Chemistry Chapter 3 Scientific Measurement 3 1 Qualitative
































- Slides: 34

Chemistry Chapter 3 Scientific Measurement

3. 1 Qualitative measurements – measurements that give results in a descriptive, non -numerical form. Examples: He is tall Electrons are tiny

Quantitative measurements – measurement that gives results in a definite form, usually as numbers and units. Examples: He is 2. 2 m tall Electrons are 1/1840 times the mass of a proton

Scientific Notation –a number is written as the product of two numbers: a coefficient and 10 raised to a power. Examples: 567000 = 5. 67 X 105 0. 00231 = 2. 31 X 10 -3

Examples: Convert to or from Scientific Notation: 241 6015. 0162. 512 6. 62 x 102 3. 4 x 10 -3 = = = 2. 41 x 102 6. 015 x 103 1. 62 x 10 -2 5. 12 x 10 -1 662. 0034

CALCULATOR PRACTICE (This is important to master!!!) FYI: “EE” button on calc= typing “X 10^” 6. 25 x 103 - 2. 01 x 102 = 6. 05 x 103 (2. 15 x 103)(6. 1 x 105)(5. 0 x 10 -6) = 3. 25 x 108 3. 6 x 107 = 9. 03 6. 6 x 103

3. 2 Accuracy – the measure of how close a measurement comes to the actual or true value of whatever is measured. – how close a measured value is to the accepted value. Precision – the measure of how close a series of measurements are to one another.

Acc ura and te Pre cis e i e N r e th Precise

Percent Error Formula: % Error = accepted value- experimental value accepted value x 100 *always a positive number- indicated by the absolute value sign* You will use this formula when checking the accuracy of your experiment.

Significant Figures – includes all of the digits that are known plus a last digit that is estimated. FYI: These rules are IMPORTANT and they will save you many points in the future if you learn them NOW! !

Rules for determining Significant Figures 1. All non-zero digits are significant. 1, 2, 3, 4, 5, 6, 7, 8, 9

2. Zeros between non-zero digits are significant. (AKA zero sandwich) 102 7002

3. Leading zeros (zeros at the beginning of a measurement) are NEVER significant. 0. 0152 00542

4. Trailing zeros (zeros after last integer) are significant only if the number contains a decimal point. 210. 0 0. 860 210

5. All digits are significant in scientific notation. 2. 1 x 10 -5 6. 02 x 1023

Examples: How many significant digits do each of the following numbers contain: a) 1. 2 b) 2. 0 c) 3. 002 2 2 4 d) 4600 2 e) 23. 450 5 f) 6. 02 x 1023 3

Exact numbers have unlimited Significant Figures Do not use these when you are figuring out sig figs… Examples: 1 dozen = exactly 12 29 people in this room

Rounding Rules: 5 round up < 5 round down (don’t change) Examples: Round 42. 63 to 1 significant digit = 40 Round 61. 57 to 3 sig. digs. = 61. 6 Round 0. 01621 to 2 = 0. 016 Round 65, 002 to 2 sig. digs. = 65, 000

Bell. Work Thursday • • • How many sig figs? 45, 000 7. 80 x 10 -3 20040 0. 00243 700 940. 0 Round to 2 sig figs Round to 3 sig figs 70500 72. 562

Addition and Subtraction The measurement with the fewest significant figures to the right of the decimal point determines the number of significant figures in the answer.

Examples: Solve using correct significant figures 45. 756 m + 75. 263 m + 62. 1 m = 1123. 93 m 107. 9 = 1199. 19

Multiplying and Dividing Measurements The measurement with the fewest significant figures determines the number of significant figures in the answer.

Examples: Solve using correct significant figures: 3. 43 m X 45. 756 m 6. 4253 m = X 1. 2 m = 45. 01 m 2 / 2. 2 m = 22. 0 m 2 55 m 2 20. m Notice the decimal!

Uncertainty In lab, you record all numbers you know for sure plus the first uncertain digit. The last digit is estimated and is said to be uncertain but still considered significant. ØGraduated cylinders have markings to the nearest m. L (milliliter) and you will determine volume to the nearest 0. 1 m. L… because that is ONE DIGIT OF UNCERTAINTY.

International System of Units • revised version of the metric system • abbreviated SI All units, their meanings and values can be found on pgs. 63, 64, 65. Meter (m) – SI unit for length Liter (L) – SI unit for volume Gram (g) – SI unit for mass

-Mass – (g) amount of matter in an object -Volume – (m. L) amount of space occupied by an object -Density – (g/m. L) a ratio of mass to volume

Formula: m D= v Rewrite this formula to solve for m & v! What is the unit for Density? ? Remember: A material has the same density no matter how big or small it is!

Example: • A piece of metal has a volume of 4. 70 m. L and a mass of 57. 3 g. What is the density? M = 57. 3 g D=M/V V = 4. 70 m. L D = 57. 3 g / 4. 70 m. L D = 12. 19148936 g/m. L D = 12. 2 g/m. L

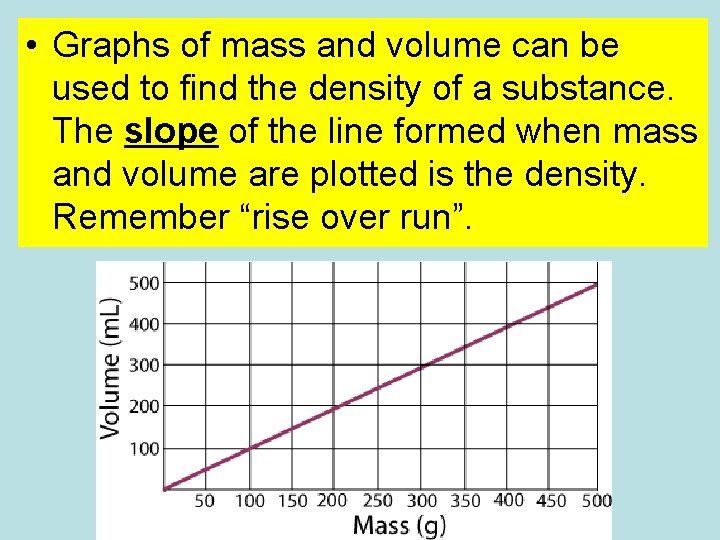
• Graphs of mass and volume can be used to find the density of a substance. The slope of the line formed when mass and volume are plotted is the density. Remember “rise over run”.

Temperature – measurement of the average kinetic energy of a system.

Temperature Scales Celsius • Sets the freezing point of water at 0 C and the boiling point at 100 C Kelvin • Absolute zero is set as the zero on the Kelvin scale. It is the temperature at which all motion theoretically ceases.

To convert: K = ºC + 273 (Kelvin does not use “degrees”. ) -273 º C = 0 K = absolute zero

Examples: Convert 25 º C to Kelvin. K = ºC + 273 K = 25ºC + 273 = 298 K

Bell Work Friday • Give Answer in proper sig figs 1. 1. 36 - 0. 253 = 2. 0. 25 m + 6. 0 m + 0. 3 m = 3. ( 2. 3 m)( 2. 8 m) = 4. 10. 33 s 2 / 1. 25 s =