Chemistry Atoms First Julia Burdge Jason Overby Chapter

Chemistry: Atoms First Julia Burdge & Jason Overby Chapter 15 Chemical Equilibrium Kent L. Mc. Corkle Cosumnes River College Sacramento, CA Copyright (c) The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display.

15 Chemical Equilibrium 15. 1 The Concept of Equilibrium 15. 2 The Equilibrium Constant Calculating Equilibrium Constants Magnitude of the Equilibrium Constant 15. 3 Equilibrium Expressions Heterogeneous Equilibria Manipulating Equilibrium Expressions Gaseous Equilibria 15. 4 Using Equilibrium Expressions to Solve Problems Predicting the Direction of a Reaction Calculating Equilibrium Concentrations 15. 5 Factors that Affect Chemical Equilibrium Addition or Removal of a Substance Changes in Volume and Pressure Changes in Temperature Catalysis

15. 1 The Concept of Equilibrium The decomposition of N 2 O 4 is a reversible process, meaning the products of the reaction can react to form reactants. N 2 O 4(g) ⇌ 2 NO 2(g) The system is in equilibrium when the rates of the forward reaction and the reverse reaction are the same. rate forward = kf[N 2 O 4] and rate reverse = kr[NO 2]2
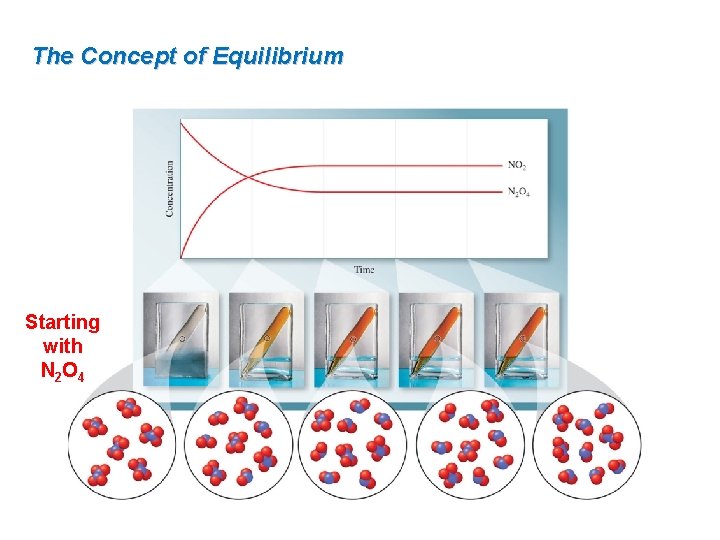
The Concept of Equilibrium Starting with N 2 O 4
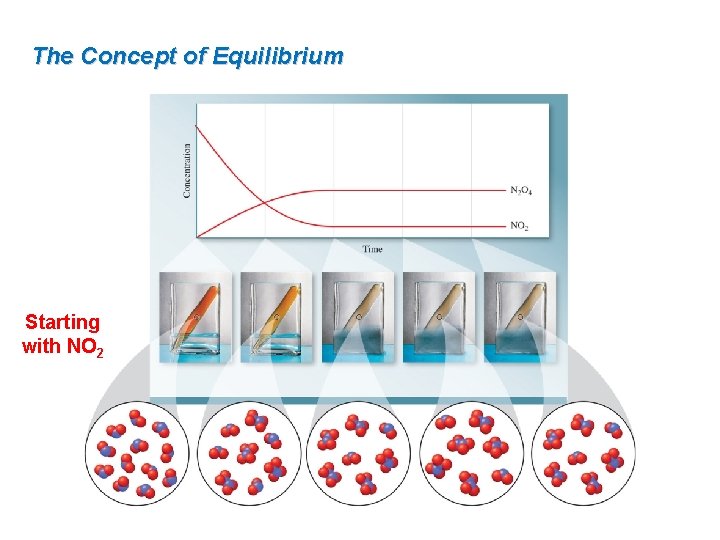
The Concept of Equilibrium Starting with NO 2

The Concept of Equilibrium Some important things to remember about equilibrium are: Equilibrium is a dynamic state—both the forward and reverse reactions continue to occur, although there is no net change in reactant and product concentration over time. At equilibrium, the rates of the forward and reverse reactions are equal. Equilibrium can be established starting with only reactants, with only products, or with any mixture of reactants and products.

15. 2 The Equilibrium Constant N 2 O 4(g) ⇌ 2 NO 2(g) rate forward = rate reverse kf[N 2 O 4] eq = kr[NO 2]2 eq The subscript “eq” denotes a concentration at equilibrium. Rearranging The ratio of two constants (kf/kr) is also a constant: equilibrium expression equilibrium constant
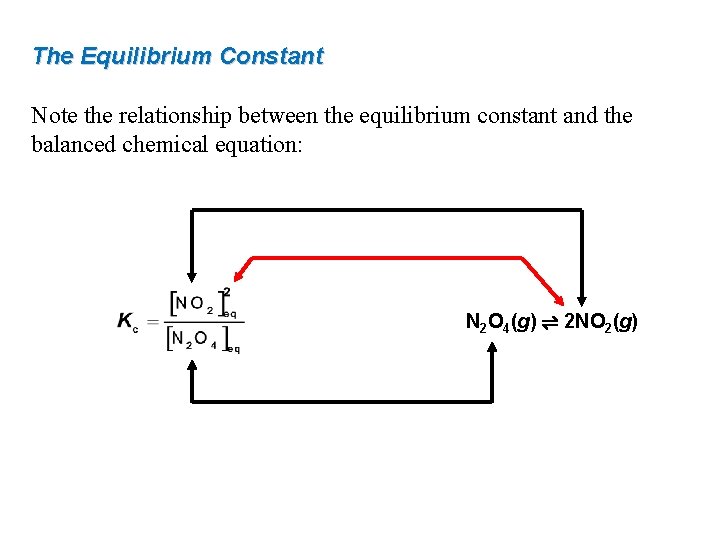
The Equilibrium Constant Note the relationship between the equilibrium constant and the balanced chemical equation: N 2 O 4(g) ⇌ 2 NO 2(g)

The Equilibrium Constant The reaction quotient (Qc ) is a fraction with product concentrations in the numerator and reactant concentrations in the denominator. Each concentration is raised to a power equal to the corresponding stoichiometric coefficient in the balanced chemical equation. a. A + b. B ⇌ c. C + d. D (at equilibrium) This law of mass action applies to not only elementary reactions, but also to more complex reactions.

Worked Example 15. 1 Carbonyl chloride (COCl 2), also called phosgene, is a highly poisonous gas that was used on the battlefield of World War I. It is produced by the reaction of carbon monoxide with chlorine gas: CO(g) + Cl 2(g) ⇌ COCl 2(g) In an experiment conducted at 74°C, the equilibrium concentrations of the species -2 M, [Cl ] = 0. 054 M, and involved in the. About reaction follows: = 1. 2× 10 concentrations 2 Think It were Whenasputting the[CO] equilibrium into [COClthe 0. 14 M. (a) expression, Write the equilibrium expression, determine the we leave out the units. and It is(b) common 2] =equilibrium practice to expressconstant equilibrium constants without units. value of the equilibrium for this reaction at 74°C. Strategy Use the law of mass action to write the equilibrium expression and plug in the equilibrium concentrations of all three species to evaluate Kc. [COCl 2] [CO][Cl 2] (0. 14) (b) Kc = = 216 or 2. 2× 102 -2 (1. 2× 10 )(0. 054) Solution (a) Kc =

Worked Example 15. 2 Write reaction quotients for the following reactions: (a) N 2(g) + 3 H 2(g) ⇌ 2 NH 3(g) (b) H 2(g) + I 2(g) ⇌ 2 HI(g) (c) Ag+(aq) + 2 NH 3(aq) ⇌ Ag(NH 3)2+(aq) (d) 2 OThink About 3(g) ⇌ 3 O 2(g) It With practice, writing reaction quotients becomes second Without practice, it will seem inordinately 2 -(aq) (e) Cd 2+ (aq) +nature. 4 Br-(aq) ⇌ Cd. Brsufficient 4 difficult. It is important that you become proficient at this. It is very (f) 2 NO(g) O 2 first (g) ⇌step 2 NOin 2(g) often+the solving equilibrium problems. Strategy Use the law of mass action to write reaction quotients. [HI]2 [NH 3]2 [Ag(NH 3)2+] Solution (a) Qc = (b) Qc = (c) Qc = [H 2][I 2] [N 2][H 2]3 [Ag+][NH 3]2 [O 2]3 [Cd. Br 42 -] [NO 2]2 (d) Qc = (e) Qc = (f) Qc = [O 3]2 [Cd 2+][Br-]4 [NO]2[O 2]

The Equilibrium Constant At any point during the progress of a reaction: a. A + b. B ⇌ c. C + d. D
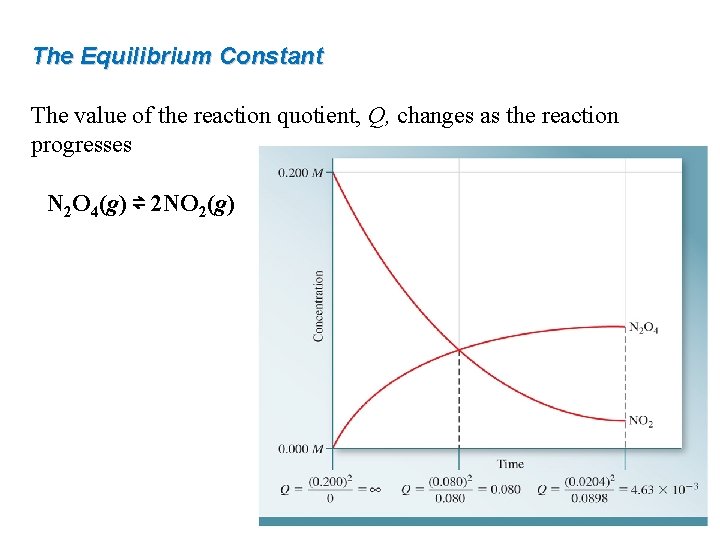
The Equilibrium Constant The value of the reaction quotient, Q, changes as the reaction progresses N 2 O 4(g) ⇌ 2 NO 2(g)

The Equilibrium Constant The equilibrium constant gives the extent a reaction will proceed at a particular temperature. Three outcomes are possible: 1) The reaction will go essentially to completion and the equilibrium mixture will consist predominately of products. Ag+(aq) + 2 NH 3(aq) ⇌ Ag(NH 3)2+(g) Kc = 1. 5 x 107 (at 25°C) Large Kc, product favored

The Equilibrium Constant The equilibrium constant gives the extent a reaction will proceed at a particular temperature. Three outcomes are possible: 2) The reaction will not occur to any significant degree, and the equilibrium mixture will consist predominantly of reactant. N 2(g) + O 2(g) ⇌ 2 NO(g) Kc = 4. 3 x 10– 25 (at 25°C) Small Kc, reactant favored 3) The reaction will proceed a significant degree but will not go to completion, and the equilibrium mixture will contain comparable amounts of both reactants and products.

15. 3 Equilibrium Expressions When the species in a reversible chemical reaction are not all in the same phase, the equilibrium is heterogeneous. Only gaseous species and aqueous species appear in equilibrium expressions, pure solids and pure liquids do not. CO 2(g) + C(s) ⇌ 2 CO(g) 2 Fe(s) + 3 H 2 O(l) ⇌ Fe 2 O 3(s) + 2 H 2(g)

Worked Example 15. 3 Write equilibrium expressions for each of the following reactions: (a) Ca. CO 3(s) ⇌ Ca. O(s) + CO 2(g) (b) Hg(l) + Hg 2+(aq) ⇌ Hg 22+(aq) (c) 2 Fe(s) + 3 H + 2 H 2 equilibrium (g) 2 O(l) ⇌It. Fe. Like 2 O 3(s) Think About writing expressions for homogeneous equilibria, writing equilibrium expressions for (d) O 2(g) + 2 H 2(g) ⇌ 2 H 2 O(l) heterogeneous equilibria becomes second nature if you practice. The importance developing this skill now cannot be overstated. Your for Strategy Use theoflaw of mass action to write the equilibrium expressions ability to. Only understand theaqueous principles and toappear solve in many of the each reaction. gases and species the expression. problems in and Chapters 16 to 19 depends 2+on your ability to write [Hg 2 ] Solution (a) Kc =expressions [CO 2] equilibrium correctly (b) Kc and = easily. [Hg 2+] (c) Kc = [H 2 ]2 (d) Kc = 1 [O 2][H 2]2

Equilibrium Expressions When a reversible chemical equation is manipulated, it is also necessary to make appropriate changes in the equilibrium expression and the equilibrium constant.

Worked Example 15. 4 The following reactions have the indicated equilibrium constants at 100°C: (1) 2 NOBr(g) ⇌ 2 NO(g) + Br 2(g) (2) Br 2(g) + Cl 2(g) ⇌ 2 Br. Cl(g) Kc = 0. 014 Kc = 7. 2 Determine the value of Kc for the following reactions at 100°C: (a) 2 NO(g) + Br 2(g) ⇌ 2 NOBr(g) (d) 2 NOBr(g) + Cl 2(g) ⇌ 2 NO(g) + 2 Br. Cl(g) 1 (b) 4 NOBr(g) ⇌ 4 NO(g) + 2 Br 2(g) (e) NO(g) + Br. Cl(g) ⇌ NOBr(g) + Cl 2(g) 2 (c) NOBr(g) ⇌ NO(g) + 1 Br (g) 2 2 Strategy Begin by writing the equilibrium expressions for the reactions that are given. Then, determine the relationship of each equation’s equilibrium expression to the equilibrium expression of the original equations, and make the corresponding change to the equilibrium constant for each. [NO]2[Br 2] [Br. Cl]2 Kc = and Kc = [NOBr]2 [Br 2][Cl 2]

Worked Example 15. 4 (cont. ) Solution (a) This equation is the reverse of original equation 1. Its equilibrium expression is the reciprocal of that for the original equation: [NOBr]2 Kc = = 1/0. 014 = 71 [NO]2[Br 2] (b) This is original equation 1 multiplied by a factor of 2. Its equilibrium expression is the original expression squared: [NO]2[Br 2] Kc = [NOBr]2 2 = (0. 014)2 = 2. 0× 10 -4 (c) This is the original equation 1 multiplied by the square root of the original Kc = or 1 2 . Its equilibrium expression is [NO]2[Br 2] Kc = [NOBr]2 1/2 = (0. 014)1/2 = 0. 12

Worked Example 15. 4 (cont. ) Solution (d) This is the sum of the original equations 1 and 2. Its equilibrium expression is the product of the two individual expressions: [NO]2[Br. Cl]2 Kc = = (0. 014)(7. 2) = 0. 10 [NOBr]2[Cl 2] (e) Probably the simplest way to analyze this reaction is to recognize that it is the 1 reverse of the reaction in part (d), multiplied by 2. Its equilibrium expression is the square root of the reciprocal of the expression in part (d): [NO]2[Br. Cl]2 Kc = [NOBr]2[Cl 2] 1/2 = (1/0. 10)1/2 = 3. 2

Worked Example 15. 4 (cont. ) Think About It The magnitude of an equilibrium constant reveals whether products or reactants are favored, so the reciprocal relationship between Kc values of forward and reverse reactions should make sense. A very large Kc value means that products are favored. In the reaction of hydrogen ion and hydroxide ion to form water, the value of Kc is very large, indicating that the product, water, is favored. H+(aq) + OH-(aq) ⇌ H 2 O(l) Kc = 1. 0× 1014 (at 25°C) Simply writing the equation backward doesn’t change the fact that water is the predominate species. In the reverse reaction, therefore, the favored species is on the reactant side: H 2 O(l) ⇌ H+(aq) + OH-(aq) Kc = 1. 0× 10 -14 (at 25°C) As a result, the magnitude of Kc should correspond to reactants being favored; that is, it should be very small.

Gaseous Equilibria When an equilibrium expression contains only gases, we can write an alternate form of the expression in which the concentrations of gases are expressed as partial pressures (atm). Thus, for the equilibrium N 2 O 4(g) ⇌ 2 NO 2(g) we can write as Kc = [NO 2]2 [N 2 O 4] or KP = (PNO 2)2 PN 2 O 4 The relationship between Kc and KP can be expressed as KP = Kc[(0. 08206 L∙atm/K∙mol)×T]Δn where Δn = moles of gaseous products – moles of gaseous reactants.

Worked Example 15. 5 Write KP expressions for (a) PCl 3(g) + Cl 2(g) ⇌ PCl 5(g), (b) O 2(g) + 2 H 2(g) ⇌ 2 H 2 O(l), and (c) F 2(g) + H 2(g) ⇌ 2 HF(g). Strategy Write equilibrium expressions for each equation, expressing the concentrations of the gases in partial pressures. Solution (a) All the species in this equation are gases, so they will appear in the KP expression. (PPCl 5) KP = (PPCl 3)(PCl 2) 1 (b) Only the reactants are gases. KP = (PO 2)(PH 2)2 (PHF)2 (c) All species are gases. KP = (PF 2)(PH 2) Think About It It isn’t necessary for every species in the reaction to be a gas– only those species that appear in the equilibrium expression.

Worked Example 15. 6 The equilibrium constant, Kc, for the reaction N 2 O 4(g) ⇌ 2 NO 2(g) is 4. 63× 10 -3 at 25°C. What is the value of KP at this temperature. Strategy Use KP = Kc[(0. 08206 L∙atm/K∙mol)×T]Δn. Be sure to convert temperature in degrees Celsius to kelvins. Using Δn = moles of gaseous products – moles of gaseous reactants, Δn = 2(NO 2) – 1(N 2 O 4) = 1. T = 298 K. Think About It Note that we have essentially disregarded the units Solution of R and T so that the resulting equilibrium constant, KP, is unitless. Equilibrium constants commonly are treated as unitless quantities. 0. 08206 L∙atm KP = Kc ×T K∙mol = (4. 63× 10 -3)(0. 08206 × 298) = 0. 113

15. 4 Using Equilibrium Expressions to Solve Problems The equilibrium expression may be used to predict the direction of a reaction and to calculate equilibrium concentrations. Predictions are made based on comparisons between Qc and Kc. There are three possibilities: 1) Q < K The ratio of initial concentrations of products to reactants is too small. To reach equilibrium, reactants must be converted to products. The system proceeds in the forward direction. 2) Q = K The initial concentrations are equilibrium concentrations. The system is at equilibrium. 3) Q > K The ratio of initial concentrations of products to reactants is too large. To reach equilibrium products must be converted to reactants. The system proceeds in the reverse direction.

Worked Example 15. 7 At 375°C, the equilibrium constant for the reaction N 2(g) + 3 H 2(g) ⇌ 2 NH 3(g) is 1. 2. At the state of a reaction, the concentrations of N 2, H 2, and NH 3 are 0. 071 M, 9. 2× 10 -3 M, and 1. 83× 10 -4 M, respectively. Determine whether the system is at equilibrium, and if not, determine in which direction it must proceed Think About It In proceeding to the right, a reaction consumes to establish equilibrium. reactants and produces more products. This increases the numerator in the. Use reaction quotient and decreases the denominator. The compare result is Qc Strategy the initial concentrations to calculate Qc, and then increase in Qc until it is equal to Kc, at which point equilibrium with Kan c. will be established. 2 [NH 3]i (1. 83× 10 -4)2 Qc = = = 0. 61 [N 2]i[H 2]i 3 (0. 071)(9. 2× 10 -3)3 Strategy The calculated value of Qc is less than Kc. Therefore, the reaction is not at equilibrium and must proceed to the right to establish equilibrium.

Calculating Equilibrium Concentrations Equilibrium concentrations can be calculated from initial concentrations if the equilibrium constant is known. Kc = 24. 0 (200°C) cis-Stilbene trans-Stilbene cis-Stilbene Initial concentration (M) ⇌ trans-Stilbene 0. 850 0 Change in concentration (M) –x +x Equilibrium concentration (M) 0. 850 – x x

Using Equilibrium Expressions to Solve Problems cis-Stilbene Initial concentration (M) ⇌ trans-Stilbene 0. 850 0 Change in concentration (M) –x +x Equilibrium concentration (M) 0. 850 – x x Use the equilibrium concentrations, defined in terms of x, in the equilibrium expression: x = 0. 816 M (Represents the change in initial concentrations)

Using Equilibrium Expressions to Solve Problems cis-Stilbene ⇌ trans-Stilbene Initial concentration (M) 0. 850 0 Change in concentration (M) – 0. 816 +0. 816 Equilibrium concentration (M) 0. 850 – 0. 816 Calculate the equilibrium concentrations of cis- and trans-stilbene: [cis-stilbene] = (0. 850 – x) M = (0. 850 – 0. 816) M = 0. 034 M [trans-stilbene] = x M = 0. 816 M

Worked Example 15. 8 Kc for the reaction of hydrogen and iodine to produce hydrogen iodide, H 2(g) + I 2(g) ⇌ 2 HI(g) is 54. 3 at 430°C. What will the concentrations be at equilibrium if we start with 0. 240 M concentrations of both H 2 and I 2? Strategy Insert the starting concentrations that we know into the equilibrium table: + ⇌ H 2 I 2 2 HI Initial concentration (M) Change in concentration (M) Equilibrium concentration (M) 0. 240 0
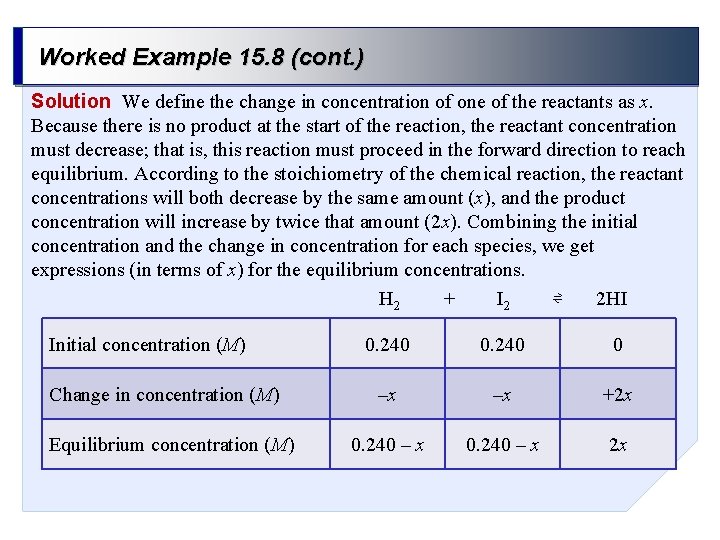
Worked Example 15. 8 (cont. ) Solution We define the change in concentration of one of the reactants as x. Because there is no product at the start of the reaction, the reactant concentration must decrease; that is, this reaction must proceed in the forward direction to reach equilibrium. According to the stoichiometry of the chemical reaction, the reactant concentrations will both decrease by the same amount (x), and the product concentration will increase by twice that amount (2 x). Combining the initial concentration and the change in concentration for each species, we get expressions (in terms of x) for the equilibrium concentrations. + ⇌ H 2 I 2 2 HI Initial concentration (M) Change in concentration (M) Equilibrium concentration (M) 0. 240 0 –x –x +2 x 0. 240 – x 2 x

Worked Example 15. 8 (cont. ) Solution Next, we insert these expressions for the equilibrium concentrations into the equilibrium expression and solve for x. [HI]2 Kc = [H 2][I 2] (2 x)2 54. 3 = = Think(0. 240 About– It Always check (0. 240 your answer x)(0. 240 – x)2 by inserting the calculated concentrations into the equilibrium expression: 2 x 2 2 = [HI] (0. 378) 0. 240 – x = 54. 9 ≈ Kc [H 2][I 2] = (0. 051)2 x= 0. 189 The small difference between the calculated Kc and the one given in the problem statement is due to rounding. Use the calculated value of x, we can determine the equilibrium concentration of each species as follows: [H 2] = (0. 240 – x) M = 0. 051 M [I 2] = (0. 240 – x) M = 0. 051 M [HI] = 2 x = 0. 378 M

Worked Example 15. 9 For the same reaction and temperature as in Worked Example 15. 8, calculate the equilibrium concentrations of all three species if the starting concentrations are as follows: [H 2] = 0. 00623 M, [I 2] = 0. 00414 M, [HI] = 0. 0424 M. Strategy Using the initial concentrations, calculate the reaction quotient, Qc, and compare it to the value of Kc (given in the problem statement of Worked Example 15. 8) to determine which direction the reaction will proceed to establish equilibrium. Then, construct an equilibrium table to determine the equilibrium concentrations. [HI]2 (0. 0424)2 = = 69. 7 [H 2][I 2] (0. 00623)(0. 00414)
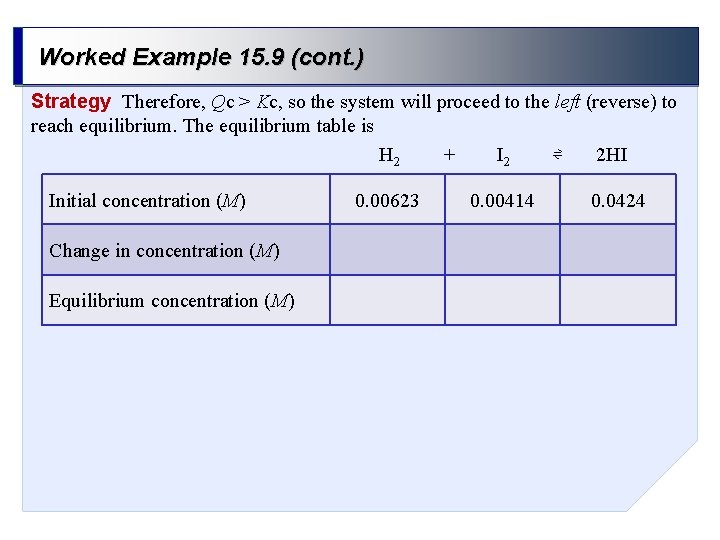
Worked Example 15. 9 (cont. ) Strategy Therefore, Qc > Kc, so the system will proceed to the left (reverse) to reach equilibrium. The equilibrium table is + ⇌ H 2 I 2 2 HI Initial concentration (M) Change in concentration (M) Equilibrium concentration (M) 0. 00623 0. 00414 0. 0424
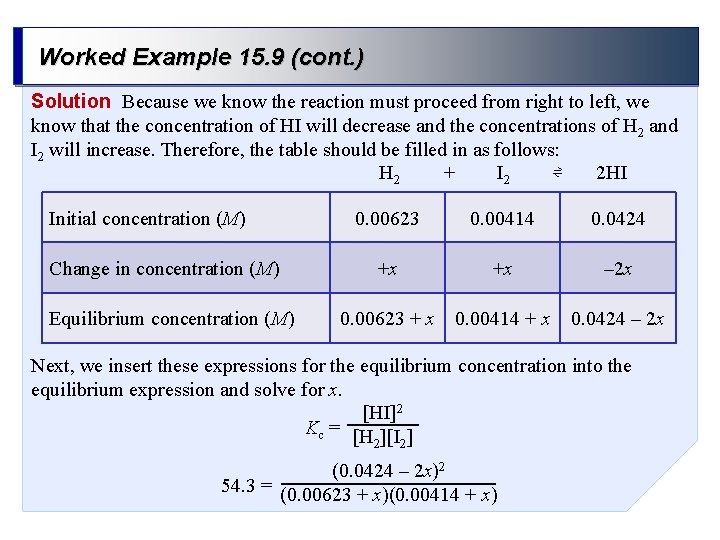
Worked Example 15. 9 (cont. ) Solution Because we know the reaction must proceed from right to left, we know that the concentration of HI will decrease and the concentrations of H 2 and I 2 will increase. Therefore, the table should be filled in as follows: + ⇌ H 2 I 2 2 HI Initial concentration (M) Change in concentration (M) Equilibrium concentration (M) 0. 00623 0. 00414 0. 0424 +x +x – 2 x 0. 00623 + x 0. 00414 + x 0. 0424 – 2 x Next, we insert these expressions for the equilibrium concentration into the equilibrium expression and solve for x. [HI]2 Kc = [H ][I ] 2 2 (0. 0424 – 2 x)2 54. 3 = (0. 00623 + x)(0. 00414 + x)

Worked Example 15. 9 (cont. ) Solution It isn’t possible to solve this equation the way we did in Worked Example 15. 8 (by taking the square root of both sides) because the concentrations of H 2 and I 2 are unequal. Instead, we have to carry out the multiplications. 54. 3(2. 58× 10 -5 + 1. 04× 10 -2 x + x 2) = 1. 80× 10 -3 – 1. 70× 10 -1 x + 4 x 2 Collecting terms we get 50. 3 x 2 + 0. 735 x – 4. 00× 10 -4 = 0 This is a quadratic of the form ax 2 + bx + c = 0. The solution for the quadratic equation [Appendix 1] is x= Here we have a = 50. 3, b = 0. 735, and c = -4. 00× 10 -4, so x=

Worked Example 15. 9 (cont. ) Solution x = 5. 25× 10 -4 or x = – 0. 0151 Only the first of these values, 5. 25× 10 -4, makes sense because concentration cannot be a negative number. Using the calculated value of x, we can determine the equilibrium concentration of each species as follows: [H 2] = (0. 00623 + x) M = 0. 00676 M [I 2] = (0. 00414 + x) M = 0. 00467 M [HI] = (0. 0424 – 2 x) M = 0. 0414 M Think About It Checking this result gives [HI]2 (0. 0414)2 Kc = = = 54. 3 [H 2][I 2] (0. 00676)(0. 00467)
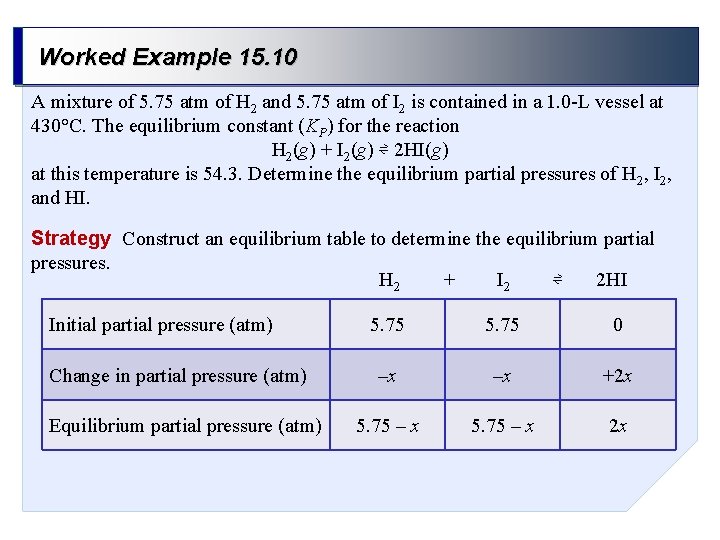
Worked Example 15. 10 A mixture of 5. 75 atm of H 2 and 5. 75 atm of I 2 is contained in a 1. 0 -L vessel at 430°C. The equilibrium constant (KP) for the reaction H 2(g) + I 2(g) ⇌ 2 HI(g) at this temperature is 54. 3. Determine the equilibrium partial pressures of H 2, I 2, and HI. Strategy Construct an equilibrium table to determine the equilibrium partial pressures. + ⇌ H 2 I 2 2 HI Initial partial pressure (atm) Change in partial pressure (atm) Equilibrium partial pressure (atm) 5. 75 0 –x –x +2 x 5. 75 – x 2 x

Worked Example 15. 10 (cont. ) Solution Setting the equilibrium expression equal to KP, (2 x)2 54. 3 = (5. 75 – x)2 Taking the square root of both sides of the equation gives 2 x = It Plugging the calculated partial pressures into the Think About 5. 75 – x equilibrium expression 2 x gives 7. 369 = (P–HIx)2 (9. 04)2 5. 75 = (1. 23)2 = 54. 0 7. 369(5. 75 – x) = 2 x(PH 2)(PI 2) The–small difference between this result and the equilibrium constant 42. 37 7. 369 x = 2 x given in the problem is due to rounding. 42. 37 = 9. 369 x x = 4. 52 The equilibrium partial pressures are PH 2 = PI 2 = 5. 75 – 4. 52 = 1. 23 atm, and PHI = 9. 04 atm.

15. 5 Factors That Affect Chemical Equilibrium Le Châtelier’s principle states that when a stress is applied to a system at equilibrium, the system, will respond by shifting in the direction that minimizes the effect of the stress. Stress refers to any of the following: Ø The addition of a reactant or product Ø The removal of a reactant or product Ø A change in volume of the system, resulting in a change in concentration or partial pressure of the reactants and products Ø A change in temperature

Factors That Affect Chemical Equilibrium Consider the Haber process at 700 K: N 2(g) + 3 H 2(g) ⇌ 2 NH 3(g) At equilibrium: [N 2] = 2. 05 M [H 2] = 1. 56 M [NH 3] = 1. 52 M Applying stress by the addition of N 2 to give the following concentrations: [N 2] = 3. 51 M [H 2] = 1. 56 M [NH 3] = 1. 52 M The reaction shifts to the right. N 2(g) + 3 H 2(g) ⇌ 2 NH 3(g)
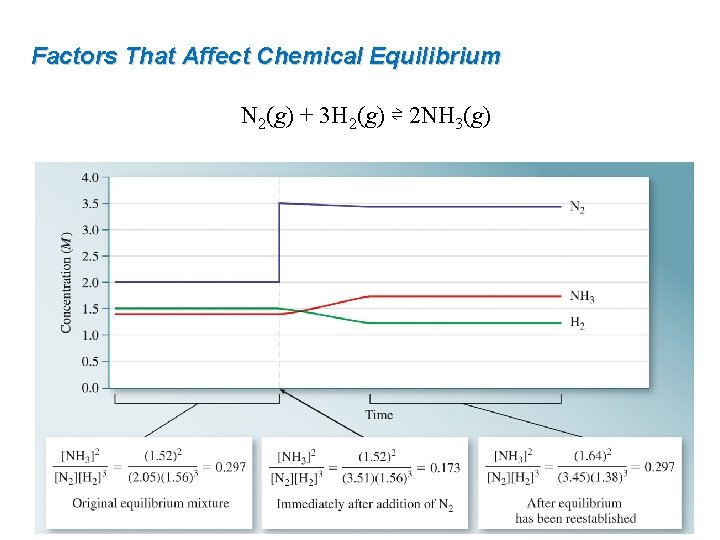
Factors That Affect Chemical Equilibrium N 2(g) + 3 H 2(g) ⇌ 2 NH 3(g)
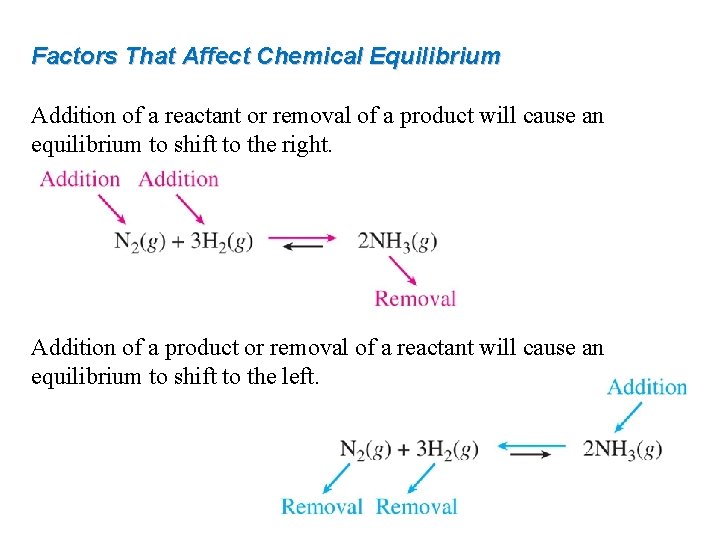
Factors That Affect Chemical Equilibrium Addition of a reactant or removal of a product will cause an equilibrium to shift to the right. Addition of a product or removal of a reactant will cause an equilibrium to shift to the left.

Worked Example 15. 11 Hydrogen sulfide (H 2 S) is a contaminant commonly found in natural gas. It is removed by reaction with oxygen to produce elemental sulfur. 2 H 2 S(g) + O 2(g) ⇌ 2 S(s) + 2 H 2 O(g) For each of the following scenarios, determine whether the equilibrium will shift to the right, shift to the left, or neither: (a) addition of O 2(g), (b) removal of H 2 S(g), (c) removal of H 2 O(g), and (d) addition of S(s). Strategy Use Le Châtelier’s principle to predict the direction of shift in each case. Remember that the position of the equilibrium is only changed by the addition or removal of a species that appears in the reaction quotient expression. [H 2 O]2 Qc = [H 2 S][O 2] Because sulfur is a solid, it does not appear in the expression.
![Worked Example 15. 11 (cont. ) Solution [H 2 O]2 Qc = [H 2 Worked Example 15. 11 (cont. ) Solution [H 2 O]2 Qc = [H 2](http://slidetodoc.com/presentation_image_h2/f4e001bbf817ff25124156ae0f25cb34/image-46.jpg)
Worked Example 15. 11 (cont. ) Solution [H 2 O]2 Qc = [H 2 S][O 2] Changes in concentration of any of the other species will cause a change in the equilibrium a reactant or the removal productwill that appears Thinkposition. About Addition It In eachofcase, analyze effectof thea change in the expression shift equilibrium to the right: have on the. Qvalue Qc. the In part (a), for example, O 2 is added, so its c willof addition increases. addition. Looking at the reaction quotient expression, concentration + O 2(g) 2 S(s) + 2 H 2 corresponds O(g) we can see that 2 H a larger of oxygen to a 2 S(g)concentration larger overall denominator – giving the overall fraction a smaller removal Removal of a. Thus, reactant or addition of a be product that appears thereaction expression Qc value. Q will temporarily smaller K andinthe will shift equilibrium theright, left: consuming some of the added O 2 willthe have to shift totothe addition (along with some of the H 2 S in the mixture) to reestablish 2 S(s) + 2 H 2 O(g) equilibrium. 2 H 2 S(g) + O 2(g) removal (a) Shift to the right (d) No change removal (b) Shift to the left (c) Shift to the right
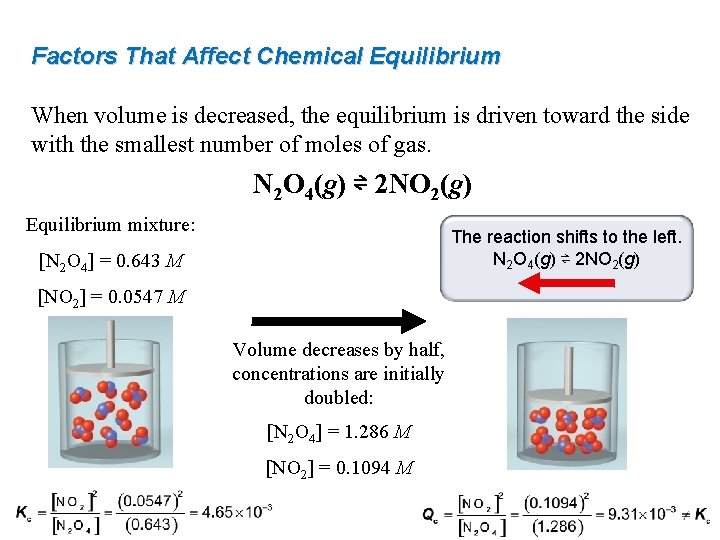
Factors That Affect Chemical Equilibrium When volume is decreased, the equilibrium is driven toward the side with the smallest number of moles of gas. N 2 O 4(g) ⇌ 2 NO 2(g) Equilibrium mixture: The reaction shifts to the left. N 2 O 4(g) ⇌ 2 NO 2(g) [N 2 O 4] = 0. 643 M [NO 2] = 0. 0547 M Volume decreases by half, concentrations are initially doubled: [N 2 O 4] = 1. 286 M [NO 2] = 0. 1094 M

Worked Example 15. 12 For each reaction, predict in what direction the equilibrium will shift when the volume of the reaction vessel is decreased. (a) PCl 5(g) ⇌ PCl 3(g) + Cl 2(g) (b) 2 Pb. S(s) + 3 O 2(g) ⇌ 2 Pb. O(s) + 2 SO 2(g) (c) H 2(g) + I 2(g) ⇌ 2 HI(g) Think About It When there is a difference in the number of moles of gas, changingwhich the volume of the reaction the vessel will change theof gas in Strategy Determine direction minimized number of moles concentrations of reactant(s) and product(s)–but the system will the reaction. Count only moles of gas. remain at equilibrium. (Q will remain equal to K. ) Solution (a) We have 1 mole of gas on the reactant side and 2 moles of gas on the product side, so it will shift to the left. (b) 3 moles of gas on the reactant side and 2 moles of gas on the product side, so it will shift to the right. (c) 2 moles of gas on each side, so no shift.
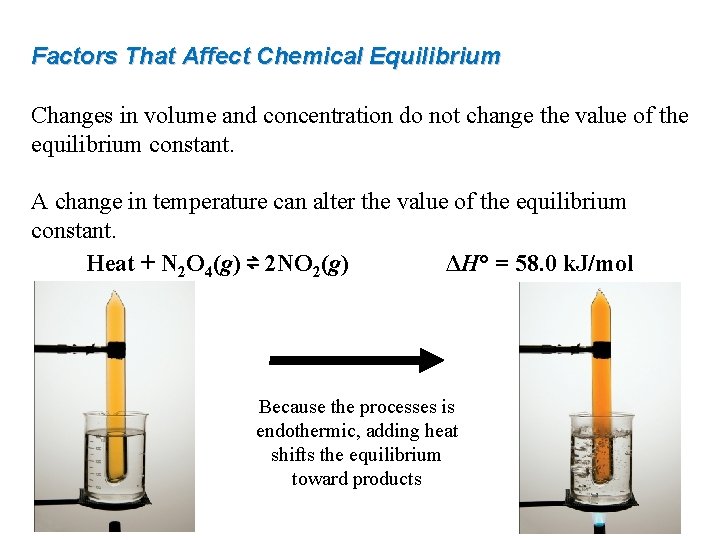
Factors That Affect Chemical Equilibrium Changes in volume and concentration do not change the value of the equilibrium constant. A change in temperature can alter the value of the equilibrium constant. Heat + N 2 O 4(g) ⇌ 2 NO 2(g) ΔH° = 58. 0 k. J/mol Because the processes is endothermic, adding heat shifts the equilibrium toward products

Factors That Affect Chemical Equilibrium For any endothermic reaction, heat is a reactant: heat + reactants ⇌ products ΔH° > 0 k. J/mol Adding heat shifts the reaction towards products, Kc increases Removing heat shifts the reaction towards reactants, Kc decreases. For any exothermic reaction, heat is a product: reactants ⇌ products + heat ΔH° < 0 k. J/mol Adding heat shifts the reaction towards reactants, Kc decreases Removing heat shifts the reaction towards products, Kc increases

Factors That Affect Chemical Equilibrium Co. Cl 42 - + 6 H 2 O ⇌ Co(H 2 O)62+ + 4 Cl- + heat blue pink
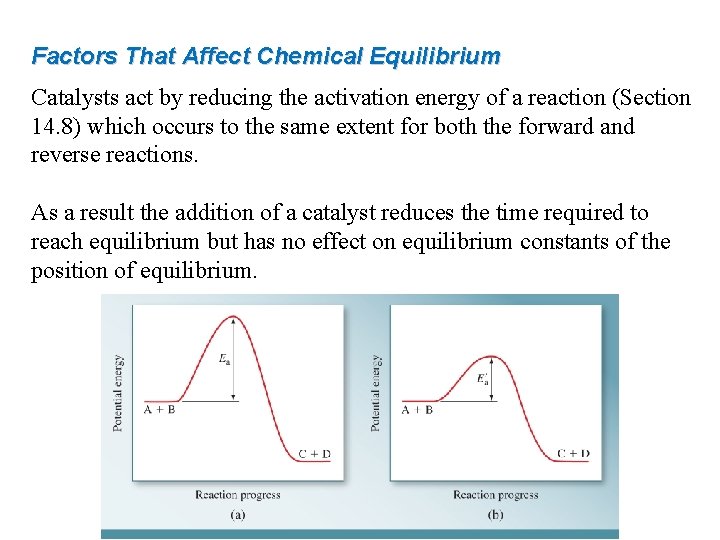
Factors That Affect Chemical Equilibrium Catalysts act by reducing the activation energy of a reaction (Section 14. 8) which occurs to the same extent for both the forward and reverse reactions. As a result the addition of a catalyst reduces the time required to reach equilibrium but has no effect on equilibrium constants of the position of equilibrium.
15 Key Points The Concept of Equilibrium The Equilibrium Constant Calculating Equilibrium Constants Magnitude of the Equilibrium Constant Equilibrium Expressions Heterogeneous Equilibria Manipulating Equilibrium Expressions Using Equilibrium Expressions to Solve Problems Predicting the Direction of a Reaction Calculating Equilibrium Concentrations Factors that Affect Chemical Equilibrium Addition or Removal of a Substance Changes in Volume and Pressure Changes in Temperature
- Slides: 53