Chemistry Basics Formal Tables and Graphs Ray Tedder

Chemistry Basics: Formal Tables and Graphs Ray Tedder NBC Chemistry Teacher

Foundations of Chemistry Formal Tables must have: • Table numbers look like this: Table 1. • Notice that the number is preceded by the word “Table” and that the word is capitalized. • Notice the number is followed by a period.
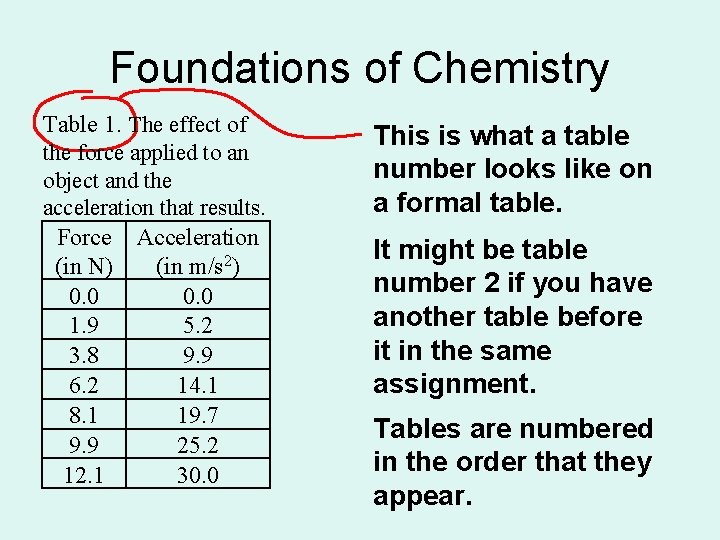
Foundations of Chemistry Table 1. The effect of the force applied to an object and the acceleration that results. Force Acceleration (in N) (in m/s 2) 0. 0 1. 9 5. 2 3. 8 9. 9 6. 2 14. 1 8. 1 19. 7 9. 9 25. 2 12. 1 30. 0 This is what a table number looks like on a formal table. It might be table number 2 if you have another table before it in the same assignment. Tables are numbered in the order that they appear.

Foundations of Chemistry Formal Tables must have: • Table description • The table description tells the reader • what is in the table and • why the information in the table is important.
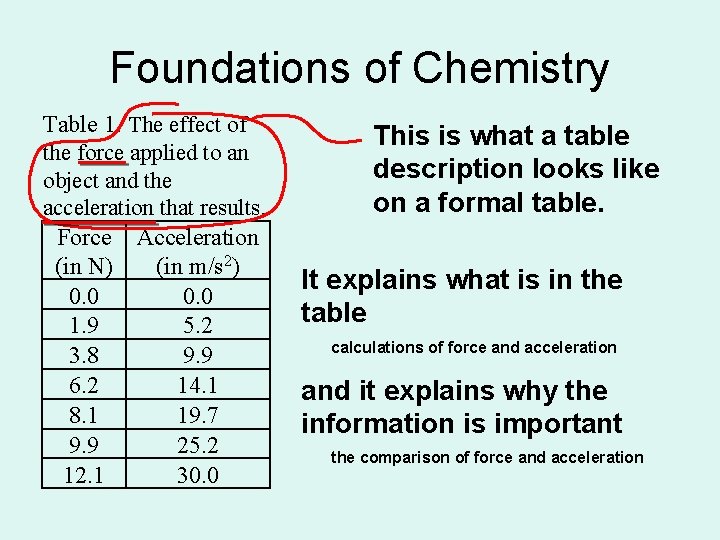
Foundations of Chemistry Table 1. The effect of the force applied to an object and the acceleration that results. Force Acceleration (in N) (in m/s 2) 0. 0 1. 9 5. 2 3. 8 9. 9 6. 2 14. 1 8. 1 19. 7 9. 9 25. 2 12. 1 30. 0 This is what a table description looks like on a formal table. It explains what is in the table calculations of force and acceleration and it explains why the information is important the comparison of force and acceleration
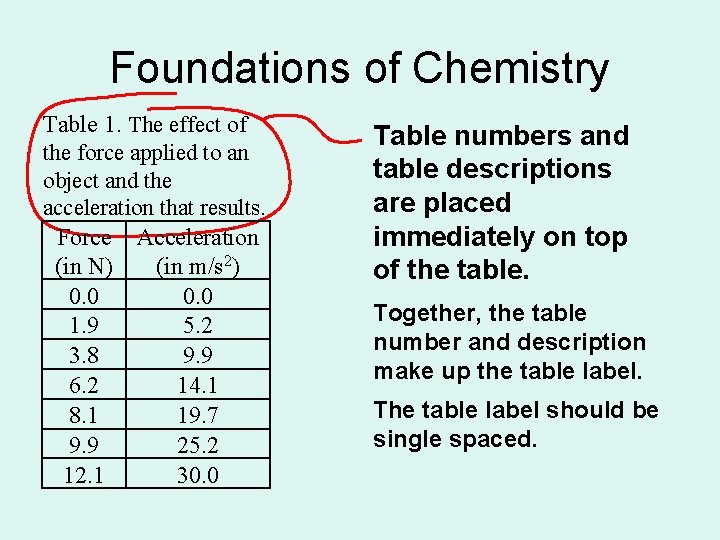
Foundations of Chemistry Table 1. The effect of the force applied to an object and the acceleration that results. Force Acceleration (in N) (in m/s 2) 0. 0 1. 9 5. 2 3. 8 9. 9 6. 2 14. 1 8. 1 19. 7 9. 9 25. 2 12. 1 30. 0 Table numbers and table descriptions are placed immediately on top of the table. Together, the table number and description make up the table label. The table label should be single spaced.

Foundations of Chemistry Formal Tables must have: • Column labels describe what observations (including measurements) or what calculated numbers are in the column • Column labels include the units of measurement in parentheses
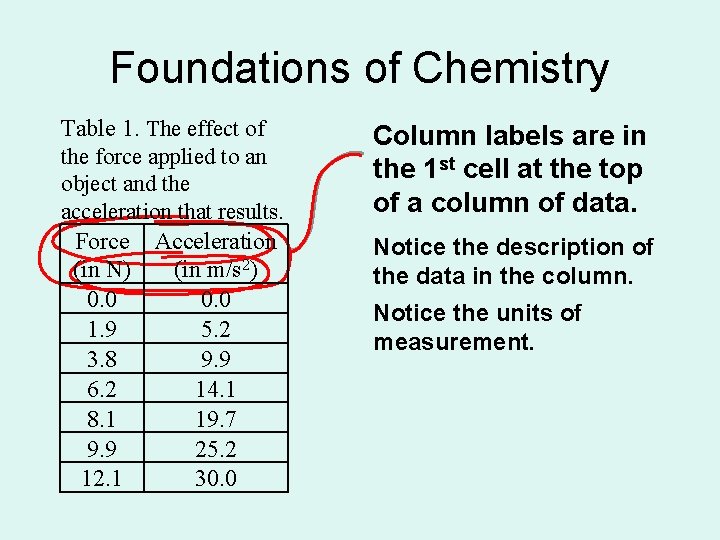
Foundations of Chemistry Table 1. The effect of the force applied to an object and the acceleration that results. Force Acceleration (in N) (in m/s 2) 0. 0 1. 9 5. 2 3. 8 9. 9 6. 2 14. 1 8. 1 19. 7 9. 9 25. 2 12. 1 30. 0 Column labels are in the 1 st cell at the top of a column of data. Notice the description of the data in the column. Notice the units of measurement.

Foundations of Chemistry Formal Tables must have: • Data cells have the actual observations (including measurements) and calculated answers • Data cells with measurements and calculated answers should not have units of measurement • Units of measurement belong in the column label
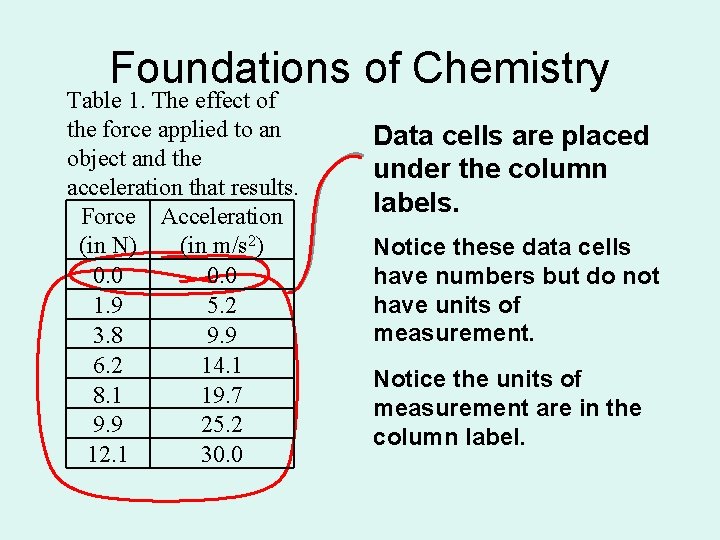
Foundations of Chemistry Table 1. The effect of the force applied to an object and the acceleration that results. Force Acceleration (in N) (in m/s 2) 0. 0 1. 9 5. 2 3. 8 9. 9 6. 2 14. 1 8. 1 19. 7 9. 9 25. 2 12. 1 30. 0 Data cells are placed under the column labels. Notice these data cells have numbers but do not have units of measurement. Notice the units of measurement are in the column label.
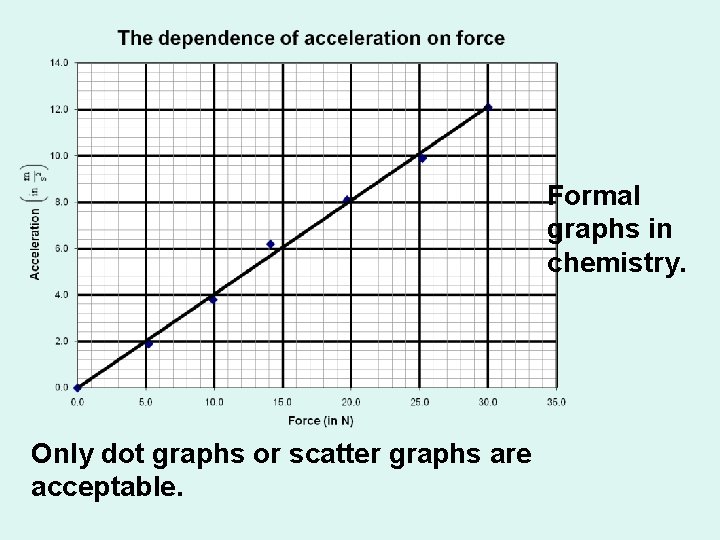
Formal graphs in chemistry. Only dot graphs or scatter graphs are acceptable.

Foundations of Chemistry Formal Graphs must have: • Titles • The title should have the following form: The dependence of (your dependent variable) on (your independent variable). • The title should be centered above the graph.

This is what a formal graph should look like. The title is written in the form where the dependent variable is mentioned 1 st and the independent variable is mentioned 2 nd.

Foundations of Chemistry Formal Graphs must have: • Axis labels must include a description of the measurements or calculations being plotted on that axis and • The units of measurement for those measurements or calculated answers.
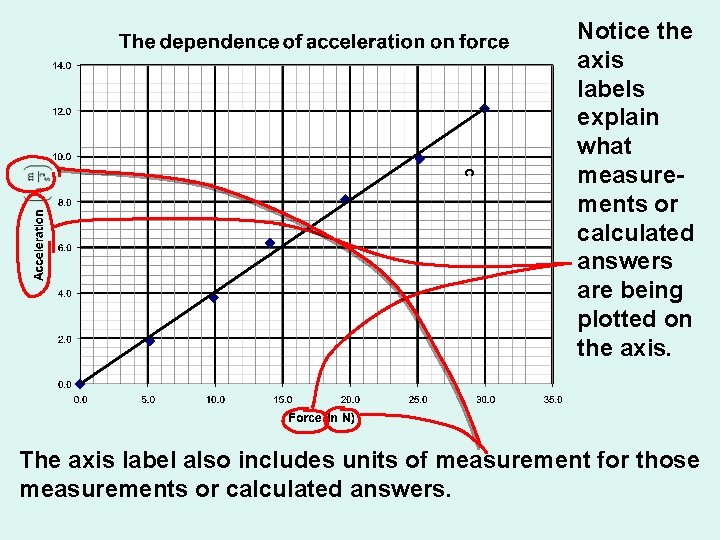
c Notice the axis labels explain what measurements or calculated answers are being plotted on the axis. The axis label also includes units of measurement for those measurements or calculated answers.

Foundations of Chemistry Formal Graphs must have: • Equally spaced number lines on each axis • Number labels are required!
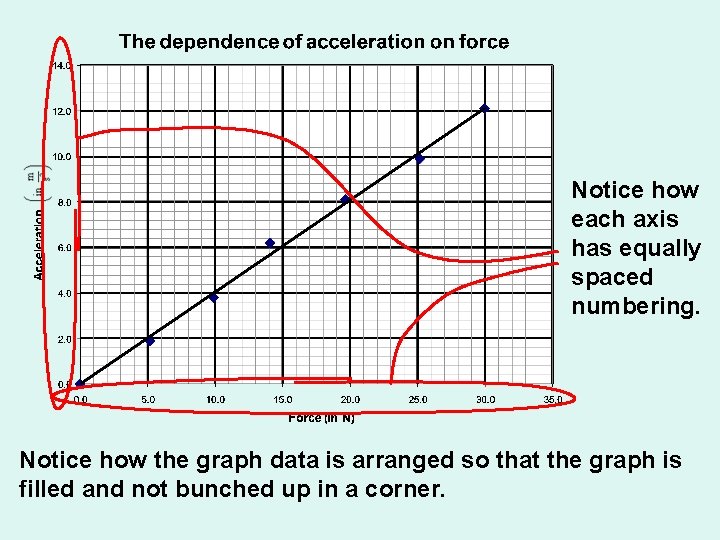
Notice how each axis has equally spaced numbering. Notice how the graph data is arranged so that the graph is filled and not bunched up in a corner.

Foundations of Chemistry Formal Graphs must have: • Figure labels go under the graph • Single spaced • Figure labels include a number (just like table numbers) • Figure labels have a description that explains to the reader what the graph shows, why it’s important, and what analysis can be determined from it.
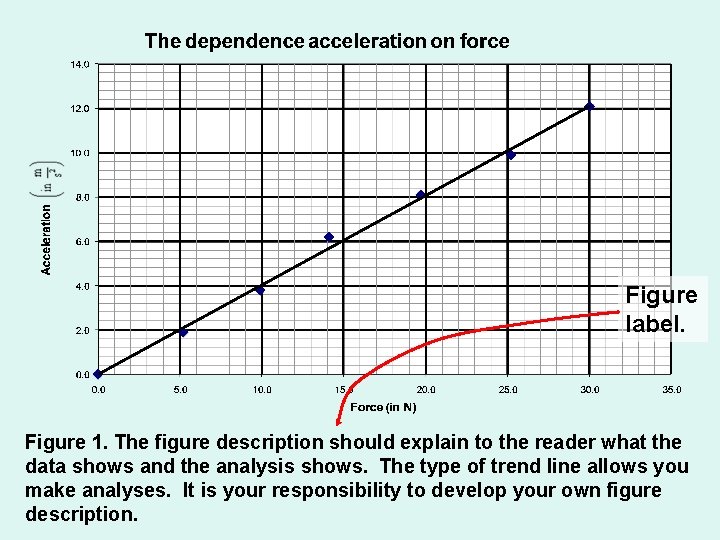
Figure label. Figure 1. The figure description should explain to the reader what the data shows and the analysis shows. The type of trend line allows you make analyses. It is your responsibility to develop your own figure description.

You MUST estimate the final digit when using an analogue device. You MUST record ALL digits measured with a digital device.
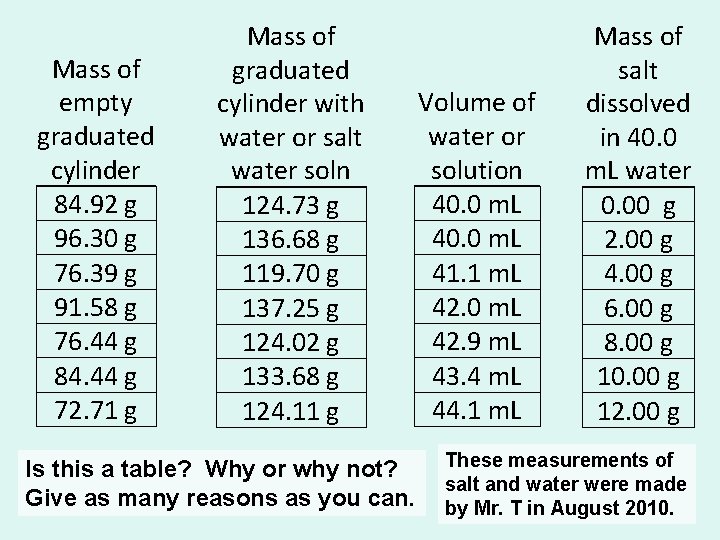
Mass of empty graduated cylinder 84. 92 g 96. 30 g 76. 39 g 91. 58 g 76. 44 g 84. 44 g 72. 71 g Mass of graduated cylinder with water or salt water soln 124. 73 g 136. 68 g 119. 70 g 137. 25 g 124. 02 g 133. 68 g 124. 11 g Is this a table? Why or why not? Give as many reasons as you can. Volume of water or solution 40. 0 m. L 41. 1 m. L 42. 0 m. L 42. 9 m. L 43. 4 m. L 44. 1 m. L Mass of salt dissolved in 40. 0 m. L water 0. 00 g 2. 00 g 4. 00 g 6. 00 g 8. 00 g 10. 00 g 12. 00 g These measurements of salt and water were made by Mr. T in August 2010.
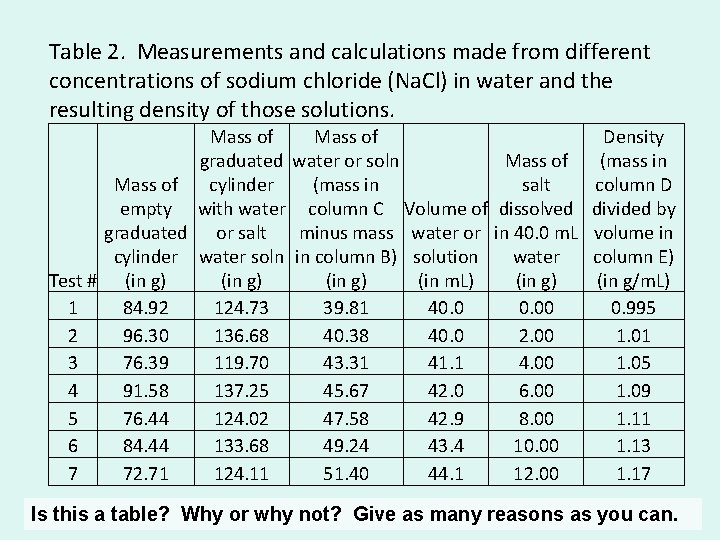
Table 2. Measurements and calculations made from different concentrations of sodium chloride (Na. Cl) in water and the resulting density of those solutions. Mass of Density graduated water or soln Mass of (mass in Mass of cylinder (mass in salt column D empty with water column C Volume of dissolved divided by graduated or salt minus mass water or in 40. 0 m. L volume in cylinder water soln in column B) solution water column E) Test # (in g) (in m. L) (in g/m. L) 1 84. 92 124. 73 39. 81 40. 00 0. 995 2 96. 30 136. 68 40. 38 40. 0 2. 00 1. 01 3 76. 39 119. 70 43. 31 41. 1 4. 00 1. 05 4 91. 58 137. 25 45. 67 42. 0 6. 00 1. 09 5 76. 44 124. 02 47. 58 42. 9 8. 00 1. 11 6 84. 44 133. 68 49. 24 43. 4 10. 00 1. 13 7 72. 71 124. 11 51. 40 44. 1 12. 00 1. 17 Is this a table? Why or why not? Give as many reasons as you can.
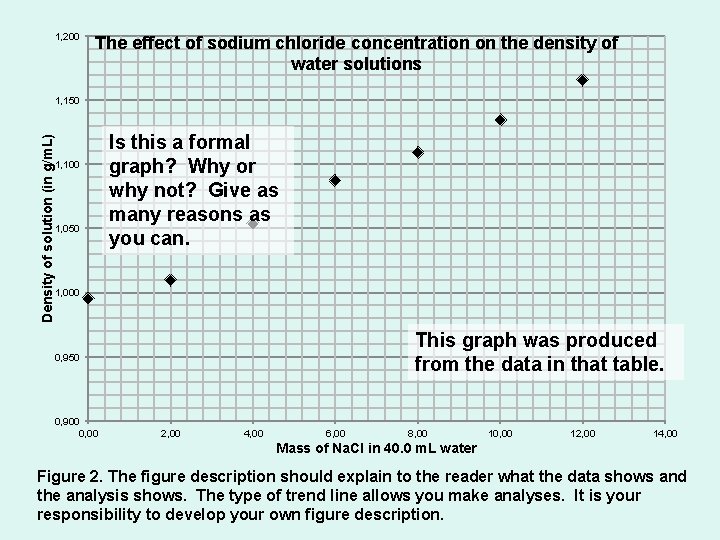
1, 200 The effect of sodium chloride concentration on the density of water solutions Density of solution (in g/m. L) 1, 150 1, 100 1, 050 Is this a formal graph? Why or why not? Give as many reasons as you can. 1, 000 This graph was produced from the data in that table. 0, 950 0, 900 0, 00 2, 00 4, 00 6, 00 8, 00 10, 00 12, 00 14, 00 Mass of Na. Cl in 40. 0 m. L water Figure 2. The figure description should explain to the reader what the data shows and the analysis shows. The type of trend line allows you make analyses. It is your responsibility to develop your own figure description.

End
- Slides: 24