Chemistry Chapter 7 Hydrocarbons crude oil fractional distillation
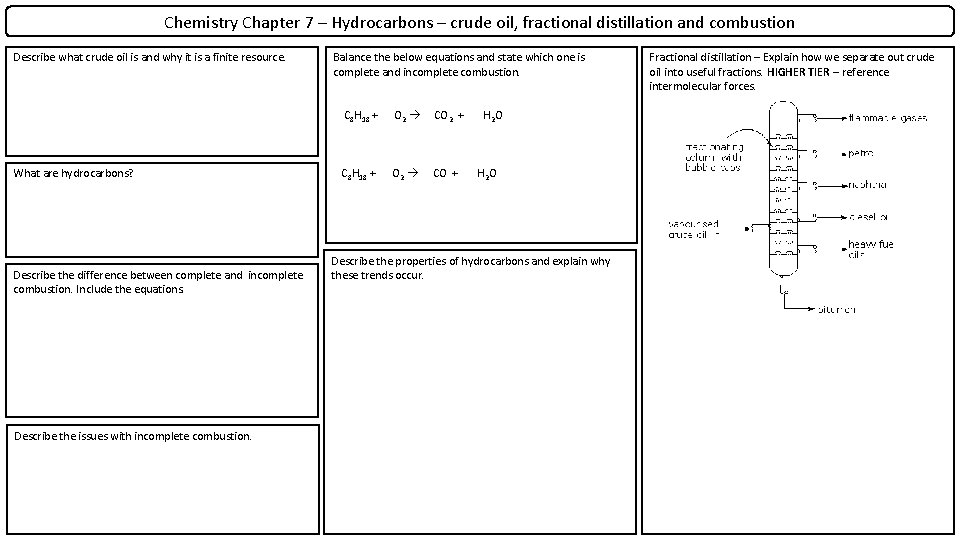
Chemistry Chapter 7 – Hydrocarbons – crude oil, fractional distillation and combustion Describe what crude oil is and why it is a finite resource. What are hydrocarbons? Describe the difference between complete and incomplete combustion. Include the equations. Describe the issues with incomplete combustion. Balance the below equations and state which one is complete and incomplete combustion. C 8 H 18 + O 2 CO 2 + C 8 H 18 + O 2 CO + H 2 O Describe the properties of hydrocarbons and explain why these trends occur. Fractional distillation – Explain how we separate out crude oil into useful fractions. HIGHER TIER – reference intermolecular forces.
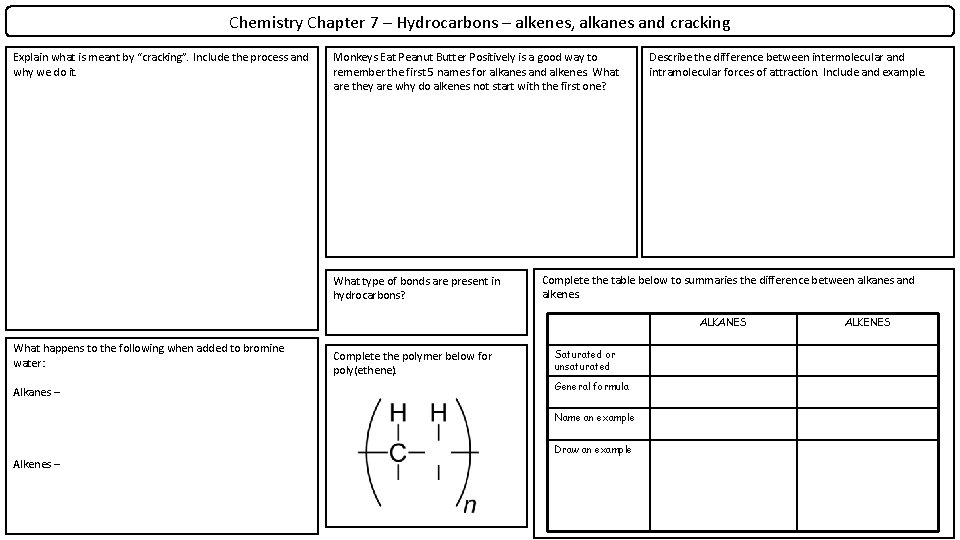
Chemistry Chapter 7 – Hydrocarbons – alkenes, alkanes and cracking Explain what is meant by “cracking”. Include the process and why we do it. Monkeys Eat Peanut Butter Positively is a good way to remember the first 5 names for alkanes and alkenes. What are they are why do alkenes not start with the first one? What type of bonds are present in hydrocarbons? Describe the difference between intermolecular and intramolecular forces of attraction. Include and example. Complete the table below to summaries the difference between alkanes and alkenes. ALKANES What happens to the following when added to bromine water: Alkanes – Complete the polymer below for poly(ethene). Saturated or unsaturated General formula Name an example Draw an example Alkenes – ALKENES
- Slides: 2