chemistry 7 2 Ionic Bonds and Ionic Compounds

chemistry

7. 2 Ionic Bonds and Ionic Compounds n In many coastal countries that have warm, relatively dry climates, salt is produced by the evaporation of seawater. You will learn how cations and anions combine to form stable compounds such as sodium chloride.

7. 2 Formation of Ionic Compounds n What is the electrical charge of an ionic compound?

7. 2 Formation of Ionic Compounds n Compounds composed of cations and anions are called ionic compounds. n Although they are composed of ions, ionic compounds are electrically neutral.
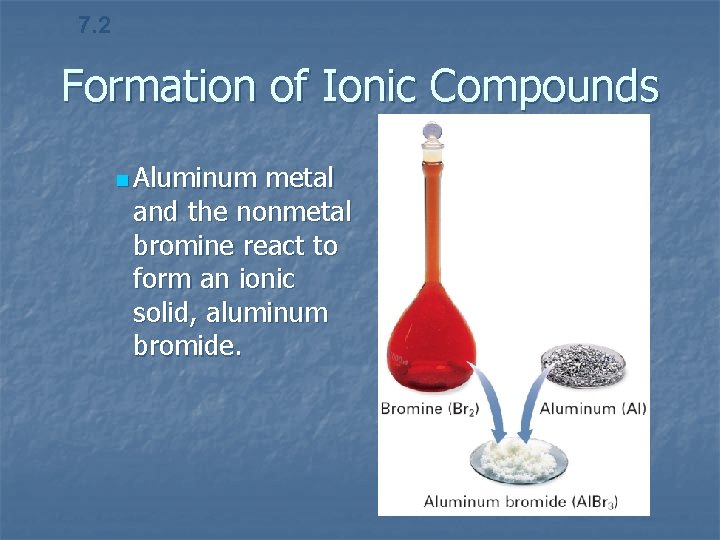
7. 2 Formation of Ionic Compounds n Aluminum metal and the nonmetal bromine react to form an ionic solid, aluminum bromide.

7. 2 Formation of Ionic Compounds n Ionic Bonds n The electrostatic forces that hold ions together in ionic compounds are called ionic bonds.

Formation of Ionic Compounds n Animation 8 Take an atomic-level look at the formation of KCl.

7. 2 Formation of Ionic Compounds n Formula Units n A chemical formula shows the kinds and numbers of atoms in the smallest representative unit of a substance. n. A formula unit is the lowest wholenumber ratio of ions in an ionic compound.
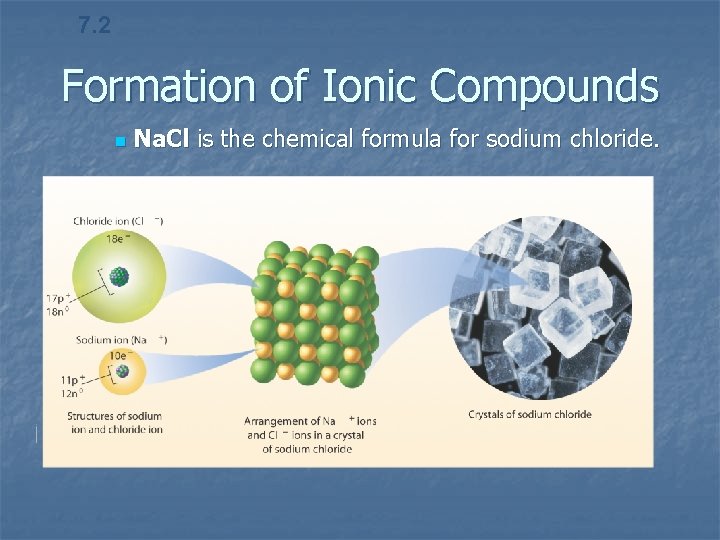
7. 2 Formation of Ionic Compounds n Na. Cl is the chemical formula for sodium chloride.

Conceptual Problem 7. 2

Conceptual Problem 7. 2

Conceptual Problem 7. 2

Conceptual Problem 7. 2

for Conceptual Problem 7. 2 Problem Solving 7. 12 Solve Problem 12 with the help of an interactive guided tutorial.

7. 2 Properties of Ionic Compounds n What are three properties of ionic compounds?

7. 2 Properties of Ionic Compounds n Most ionic compounds are crystalline solids at room temperature. n Ionic compounds generally have high melting points.

7. 2 Properties of Ionic Compounds n The orderly arrangement of component ions produces the beauty of crystalline solids.

7. 2 Properties of Ionic Compounds n Ionic compounds can conduct an electric current when melted or dissolved in water.

7. 2 Section Quiz. Assess students’ understanding of 7. 2. the concepts in Section Continue to: -or- Launch: Section Quiz Slide 19 of 25 © Copyright Pearson Prentice Hall

7. 2 Section Quiz. 1. Which chemical formula is incorrect? a. KF 2 b. Ca. S c. Mg. O d. Na. Br Slide 20 of 25 © Copyright Pearson Prentice Hall

7. 2 Section Quiz. 2. Ionic compounds can conduct an electric current a. only when melted. b. when melted or dissolved in water. c. only when dissolved in water. d. when solid or melted. Slide 21 of 25 © Copyright Pearson Prentice Hall

7. 2 Section Quiz. 3. At room temperature, most ionic compounds are a. crystalline solids. b. liquids. c. gases. d. soft, low melting-point solids. Slide 22 of 25 © Copyright Pearson Prentice Hall

END OF SHOW
- Slides: 23