Chemistry Atoms First Second Edition Julia Burdge Jason

Chemistry: Atoms First Second Edition Julia Burdge & Jason Overby Chapter 11 Gases M. Stacey Thomson Pasco-Hernando State College Copyright (c) The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display.

11. 1 Properties of Gases differ from solids and liquids in the following ways: 1) A sample of gas assumes both the shape and volume of the container. 2) Gases are compressible. 3) The densities of gases are much smaller than those of liquids and solids and are highly variable depending on temperature and pressure. 4) Gases form homogeneous mixtures (solutions) with one another in any proportion. • http: //mw. concord. org/modeler/showcase/simulation. html? s=http: //mw 2. conco rd. org/public/student/motionandforce/barometric. html • https: //phet. colorado. edu/en/simulation/balloons-and-buoyancy

11. 2 The Kinetic Molecular Theory The kinetic molecular theory explains how the molecular nature of gases gives rise to their macroscopic properties. The basic assumptions of the kinetic molecular theory are as follows: 1) A gas is composed of particles that are separated by large distances. The volume occupied by individual molecules is negligible. 2) Gas molecules are constantly in random motion, moving in straight paths, colliding with perfectly elastic collisions. 3) Gas molecules do not exert attractive or repulsive forces on one another. 4) The average kinetic energy of a gas molecules in a sample is proportional to the absolute temperature: Activity: Match the tenants of the kinetic molecular theory with the macroscopic observations of gasses that support them.

The Kinetic Molecular Theory Gases are compressible because molecules in the gas phase are separated by large distances (assumption 1). Pressure is the result of the collisions of gas molecules with the walls of their container (assumption 2). Decreasing volume increases the frequency of collisions. Pressure increases as collision frequency increases.

The Kinetic Molecular Theory Heating a sample of gas increases its average kinetic energy (assumption 4). Gas molecules must move faster. Faster molecules collide more frequently and at a greater speed. Pressure increases as collision frequency and force of collisions increases. Activity: Explain how liquids and solids react when they are heated Text Practice: 11. 5 11. 6
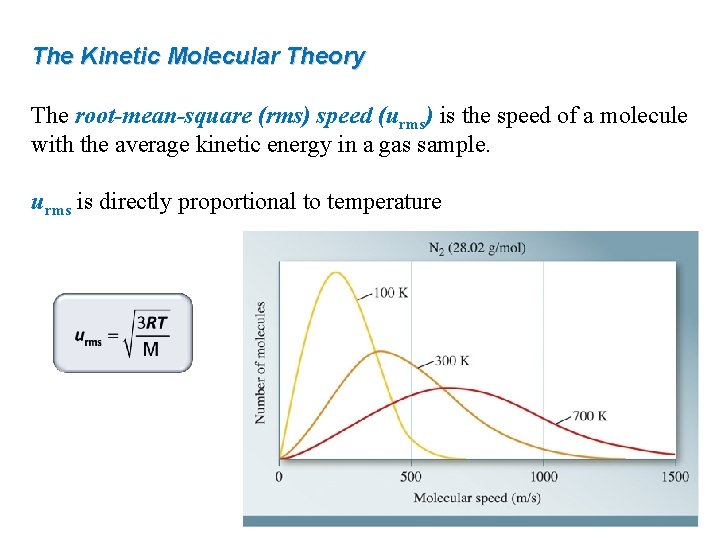
The Kinetic Molecular Theory The root-mean-square (rms) speed (urms) is the speed of a molecule with the average kinetic energy in a gas sample. urms is directly proportional to temperature
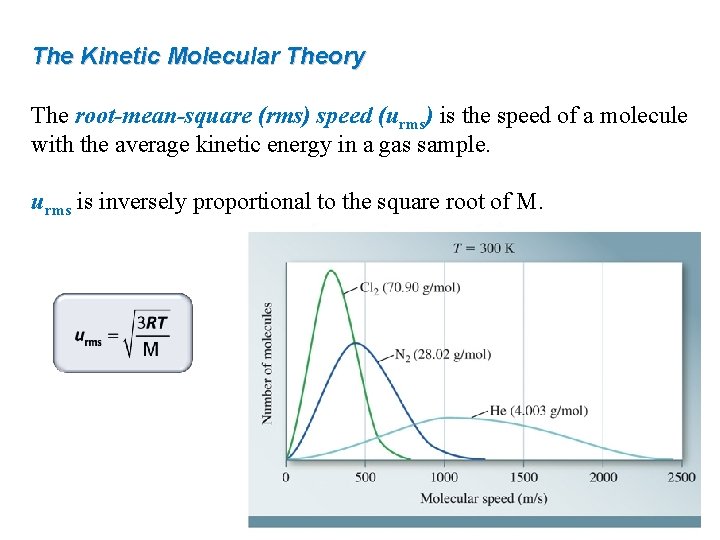
The Kinetic Molecular Theory The root-mean-square (rms) speed (urms) is the speed of a molecule with the average kinetic energy in a gas sample. urms is inversely proportional to the square root of M.

5. 8 Molecular and Formula Mass The molecular mass is the mass in atomic mass units (amu) of an individual molecule. To calculate molecular mass, multiply the atomic mass for each element in a molecule by the number of atoms of that element and then total the masses Molecular mass of H 2 O = 2(atomic mass of H) + atomic mass of O = 2(1. 008 amu) + 16. 00 amu = 18. 02 amu Because the atomic masses on the periodic table are average atomic masses, the result of such a determination is an average molecular mass, sometimes referred to as the molecular weight.

Molecular and Formula Mass Although an ionic compound does not have a molecular mass, we can use its empirical formula to determine its formula mass (the mass of a “formula unit”), sometimes called the formula weight.

Worked Example 5. 12 Calculate the molecular mass or the formula mass, as appropriate, for each of the following corresponds: (a) propane, C 3 H 8, (b) lithium hydroxide, Li. OH, and (c) barium acetate, Ba(C 2 H 3 O 2)2. Strategy Determine the molecular mass (for each molecular compound) or formula mass (for each ionic compound) by summing all the atomic masses.

The Kinetic Molecular Theory When two gases are at the same temperature, it is possible to compare the urms values of the different gases.

Worked Example 11. 1 Determine how much faster a helium atom moves, on average, than a carbon dioxide molecule at the same temperature. Strategy Use equation and the molar masses of He and CO 2 to determine the ratio of their root-mean-square speeds. When solving a problem such as. Think this, it. About is generally best to label of the two molecules It Remember thatthe thelighter relationship between molar as molecule 1 and heavier speed as molecule 2. This A ensures that the result mass andthe molecular is reciprocal. CO 2 molecule has will be greaterapproximately than 1, which is 10 relatively times theeasy masstoofinterpret. an He atom. Therefore, we should expect an He atom, on average, to be moving approximately Solution The molar 4. 003 and 44. 02 g/mol, √ 10 times (~3. 2 masses times) of as He fastand as a. CO CO 2 2 are molecule. respectively. 44. 02 g 1 mol urms(He) = = 3. 316 urms(CO 2) 4. 003 g 1 mol On average, He atoms move 3. 316 times as fast as CO 2 molecules at the same temperature.
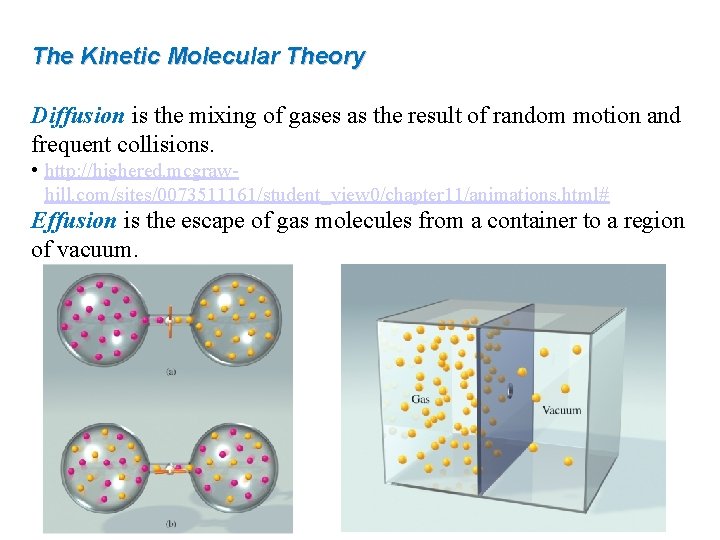
The Kinetic Molecular Theory Diffusion is the mixing of gases as the result of random motion and frequent collisions. • http: //highered. mcgrawhill. com/sites/0073511161/student_view 0/chapter 11/animations. html# Effusion is the escape of gas molecules from a container to a region of vacuum.

The Kinetic Molecular Theory Graham’s law states that the rate of diffusion or effusion of a gas is inversely proportional to the square root of its molar mass. Does the temperature impact rates of diffusion and effusion?
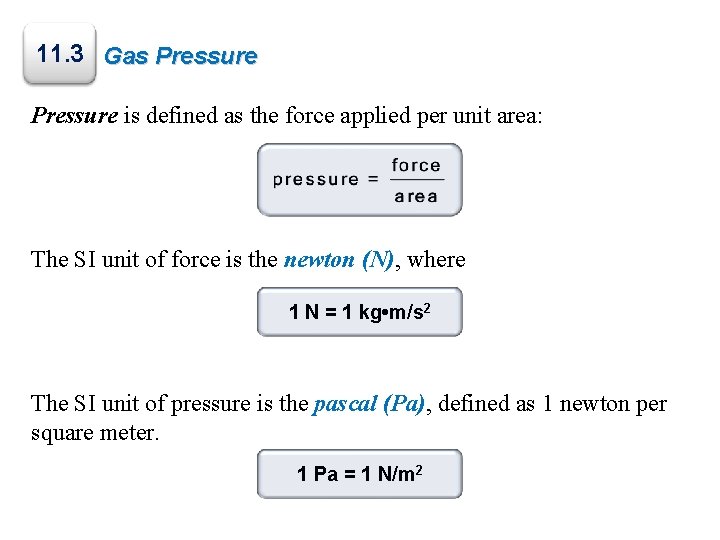
11. 3 Gas Pressure is defined as the force applied per unit area: The SI unit of force is the newton (N), where 1 N = 1 kg • m/s 2 The SI unit of pressure is the pascal (Pa), defined as 1 newton per square meter. 1 Pa = 1 N/m 2

Gas Pressure
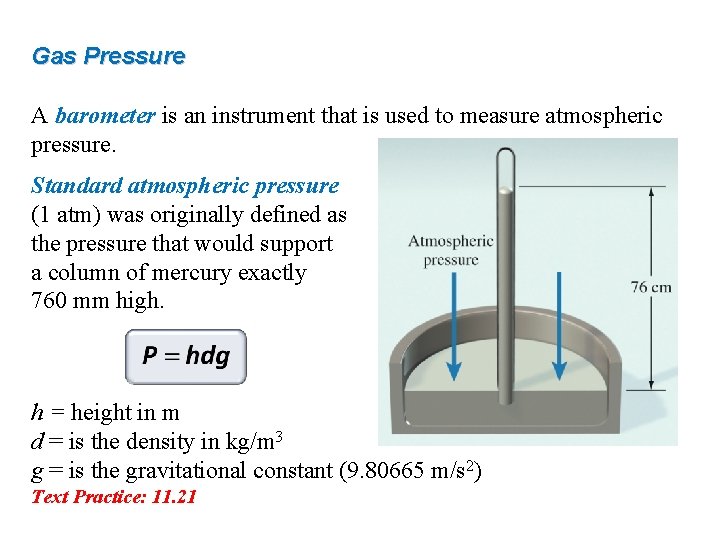
Gas Pressure A barometer is an instrument that is used to measure atmospheric pressure. Standard atmospheric pressure (1 atm) was originally defined as the pressure that would support a column of mercury exactly 760 mm high. h = height in m d = is the density in kg/m 3 g = is the gravitational constant (9. 80665 m/s 2) Text Practice: 11. 21
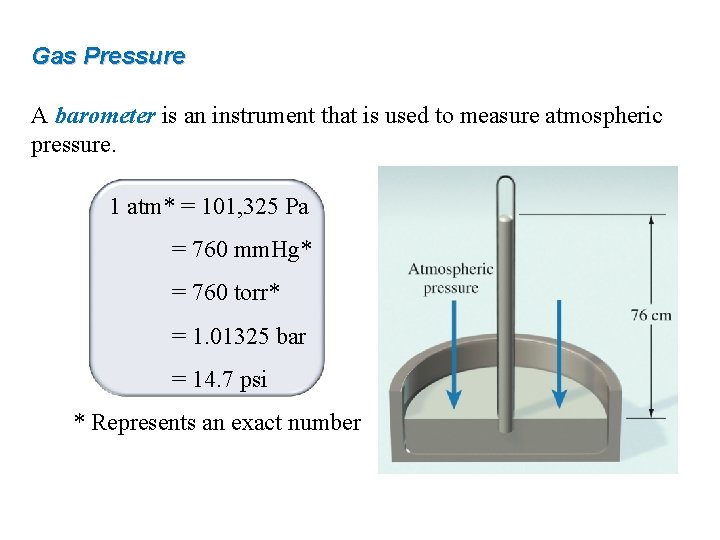
Gas Pressure A barometer is an instrument that is used to measure atmospheric pressure. 1 atm* = 101, 325 Pa = 760 mm. Hg* = 760 torr* = 1. 01325 bar = 14. 7 psi * Represents an exact number
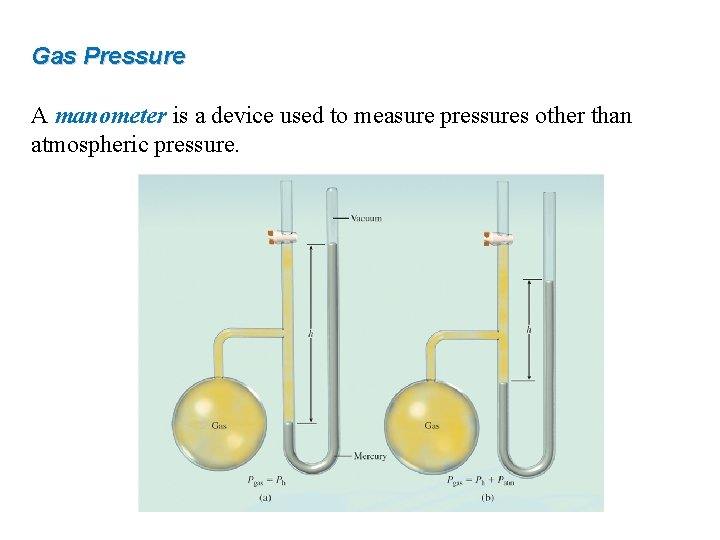
Gas Pressure A manometer is a device used to measure pressures other than atmospheric pressure.

11. 4 The Gas Laws • You are responsible for investigating the individual gas laws and for mastering all the conceptual and quantitative skills associated with them. • The Ideal Gas Law can be more universally applied to solving problems involving gasses, so we will focus our lecture time on that. • The properties of a gas and its quantitative behavior depend partially on the number of gas particles present. A mole is the unit we use to measure how many particles we have when we have A LOT of particles.

Study Guide for Sections 11. 1 -11. 2, 5. 8, 11. 3 DAY 16, Terms to know: Sections 11. 1 -11. 2, 5. 8, 11. 3 Kinetic molecular theory (KMT), root-mean-square speed, molecular mass or molecular weight, formula mass or formula weight, pressure, diffusion, effusion, Graham’s Law, barometer, standard atmospheric pressure, manometer DAY 16, Specific outcomes and skills that may be tested on exam 2: Sections 11. 1 -11. 2, 5. 8, 11. 3 • Be able to outline the key tenants of the kinetic molecular theory (KMT) of gasses, and be able to explain how each aspect of the KMT correlates with specific observations about the physical properties of gasses • Given a molecular or structural formula, be able to determine the formula or molecular mass • Given masses and temperatures of gasses, be able to determine which gas molecules have a greater average kinetic energy and which have a greater average root-mean-square speed • Be able to rank the relative rates of effusion or diffusion for given gasses at specific temperatures • Be able to explain how a measurement of length (mm. Hg) can represent a measurement of pressure for a gas • Be able to explain how both barometers and manometers are used to measure pressures for gasses

Extra Practice Problems for Sections 11. 1 -11. 2, 5. 8, 11. 3 Complete these problems outside of class until you are confident you have learned the SKILLS in this section outlined on the study guide and we will review some of them next class period. 5. 67 5. 69 5. 71 11. 15 11. 23 (just to atmospheres) 11. 141 11. 159

Prep for Day 17 Must Watch videos: https: //www. youtube. com/watch? v=2 P--f. Rgd. SF 0 (Tyler De. Witt: moles vs. molecules) https: //www. youtube. com/watch? v=HMAOr. Gpk. Ts. Q (Tyler De. Witt: Avogadro's number in conversions) https: //www. youtube. com/watch? v=CMnk. Sb 2 Ys. XI (Tyler De. Witt: molar mass in conversions) https: //www. youtube. com/playlist? list=PLXGY 0 o. S 1 Ix 4_D 79 j 7 a. Tv 0 w. Uq. S 3 SM 51 FKB (gas laws playlist, Tyler De. Witt) https: //www. youtube. com/watch? v=Bx. US 1 K 7 xu 30 (ideal gas, crash course chemistry) Other helpful videos: http: //www. learnerstv. com/video/Free-video-Lecture-29332 -Chemistry. htm (moles in conversions) https: //www. youtube. com/watch? v=Qflq 48 Foh 2 w (Tyler De. Witt: determining molar mass) https: //www. youtube. com/watch? v=0 RXB 8 x. Nm. JNM (Tyler De. Witt: molar mass in conversions part 2) https: //www. youtube. com/watch? v=8 SRAk. XMu 3 d 0 (ideal gas calculations, crash course chemistry) Read Sections 2. 7, 5. 10, 11. 4 -11. 5 and work through the simulations here: http: //mw. concord. org/modeler/showcase/simulation. html? s=http: //mw 2. concord. org/public/student/ gaslaws/index. html We will not be lecturing about section 11. 4 unless you have questions about the reading, learnsmart, or simulation, so analyze this section carefully!
- Slides: 23