Chemistry Bonding Naming Unit Covalent Compounds Covalent Bonds

Chemistry Bonding & Naming Unit Covalent Compounds
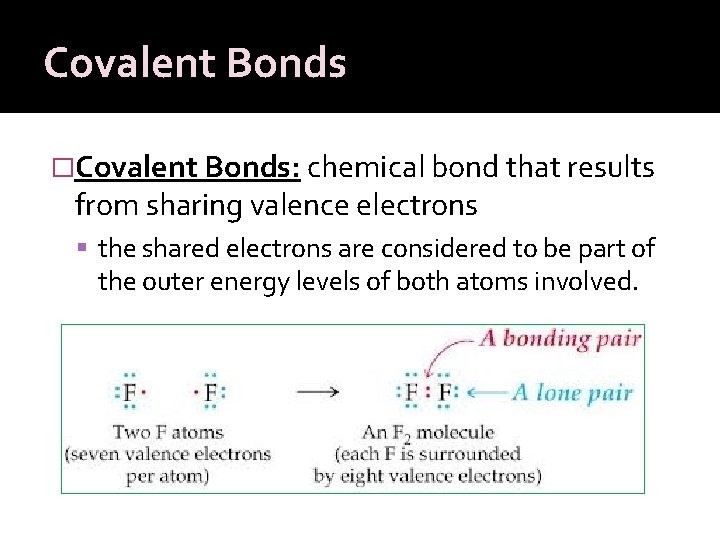
Covalent Bonds �Covalent Bonds: chemical bond that results from sharing valence electrons the shared electrons are considered to be part of the outer energy levels of both atoms involved.

Covalent Compounds �Characteristics: Between nonmetals Sharing of electrons Low electronegativity difference Attractive forces not as strong as ionic so low MP and BP

Polar Covalent �Unequal sharing of electrons, occurs between different nonmetal atoms �EN difference is b/w 0. 5 and 2. 0 �Partial charges are present on the atoms

Nonpolar Covalent �Equal sharing of electrons, occurs between same or similar nonmetals �EN difference is less than 0. 5 �All diatomic molecules (HOBr. FINCl) �No charges are present
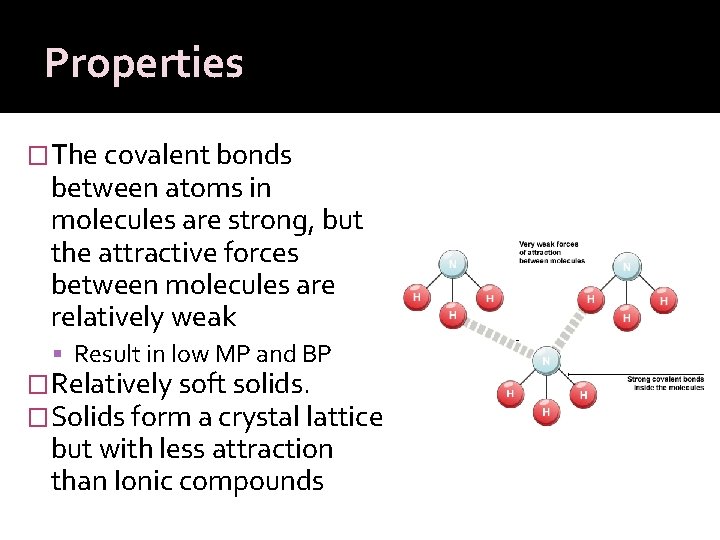
Properties �The covalent bonds between atoms in molecules are strong, but the attractive forces between molecules are relatively weak Result in low MP and BP �Relatively soft solids. �Solids form a crystal lattice but with less attraction than Ionic compounds


Writing Names � 3 Naming Rules 1. ) First element is named first, using the entire element name. 2) Second element is the formula is named using its root, adding the suffix –ide. 3. ) Prefixes are used to indicate the number of atoms of each element that are present in the compound. � Exceptions: 1) First element never used mono- prefix 2) If using a prefix results in two consecutive vowels, one of the vowels is dropped to avoid an awkward pronunciation.
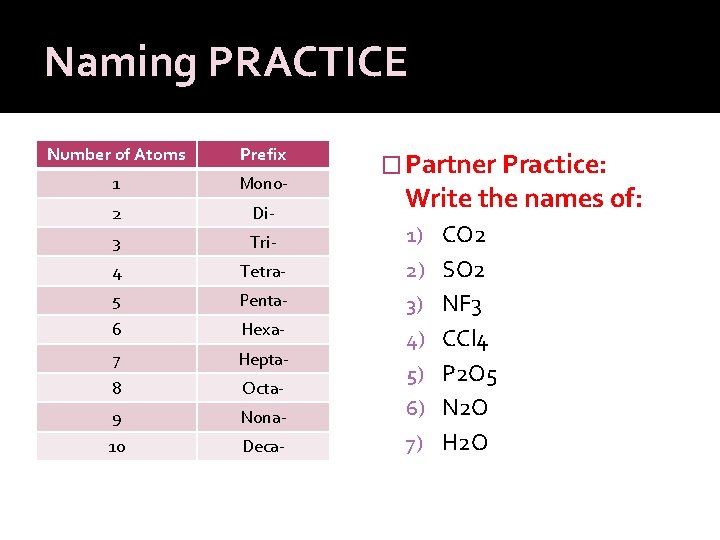
Naming PRACTICE Number of Atoms Prefix 1 Mono- 2 Di- 3 Tri- 1) CO 2 4 Tetra- 2) SO 2 5 Penta- 3) NF 3 6 Hexa- 7 Hepta- 4) CCl 4 8 Octa- 9 Nona- 10 Deca- � Partner Practice: Write the names of: 5) P 2 O 5 6) N 2 O 7) H 2 O

Writing Formulas �The prefixes used in the name indicate the exact number of each atom present in the molecule and determine the subscripts used in the formula.

Formula PRACTICE �Partner Practice: Write the formula for: 1) Chlorine Trifluoride 2) Diphosphorus trioxide 3) Disulfur decafluoride 4) Nitrogen monoxide 5) Disulfur tetroxide
- Slides: 11