CHEMISTRY 59 320 ANALYTICAL CHEMISTRY Fall 2010 Chapter













- Slides: 17

CHEMISTRY 59 -320 ANALYTICAL CHEMISTRY Fall - 2010 Chapter 8: Activity and the systematic treatment of equilibrium

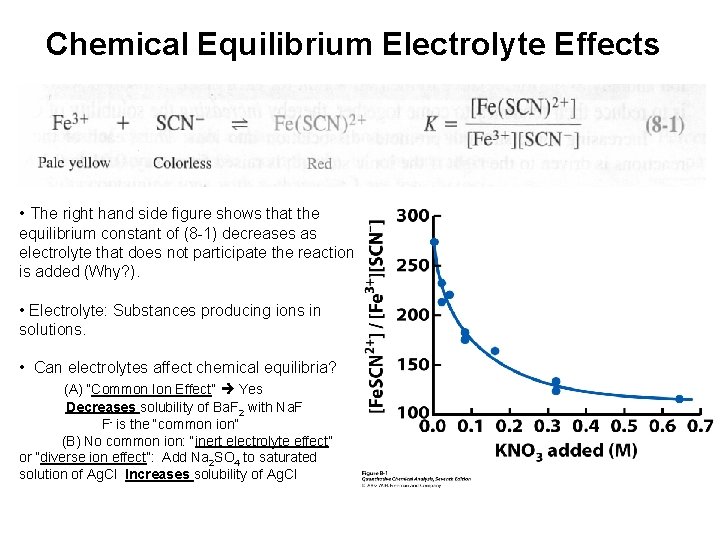
Chemical Equilibrium Electrolyte Effects • The right hand side figure shows that the equilibrium constant of (8 -1) decreases as electrolyte that does not participate the reaction is added (Why? ). • Electrolyte: Substances producing ions in solutions. • Can electrolytes affect chemical equilibria? (A) “Common Ion Effect” Yes Decreases solubility of Ba. F 2 with Na. F F- is the “common ion” (B) No common ion: “inert electrolyte effect” or “diverse ion effect”: Add Na 2 SO 4 to saturated solution of Ag. Cl Increases solubility of Ag. Cl

8 -1 The effect of ionic strength on solubility of salts • Why does the solubility increase when salts are added to the solution? • The formation of ionic atmosphere. The greater the ionic strength of a solution, the higher the charge in the ionic atmosphere. • The ionic atmosphere attenuates the attraction between ions since each ionplus-atmosphere contains less net charge. • Increasing ionic strength therefore reduces the attraction between any particular Ag+ and any Cl-, relative to the case in distilled water.

What is ionic strength? • Ionic strength, μ, is a measure of the total concentration of ions in solution. m = ½SCi. Zi 2 where Ci is the concentration of the ion, actually a ratio of Ci/1 M, Zi = charge on each individual ion. Example: Find the ionic strength of (a) 0. 10 M Na. Cl; (b) 0. 020 M KBr plus 0. 01 M Na 2 SO 4. Solution: (a) the soultion contains 0. 10 M Na+ and 0. 10 M Clm = ½[(0. 10 M/1 M)*(+1)2 + (0. 10 M/1 M)*(-1)2] = ½ [0. 10 + 0. 10] = 0. 10 (b) the solution contains 0. 020 M K+, 0. 020 M Br-, 0. 02 M Na+ and 0. 01 M SO 42 -. m = ½[(0. 020 M/1 M)*(+1)2 + (0. 020 M/1 M)*(-1)2 + (0. 020 M/1 M)*(+1)2 + (0. 010 M/1 M)*(-2)2 = ½ [0. 020 + 0. 040] = 0. 050. Ions with a larger charge number have greater contribution on the ionic strength.

8 -2 Activity Coefficients • To account for the effect of ionic strength, concentrations in the calculation of equilibrium constant show be replaced by activities: ẲC = [C]*γC • The activity of species j is its concentration multiplied by its activity coefficient, ai = Ciƒi ƒi = activity coefficient. • At low ionic strength, activity coefficients approach unity.

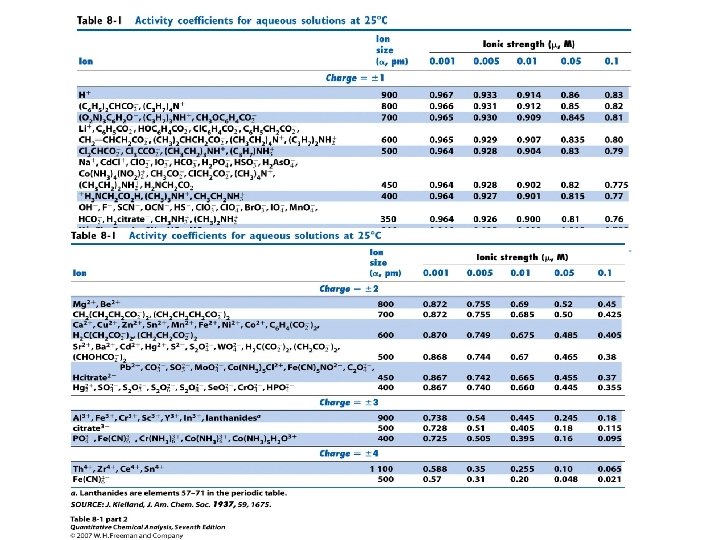
Activity and Activity Coefficients • Calculation of Activity Coefficients • Extended Debye-Huckel Equation: • ai = ion size parameter in angstrom (Å) 1 Å = 100 picometers (pm, 10 -10 meters) • Limitations: singly charged ions = 3 Å • log ƒi = - 0. 51 Zi 2(m)½ / (1+ (m)½) • On page 144: “the equation works fairly well for μ≤ 0. 1 M “ (? ? )

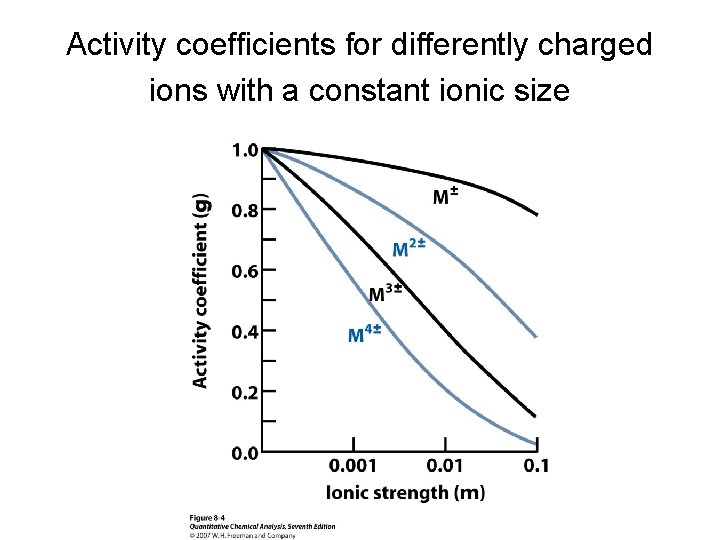
Effect of ionic strength, ion charge, and ion size on the activity coefficient • The ion size α in eq 8 -6 is an empirical parameter that provides good agreement between measured activity coefficients. • α is the diameter of the hydrated ion. • As ionic strength increases, the activity coefficient decreases. • The activity coefficient approaches unity as the ionic strength approaches 0. • As the magnitude of the charge of the ion increases, the departure of its activity coefficient from unity increases. • The smaller the ion size (α), the more important activity effects become.


Activity coefficients for differently charged ions with a constant ionic size

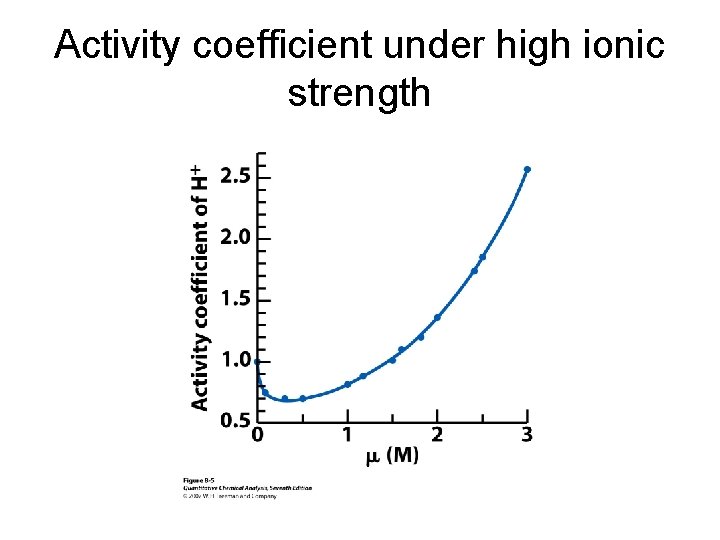
Activity coefficient under high ionic strength

Activity coefficient for non-ionic compounds • Case 1: neutral molecules in solution phase, the activity coefficient is unity and thus the activity is numerically the same as their concentration. • Case 2: Gases. The fugacity (i. e. activity) is calculated as the product of pressure and the fugacity coefficient (i. e. activity coefficient). When the pressure is below 1 bar, the fugacity coefficient is close to unity.

Diverse Ion (Inert Electrolyte) Effect: • Kspo = a. Ag+. a. Cl- = 1. 75 x 10 -10 • Adding Na 2 SO 4 to saturated solution of Ag. Cl, at high concentration of diverse (inert) electrolyte: higher ionic strength, m • a. Ag < [Ag ] ; a. Cl < [Cl ] • a. Ag. a. Cl < [Ag ] [Cl ] + + + - - - + - • Kspo < [Ag+] [Cl-] ; Kspo < [Ag+] = solubility • Solubility = [Ag+] > Kspo

Equilibrium calculations using activities • Solubility of Pb. I 2 in 0. 1 M KNO 3 m = 0. 1 = {0. 1(1+)2 + 0. 1(1 -)2}/2 (ignore Pb 2+, I-) • • ƒPb = 0. 35 ƒI = 0. 76 • Kspo = (a. Pb)1(a. I)2 = ([Pb 2+] Pb )1([I-] I )2 • Kspo = ([Pb 2+] [I-]2)( Pb I 2 ) = Ksp ( Pb I 2 ) • Ksp = Kspo / ( Pb I ) • Ksp = 7. 1 x 10 -9 /((0. 35)(0. 76)2) = 3. 5 x 10 -8 • (s)(2 s)2 = Ksp s = (Ksp/4)1/3 s =2. 1 x 10 -3 M • Note: If s = (Kspo/4)1/3 then s =1. 2 x 10 -3 M • The solubility is increased by approx. 43%

8 -3 p. H revisited Addition example: Calculate the p. H of water containing 0. 010 M KCl at 25 o. C. (see in class discussion).

8 -4 Systematic treatment of equilibrium • • Step 1: Write the pertinent reactions. Step 2: Write the charge balance equation. Step 3: Write mass balance equations. Step 4: Write equilibrium constant expression for each chemical reaction. • Step 5: Count the equations and unknowns. The numbers should be the same. • Step 6: Solve those equations.

8 -5 Applying the systematic treatment of equilibrium Solubility of calcium sulfate Step 1 Step 2 Step 3 Step 4 Step 5: counting Step 6

Solubility of magnesium hydroxide Step 6