CHEMISTRY Chapter 16 AcidBase Equilibria Acids and Bases

CHEMISTRY Chapter 16 Acid-Base Equilibria

Acids and Bases: A Brief Review • Acids: taste sour and cause dyes to change color. • Bases: taste bitter and feel soapy. • Arrhenius: acids increase [H+]; bases increase [OH-] in solution. • Arrhenius: acid + base salt + water. • Problem: the definition confines us to aqueous solution.

STRONG ACIDS • Strong Acids – totally ionize; full conversion to product, Ka>>1; no equilibrium involved in the ionization • HBr (aq) 2. 5 M H+ (aq) 2. 5 M + OH- (aq) 2. 5 M

Acid Strength • Strength of acid is given by the equilibrium position of the dissociation reaction: • HA (aq) + H 2 O (l) H 3 O + + A - • Strong acid – totally ionized and equilibrium lies far to the right • Weak acid – only partially ionized and equilibrium lies far to the left

STRONG BASES • Strong Bases – totally ionize; full conversion to product, Kb>>1; no equilibrium involved in the ionization • KOH (aq) 2. 5 M K+ (aq) 2. 5 M + OH- (aq) 2. 5 M

List of STRONG ACIDS • • 1. 2. 3. 4. 5. 6. 7. HCl HBr HI HNO 3 H 2 SO 4 HCl. O 3

List of Strong Bases • Grp I (except H) + 1 OH • Grp II (except Mg) + 2 OH • Other Metals plus # OH- depending upon the metal’s charge

Acids and Bases • Arrhenius’ Definition: • Acids - are substances that produce hydrogen ions (protons or H+) in solution. Bases - are substances that produce hydroxide ions in solution. Strong Acids and Strong Bases – totally ionize in solution Weak Acids and Weak Bases – partially ionize in solution

Bronsted-Lowry • Bronsted-Lowry Acid – any compound that donates H+ • Bronsted-Lowry Base – any compound that accepts H+ • - does not need to have OH in chemical formula • - ex. NH 3

Acid Dissociation in Water • General Rxn. when Acid dissolves in H 2 O • HCl + acid H 2 O base H 3 O+ + Clconj. Acid conj. base

Properties of H 2 O @ 25 o. C • H 2 O (l) D H+ (aq) + OH- (aq) • Neutral, can act as an acid and a base so it is AMPHOTERIC. • Kw = [H+][OH-] = 1. 0 x 10 -14 only @ 25 o. C • Kw = water dissociation constant
![Acidity vs. Basicity • If [H+] >[OH-] , solution is acidic • If [H+] Acidity vs. Basicity • If [H+] >[OH-] , solution is acidic • If [H+]](http://slidetodoc.com/presentation_image_h2/7f2aced56c5c89c40c4e94fb5dc9fbf6/image-12.jpg)
Acidity vs. Basicity • If [H+] >[OH-] , solution is acidic • If [H+] <[OH-] , solution is basic • The term p. X = -log [concentration of X] • So: p. H = -log [concentration of H+] • p. OH = -log [concentration of OH-] • p. H = power of hydrogen; the power of H to which 10 is raised
![p. H • Kw = [H+][OH-] = 1. 0 x 10 -14 @ 25 p. H • Kw = [H+][OH-] = 1. 0 x 10 -14 @ 25](http://slidetodoc.com/presentation_image_h2/7f2aced56c5c89c40c4e94fb5dc9fbf6/image-13.jpg)
p. H • Kw = [H+][OH-] = 1. 0 x 10 -14 @ 25 o. C • p. Kw = [-log H+] + [-log OH-]= - [log 1. 0 x 10 -14] = 14 • p. H = -log [H+] • p. OH = -log [OH-]

Properties of H 2 O @ 25 o. C • p. Kw = - [log 1. 0 x 10 -14] = 14 • p. H + p. OH = 14 • p. H = 7 • p. H = p. OH = 7
![Things to Remember • p. Kw = - [log 1. 0 x 10 -14] Things to Remember • p. Kw = - [log 1. 0 x 10 -14]](http://slidetodoc.com/presentation_image_h2/7f2aced56c5c89c40c4e94fb5dc9fbf6/image-15.jpg)
Things to Remember • p. Kw = - [log 1. 0 x 10 -14] = 14 @ 25 o. C • p. H + p. OH = 14 • p. H <7 ; acidic • p. H > 7; basic • p. H range is between 0 – 14 @ 25 o. C. Note that there are p. Hs below ZERO and above 14. • At different temperatures, the scale also changes and 7 is NOT the neutral p. H.
![The p. H Scale • In most solutions [H+(aq)] is quite small. • We The p. H Scale • In most solutions [H+(aq)] is quite small. • We](http://slidetodoc.com/presentation_image_h2/7f2aced56c5c89c40c4e94fb5dc9fbf6/image-16.jpg)
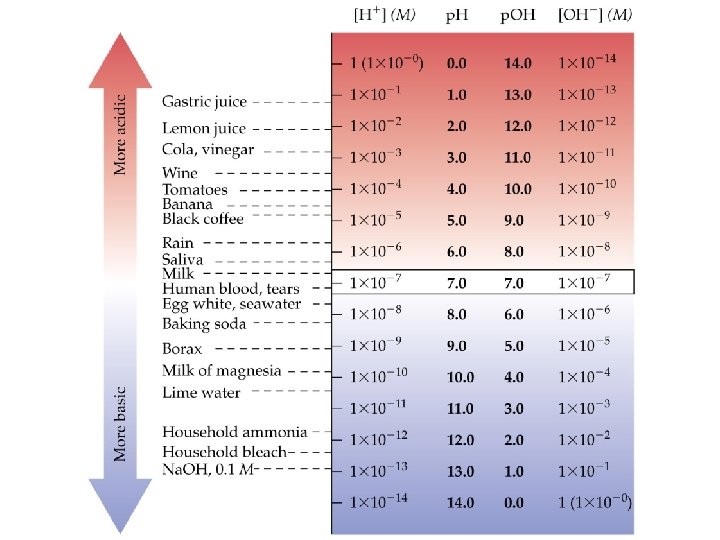
The p. H Scale • In most solutions [H+(aq)] is quite small. • We define • • In neutral water at 25 C, p. H = p. OH = 7. 00. In acidic solutions, [H+] > 1. 0 10 -7, so p. H < 7. 00. In basic solutions, [H+] < 1. 0 10 -7, so p. H > 7. 00. The higher the p. H, the lower the p. OH, the more basic the solution.

Broensted-Lowry’s Definition • • Acid – is any compound that is a proton (H+) donor. • Base – is any compound that is a proton (H+) acceptor. • * Broensted-Lowry Definition is more general – It even applies to bases that have no –OH in the chemical formula such as NH 3.

Terminologies • • • H+ OHH 3 O+ HA A- = = = proton hydroxide ion hydronium ion abbreviation for ALL acids abbreviation for ALL bases • Conjugate base –acid minus proton • Conjugate acid – base plus proton

• Consider the reactions: • HA (aq) + H 2 O (l) • A- (aq) + H 2 O (l) H 3 O+ (aq) + A- (aq) conj. acid conj. base HA (aq) + OH- (aq) conj. acid conj. base

More Terminologies • Conjugate acid-base pair – Consists of 2 substances related to each other by the donation and acceptance of a single proton (H+). • Acid Dissociation Constant (Ka)

Strong Acids and Bases Strong Acids • In solutions the strong acid is usually the only source of H+. (If the molarity of the acid is less than 10 -6 M then the autoionization of water needs to be taken into account. ) • Therefore, the p. H of the solution is the initial molarity of the acid. Strong Bases • Most ionic hydroxides are strong bases (e. g. Na. OH, KOH, and Ca(OH)2).

Strong Acids and Bases • If the molarity of the acid or base is less than 10 -6 M then the autoionization of water needs to be taken into account. In other words, water is the primary source of H+ and OH-, so the p. H would be neutral.

Strong Acids and Bases • • Strong Bases Strong bases are strong electrolytes and dissociate completely in solution. The p. OH (and hence p. H) of a strong base is given by the initial molarity of the base. Be careful of stoichiometry. In order for a hydroxide to be a base, it must be soluble. Bases do not have to contain the OH- ion: O 2 -(aq) + H 2 O(l) 2 OH-(aq) + H 2 O(l) H 2(g) + OH-(aq) N 3 -(aq) + H 2 O(l) NH 3(aq) + 3 OH-(aq)

p. H of Strong Acids and Bases • The p. H (and hence p. OH) of a strong acid is given by the initial molarity of the acid. • The p. OH (and hence p. H) of a strong base is given by the initial molarity of the base. • Be careful of stoichiometric ratios!
![Equations • • p. H = - log [H+] p. OH = - log Equations • • p. H = - log [H+] p. OH = - log](http://slidetodoc.com/presentation_image_h2/7f2aced56c5c89c40c4e94fb5dc9fbf6/image-25.jpg)
Equations • • p. H = - log [H+] p. OH = - log [OH-] [H+] = 10 – p. H [OH-] = 10 - p. OH Kw = 10 - p. Kw = p. H + p. OH p. Kw = - log [Kw] Kw = [H+][OH-]

Sample Problem • At 40 o. C, a solution has Kw = 2. 916 x 10 -14; p. H = 7. 51 • • • Calculate the following: A. p. OH of the solution B. hydrogen ion concentration [H+] C. hydroxide ion concentration [OH-] D. p. Kw E. Is the solution acidic basic or neutral?

Brønsted-Lowry Acids and Bases Conjugate Acid-Base Pairs • Whatever is left of the acid after the proton is donated is called its conjugate base. • Similarly, whatever remains of the base after it accepts a proton is called a conjugate acid. • Consider – After HA (acid) loses its proton it is converted into A- (base). Therefore HA and A- are conjugate acid-base pairs. – After H 2 O (base) gains a proton it is converted into H 3 O+ (acid). Therefore, H 2 O and H 3 O+ are conjugate acid-base pairs. • Conjugate acid-base pairs differ by only one proton.

Acid Equilibrium • Equilibrium Expression for the reaction: • HA (aq) + H 2 O (l) acid base H 3 O+ (aq) + A- (aq) conj. Acid conj. Base • Ka = [H+][A-] [HA] [H 3 O+ ][A- ] = [HA]
![Equilibrium • Ka = [H 3 O+ ][A- ] = [HA] [H+][A-] [HA] – Equilibrium • Ka = [H 3 O+ ][A- ] = [HA] [H+][A-] [HA] –](http://slidetodoc.com/presentation_image_h2/7f2aced56c5c89c40c4e94fb5dc9fbf6/image-29.jpg)
Equilibrium • Ka = [H 3 O+ ][A- ] = [HA] [H+][A-] [HA] – Note that H 3 O+ is simply H+ dissolved in water. • Remember: Keq = [products]n [reactants]n

Problems on Acid Dissociation • Write the simple dissociation reaction for each of the following acids. Omit water. • • A. ) B. ) C. ) D. ) HNO 3 CH 3 COOH (acetic acid) NH 4+ [Al(H 2 O)3]3+

Strong Acid vs. Weak Acid • Strong Acid – yields a weak conjugate base (one that has weak affinity for proton; weaker than H 2 O) • Weak Acid – yields a strong conjugate base (one that has strong affinity for proton; stronger than H 2 O)
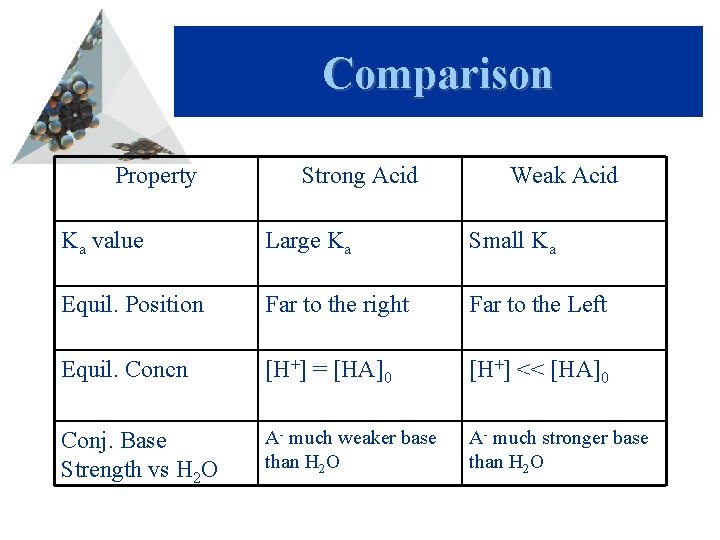
Comparison Property Strong Acid Weak Acid Ka value Large Ka Small Ka Equil. Position Far to the right Far to the Left Equil. Concn [H+] = [HA]0 [H+] << [HA]0 Conj. Base Strength vs H 2 O A- much weaker base than H 2 O A- much stronger base than H 2 O
![Sample Problems • Given [OH-] = 1. 0 x 10 -12 M, calculate p. Sample Problems • Given [OH-] = 1. 0 x 10 -12 M, calculate p.](http://slidetodoc.com/presentation_image_h2/7f2aced56c5c89c40c4e94fb5dc9fbf6/image-33.jpg)
Sample Problems • Given [OH-] = 1. 0 x 10 -12 M, calculate p. H. Is the solution basic, acidic or neutral? • Given [H+] = 4. 30 x 10 -6 M, calculate p. H. Is the solution basic, acidic or neutral?

Strong Acids and Bases • If the molarity of the acid or base is less than 10 -6 M then the autoionization of water needs to be taken into account. In other words, water is the primary source of H+ and OH-, so the p. H would be neutral.

Brønsted-Lowry Acids and Bases Relative Strengths of Acids and Bases • The stronger the acid, the weaker the conjugate base. • H+ is the strongest acid that can exist in equilibrium in aqueous solution. • OH- is the strongest base that can exist in equilibrium in aqueous solution.
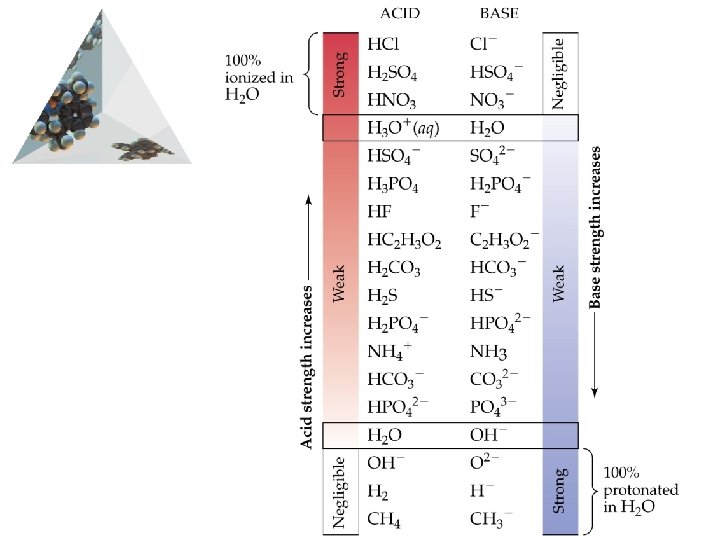

Brønsted-Lowry Acids and Bases Relative Strengths of Acids and Bases • The conjugate base of a strong acid (e. g. Cl-) has negligible acid-base properties. • Similarly, the conjugate acid of a strong base has negligible acid-base properties.

The Autoionization of Water The Ion Product of Water • In pure water the following equilibrium is established • at 25 C • The above is called the autoionization of water.

The p. H Scale • Most p. H and p. OH values fall between 0 and 14. • There are no theoretical limits on the values of p. H or p. OH. (e. g. p. H of 2. 0 M HCl is -0. 301. ) • p. Hs can be below Zero and above 14.

Strong Acids and Bases Strong Acids • The strongest common acids are HCl, HBr, HI, HNO 3, HCl. O 4, and H 2 SO 4. • are strong electrolytes. • All strong acids ionize completely in solution:

Please Note! • Tuesday’s experiment is Experiment 29: Choice I.

Bronsted-Lowry Acids and Bases • Bronsted-Lowry acids – compounds that donate a proton (H+) • Bronsted-Lowry Bases – compounds that accept a proton (H+) • Note that Bronsted-Lowry bases need not have the –OH group on the formula

Weak Acids • Weak acids are only partially ionized in solution. • There is a mixture of ions and unionized acid in solution. • Therefore, weak acids are in equilibrium:

NOTE • For Weak Acids and Weak Bases: • USE ICE to determine p. H and p. OH. ! + H, OH ,

Sample A Problem • A solution of 0. 10 M formic acid (HCOOH) has a p. H of 2. 38 at 25 o. C. • A. Calculate Ka formic acid at this temperature. • B. What percent of this solution is ionized?

Sample Problem • The Ka of acetic acid is 1. 8 x 10 -5. • A. Calculate the p. H of a 0. 30 M solution of CH 3 COOH. • B. Calculate OH- and p. OH. • C. Calculate Kb. • Calculate % ionization.

A Simple Trick • Use of approximation: eliminates the difficulty of quadratic equations. • Approximation is Valid if: X_______ x 100 < 5 % [Initial Concn. ]
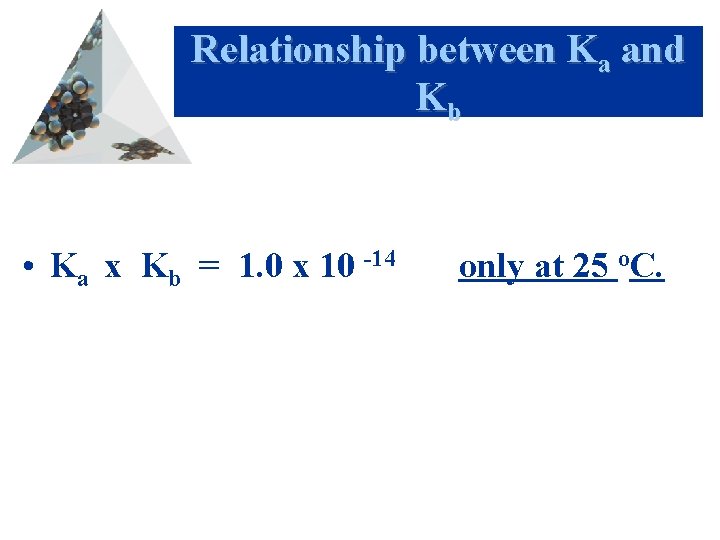
Relationship between Ka and Kb • Ka x Kb = 1. 0 x 10 -14 only at 25 o. C.

p. H of polyprotic acids • Treat polyprotic acids as separate steps! • #1. H 2 A (aq) D H+ (aq) + HA- (aq) Ka 1 • # 2. HA- (aq) D H+ (aq) + A-2 (aq) Ka 2 • Initial [H+] in Step 2 is Equil. [H+] from Step 1. • Total [H+] = SUM from Steps 1 & 2

HOMEWORK • What is the p. H of a 1. 00 M solution of tartaric acid, H 2 C 4 H 4 O 6 (aq. ) at 25. 0 o. C? • Answer: p. H = 1. 49

Sample Problem • The Ka of acetic acid is 1. 8 x 10 -5. Calculate the Kb of of CH 3 COOH.

Sample Problem • The Kb of ammonia is 1. 8 x 10 -5. Calculate the p. H of a 0. 15 M solution of NH 3.

Sample Problem • Calculate the concentration of an aqueous solution of Na. OH that has a p. H of 11. 50.

HOMEWORK • What is the p. H of a 1. 00 M solution of tartaric acid, H 2 C 4 H 4 O 6 (aq. ) at 25. 0 o. C? • Answer: p. H = 1. 49

Weak Acids Calculating Ka from p. H • Weak acids are simply equilibrium calculations. • The p. H gives the equilibrium concentration of H+. • Using Ka, the concentration of H+ (and hence the p. H) can be calculated. – Write the balanced chemical equation clearly showing the equilibrium. – Write the equilibrium expression. Find the value for Ka. – Write down the initial and equilibrium concentrations for everything except pure water. We usually assume that the change in concentration of H+ is x.

Weak Acids Calculating Ka from p. H • Substitute into the equilibrium constant expression and solve. Remember to turn x into p. H if necessary. Using Ka to Calculate p. H • Percent ionization is another method to assess acid strength. • For the reaction

Sample Problem • A solution of NH 3 in water has a p. H of 10. 50. What is the initial molarity of the solution?

Other Weak Bases • Amines ex. Methylamine (CH 3 NH 2) • carbonate ion (CO 32 -) • hypochlorite ion (Cl. O-1)

Weak Bases • Also use ICE! • Calculation is the same as for weak acids! • Main difference is that you get [OH-] and p. OH first.

Effects of Salts on p. H • • • Conjugate bases of strong acids have no effect on p. H. Conjugate acids of strong bases have no effect on p. H. Conjugate bases of weak acids increase p. H (more basic). Ex. F- (aq) + H 2 O(l) D HF (aq) + OH- (aq) Conjugate acids of weak bases decrease p. H (more acidic). NH 4+(aq) + H 2 O (l) D NH 3 (aq) + H 3 O+ (aq)

Relationship Between Ka and Kb

Acid-Base Properties of Salt Solutions Combined Effect of Cation and Anion in Solution • A cation that is the conjugate acid of a weak base will cause a decrease in the p. H of the solution. • Metal ions will cause a decrease in p. H except for the alkali metals (Grp. I) and alkaline earth metals. (Grp. II) • When a solution contains both cations and anions from weak acids and bases, use Ka and Kb to determine the final p. H of the solution.

Sample Problem • Determine whether the resulting solution in water will be acidic, basic or neutral. • • A. K+Cl. O 3 B. Na+CH 3 COOC. Na 2 HPO 4 D. NH 4+Cl- Ka for HPO 4 - = 4. 2 x 10 -13

Sample Problem • Predict whether the potassium salt of citric acid (K 2+HC 6 H 5 O 7 -) will form an acidic, basic or neutral solution in water.

Weak Acids Polyprotic Acids • Polyprotic acids have more than one ionizable proton. • The protons are removed in steps not all at once: • It is always easier to remove the first proton in a polyprotic acid than the second. • Therefore, Ka 1 > Ka 2 > Ka 3 etc.
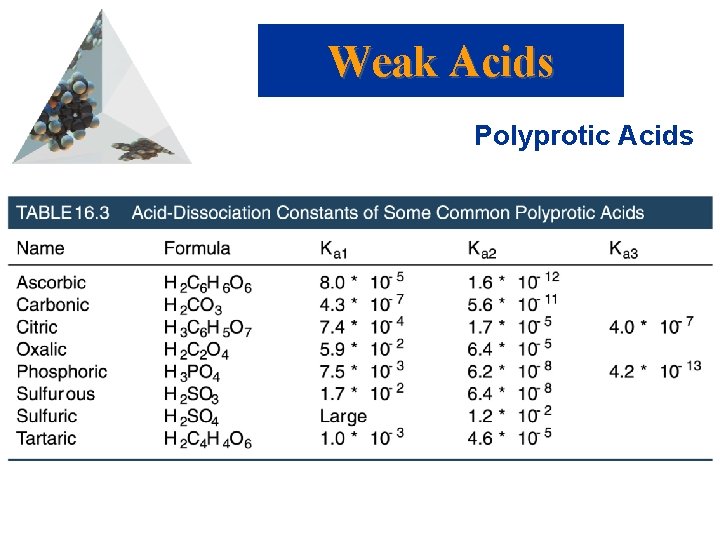
Weak Acids Polyprotic Acids

Sample Problem • The solubility of CO 2 in pure water at 25 o. C and 0. 1 atm is 0. 0037 M. The common practice is to assume that all of the dissolved CO 2 is in the form of carbonic acid (H 2 CO 3), which is produced by the reaction between the CO 2 and H 2 O. • What is the p. H of a 0. 0037 M solution of H 2 CO 3? • Ka 1 = 4. 3 x 10 -7 • Ka 2 = 5. 6 x 10 -11

Answer • p. H = 4. 4 • x 1 = 4. 0 x 10 -5 M • [CO 3 -] = 5. 6 x 10 -11 M

Sample Problem • Calculate the p. H and concentration of oxalate ion (C 2 O 42), in a 0. 020 M solution of oxalic acid (H 2 C 2 O 4)
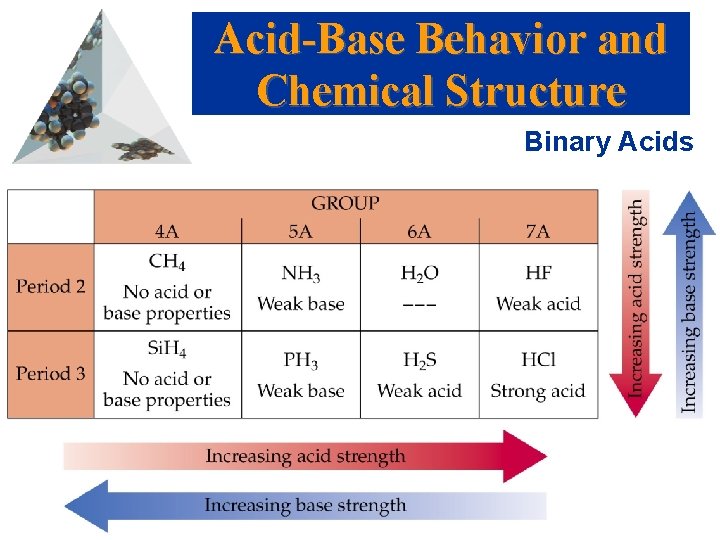
Acid-Base Behavior and Chemical Structure Binary Acids

Lewis Acids and Bases • Brønsted-Lowry acid is a proton donor. • Focusing on electrons: a Brønsted-Lowry acid can be considered as an electron pair acceptor. • Lewis acid: electron pair acceptor. • Lewis base: electron pair donor. • Note: Lewis acids and bases do not need to contain protons. • Therefore, the Lewis definition is the most general definition of acids and bases.

Lewis Acids and Bases • Lewis acids generally have an incomplete octet (e. g. BF 3). • Transition metal ions are generally Lewis acids. • Lewis acids must have a vacant orbital (into which the electron pairs can be donated). • Compounds with p-bonds can act as Lewis acids: H 2 O(l) + CO 2(g) H 2 CO 3(aq)

End of Chapter 16 Acid-Base Equilibria

Problem 1 • Give the conjugate base of the following Bronsted. Lowry acids: • H 2 SO 3 • H 2 As. O 4 • NH 4+
![Problem 2 • By what factor does [H+] change for a p. H change Problem 2 • By what factor does [H+] change for a p. H change](http://slidetodoc.com/presentation_image_h2/7f2aced56c5c89c40c4e94fb5dc9fbf6/image-76.jpg)
Problem 2 • By what factor does [H+] change for a p. H change of: • A. 2. 00 units • B. 0. 50 units
![Problem 3 • Calculate [OH-] and p. H for: • A. ) 1. 5 Problem 3 • Calculate [OH-] and p. H for: • A. ) 1. 5](http://slidetodoc.com/presentation_image_h2/7f2aced56c5c89c40c4e94fb5dc9fbf6/image-77.jpg)
Problem 3 • Calculate [OH-] and p. H for: • A. ) 1. 5 x 10 -3 M Sr(OH)2 is a strong base. • B. ) a solution formed by adding 10 m. L of 0. 100 M HBr to 20. 0 m. L of 0. 200 M HCl

Problem • • Calculate the p. H of a solution made by adding 15. 00 grams of Na. F in enough water to make 2. 5 L of solution

Problem • Write the ionization and equilibrium expressions for HBr. O 2.

Problem • A particular sample of vinegar has a p. H of 2. 9. Assuming acetic acid is the only acid in the vinegar, find the initial concentration of acetic acid in the vinegar.

Problem • The acid dissociation constant for benzoic acid (HC 7 H 5 O 2) is 6. 3 x 10 -5. Calculate the equilibrium concentrations of H 3 O+, C 7 H 5 O 2 - and HC 7 H 5 O 2 if the initial concentration of HC 7 H 5 O 2 is 0. 050 M.

Problem • Calculate the p. H of 0. 120 M pyridine (C 5 H 5 N). Kb for pyridine is 1. 7 x 10 -9.

Problem • A 0. 200 M solution of a weak acid, HA is 9. 4% ionized. Using this information, calculate [H+], [A-], [HA] and Ka for HA.

Problem • An unknown salt is either Na. F, Na. Cl, or Na. OCl. When 0. 05 mole of the salt is dissolved in water to form 0. 500 L of solution, the p. H of the solution is 8. 08. What is the identity of the salt?

Problem • Write the chemical equation and the Kb expression for the ionization of the following bases in aqueous solution: • A. Dimethylamine (CH 3)2 NH • B. Formate ion (HCOO-) • C. Carbonate ion (CO 32 -)

Problem • Calculate the molar concentration of OH- ions in a 0. 075 M solution of ethylamine. • Kb of C 2 H 5 NH 2 = 6. 4 x 10 -4. • Calculate the p. H of this solution.

Problem • Ka for acetic acid (CH 3 COOH) is 1. 8 x 10 -5 while Ka for hypochlorous (HCl. O) ion is 3. 0 x 10 -8. • A. Which is the stronger acid? • B. Which is the stronger conjugate base? Acetate ion (CH 3 COO-) or chlorous (Cl. O-) ion? • C. Calculate kb values for CH 3 COO- and Cl. O-.

Solubility vs. Ksp • Solubility – refers to the quantity that dissolves to form a saturated solution. Unit is gm/liter or moles/liter for molar solubility. • - solubility if affected by temperature • Solubility product constant – is the equilibrium constant for the equilibrium that exists between the ionic solute and its saturated aqueous solution

Ksp • Solubility product constant – the equilibrium constant indicating how soluble the product is in water. • Example: Ca. F 2 (s) D Ca 2+ (aq) + 2 F- (aq) • Ksp = [Ca 2+][F-]2

Problem 1 • Give the ionization equation and Ksp expression for the reaction: • Ag 2 Cr. O 4 (s) D ? + ?

Problem 2 • The Ksp for Ca. F 2 is 3. 9 x 10 -11 at 25 o. C. Assuming that Ca. F 2 dissociates completely upon dissolving and that there are no other important equilibria affecting its solubility: • a. calculate the solubility of Ca. F 2 in moles per liter. • b. calculate the solubility of Ca. F 2 in grams per liter.

Problem 3 • The Ksp for La. F 3 is 2. 0 x 10 -19. What is the solubility of La. F 3 in water in moles per liter? • What is the solubility of La. F 3 in water in grams per liter?

Factors Affecting Solubility • Common-Ion Effect • Concentration

Problem 4 • Calculate the molar solubility of Ca. F 2 at 25 o. C in a solution that is: • A. 0. 010 M in Ca(NO 3)2 • B. 0. 025 M in Na. F

Precipitation of Ions • Remember Q, the reaction quotient? • If: Q > Ksp, prepitations occurs until Q = Ksp, equilibrium exists (saturated solution) Q < Ksp, solid dissolves until Q = Ksp.

Problem 1 • A solution contains 1. 0 x 10 -12 M Ag+ and 2. 0 x 10 -2 M Pb 2+. When Cl- is added, both Ag. Cl and Ksp precipitate from the solution. • What concentration of Cl- is necessary to begin the precipitation of each salt? • Which salt precipitates first?

Insoluble Chlorides • Of the common metals ions, only Ag+, Hg 2 2+, Pb 2+ form insoluble chlorides.

Qualitative Analysis • Order of separation of ions: Cl- S 2 - (OH)- (PO 4) 3 - NH 4+ 1 st step: 2 nd step: 3 rd step: 4 th step: add 6 M HCl add H 2 S and 0. 20 M HCl add (NH 4)2 S add (NH 4)2 HPO 4 and NH 3
- Slides: 98