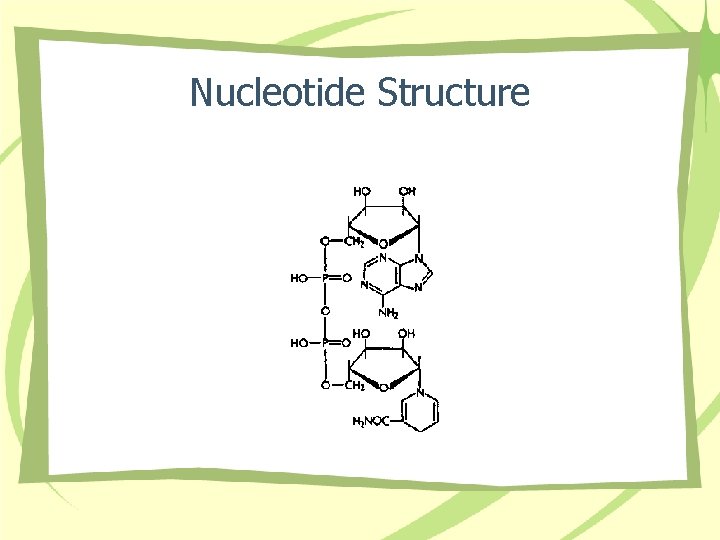
Nucleotide Structure
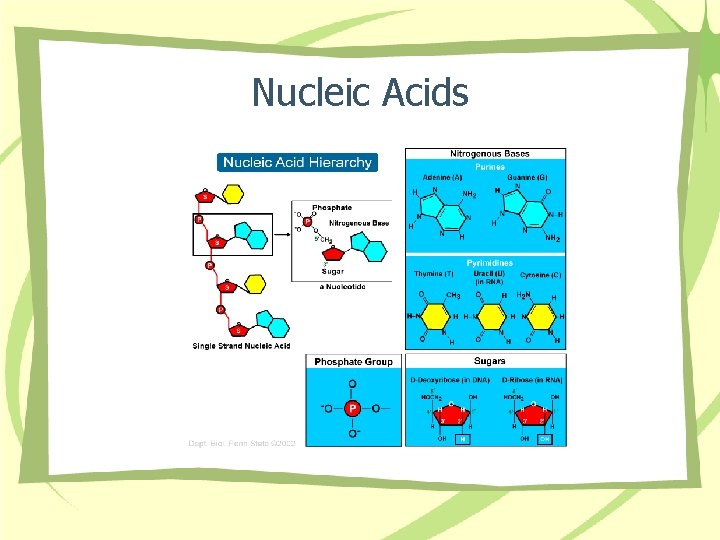
Nucleic Acids
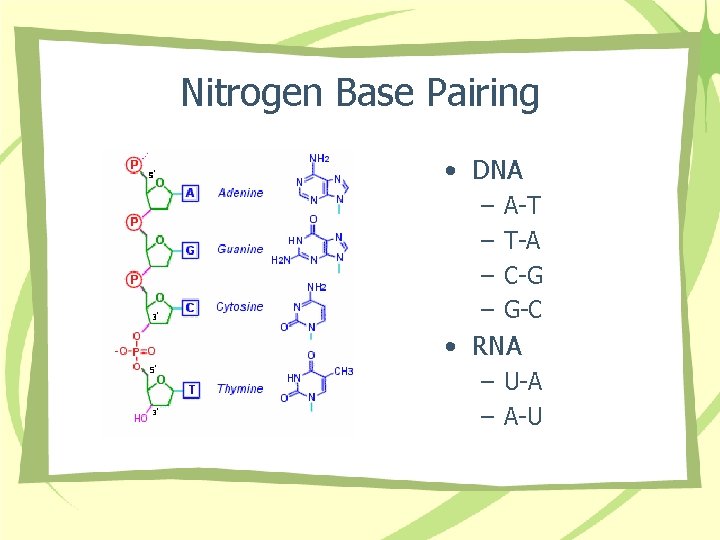
Nitrogen Base Pairing • DNA – – A-T T-A C-G G-C • RNA – U-A – A-U

Nucleic Acid Comparison • DNA • RNA – – – – – Double stranded Alpha Helix AT and CG base pairs Nucleus (eukaryotes) Semi-conservative replication creates daughter strands during S phase of cell cycle Single stranded Different for each type AU and CG base pairs Cytoplasm Created from DNA for transcription and translation for protein synthesis

DNA
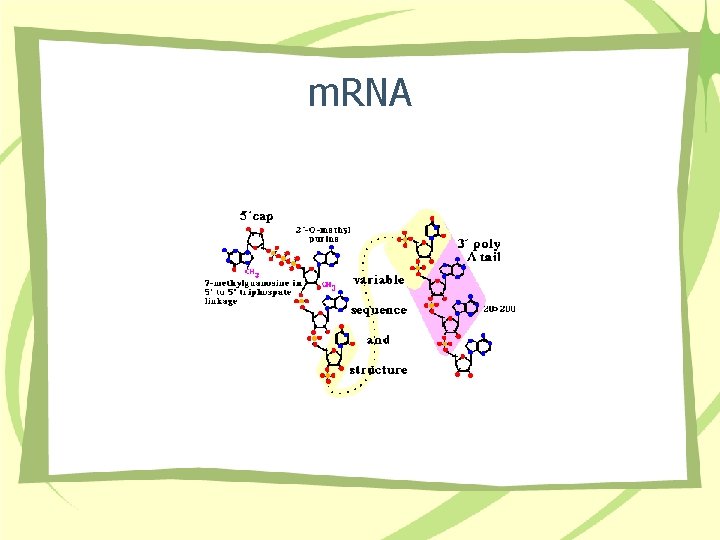
m. RNA
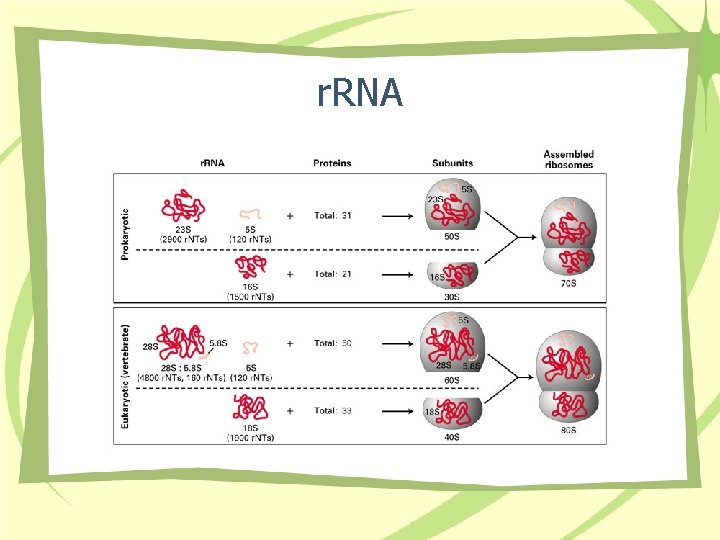
r. RNA
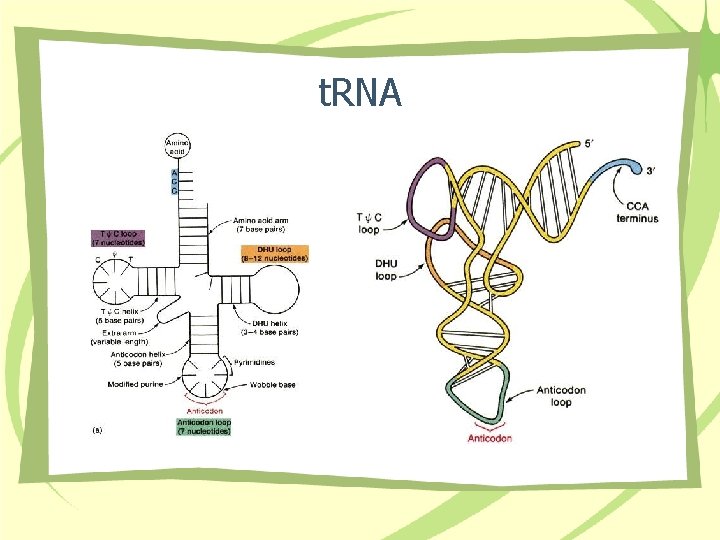
t. RNA
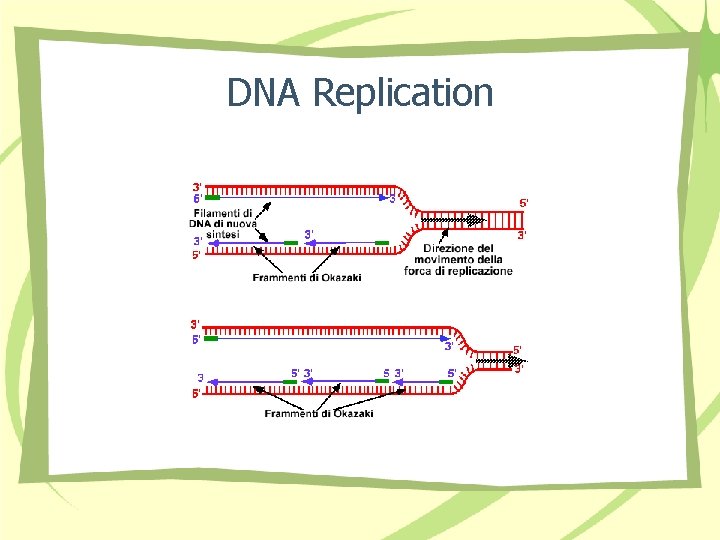
DNA Replication
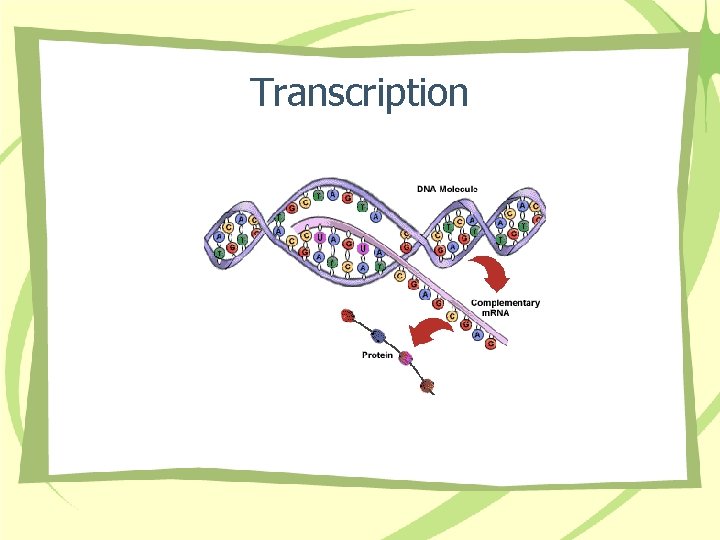
Transcription
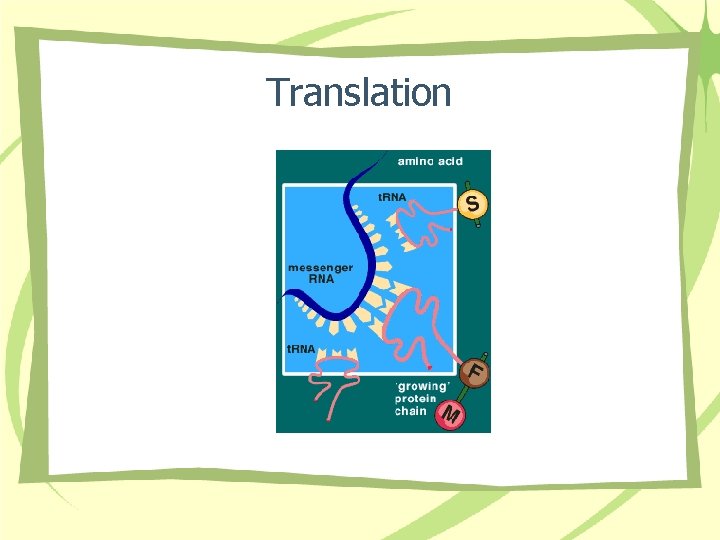
Translation
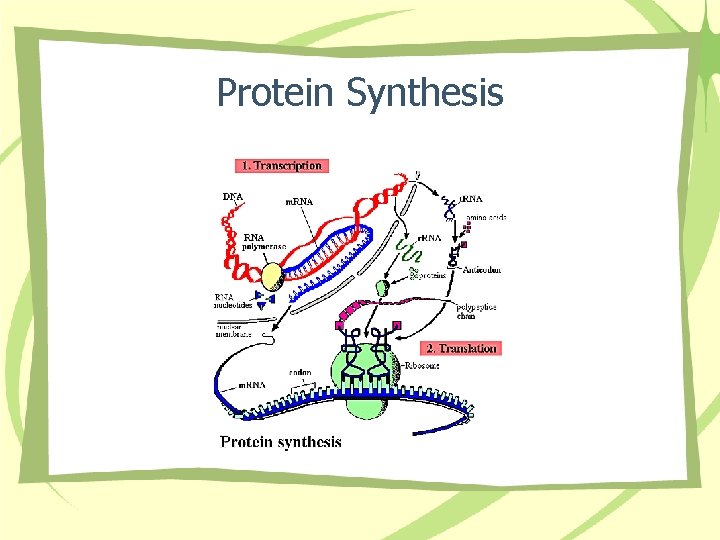
Protein Synthesis
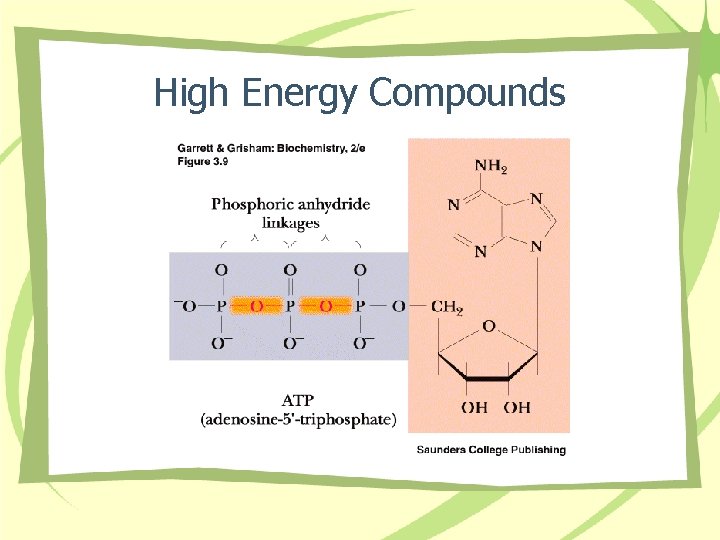
High Energy Compounds

Questions?
- Slides: 59