Chemistry 2100 Lecture 8 Enantiomers Nonsuperposable mirror images

Chemistry 2100 Lecture 8
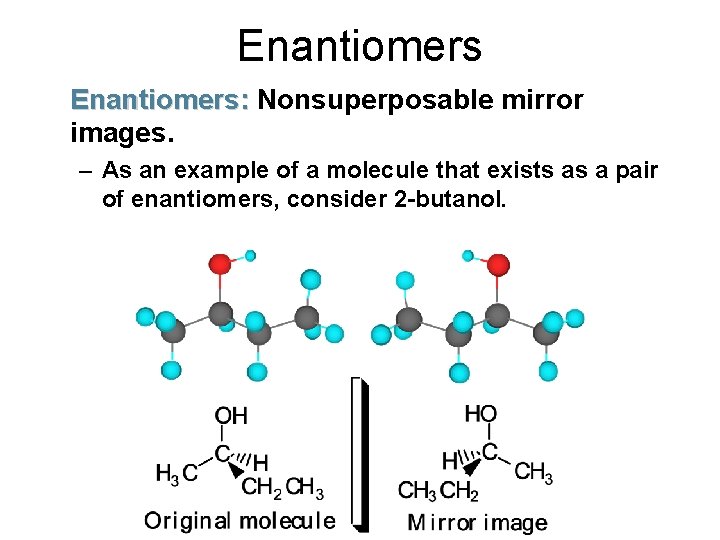
Enantiomers: Nonsuperposable mirror images. – As an example of a molecule that exists as a pair of enantiomers, consider 2 -butanol.
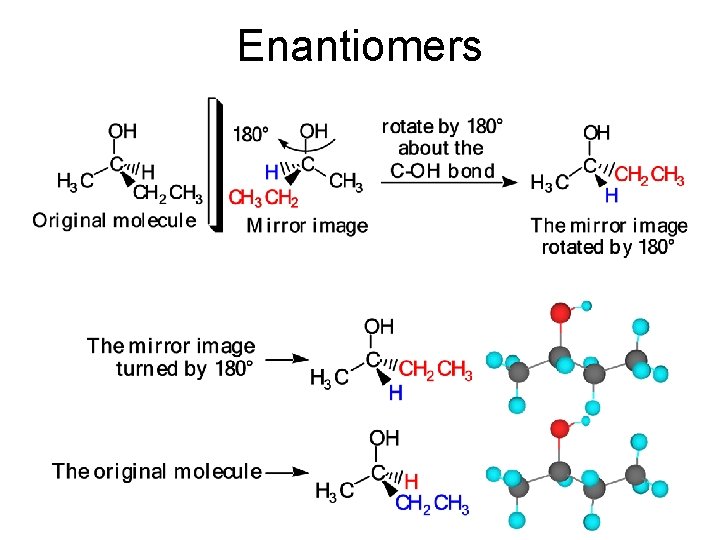
Enantiomers
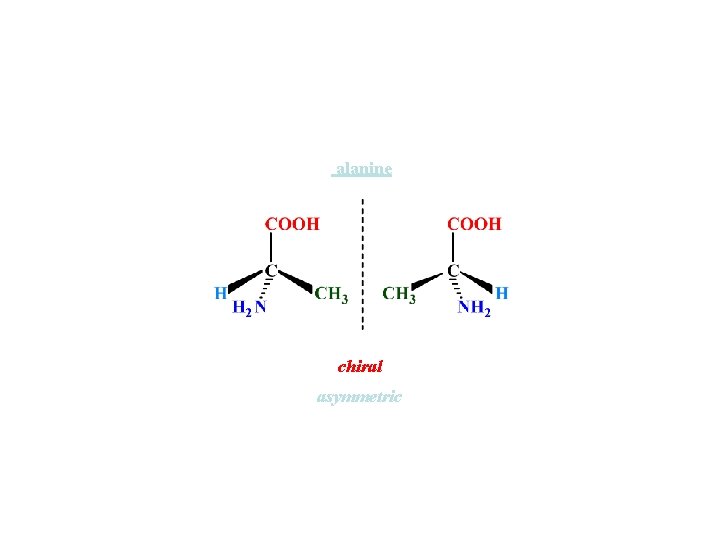
alanine chiral asymmetric
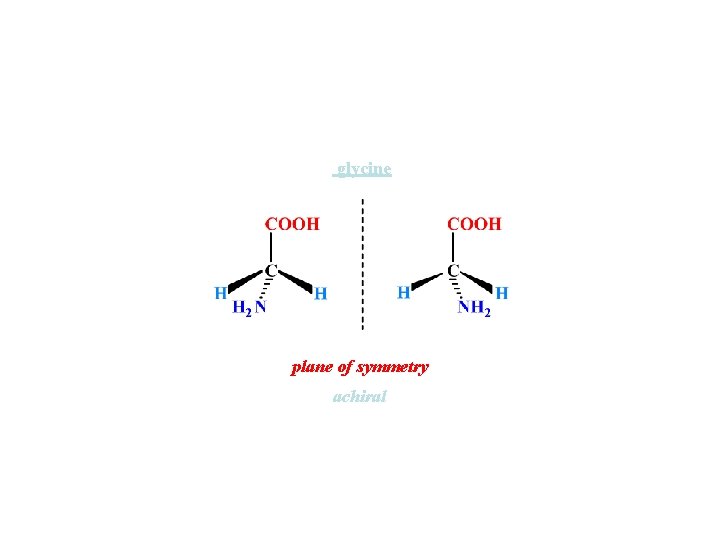
glycine plane of symmetry achiral

Enantiomers Objects that are nonsuperposable on their mirror images are chiral (from the Greek: cheir, hand). – They show handedness. The most common cause of enantiomerism in organic molecules is the presence of a carbon with four different groups bonded to it. – A carbon with four different groups bonded to it is called a stereocenter

Drawing Enantiomers

Enantiomers matter! * * *

Properties of Enantiomers
Optical activity

plane-polarized light

plane-polarized light

plane-polarized light

plane-polarized light

plane-polarized light
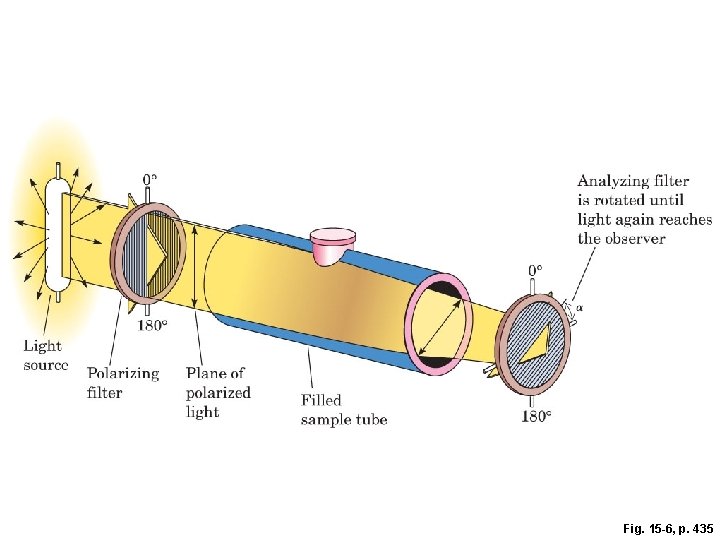
Fig. 15 -6, p. 435

Assigning Rotation dextrorotatory (d) levorotatory (l) (±) or (dl) (+) rotation "right-hand isomer" (–) rotation "left-hand isomer" optically inactive raceme

Multiple Chiral Centers For a molecule with n stereocenters, the maximum number of possible stereoisomers is 2 n. – We have already verified that, for a molecule with one stereocenter, 21 = 2 stereoisomers (one pair of enantiomers) are possible. – For a molecule with two stereocenters, a maximum of 22 = 4 stereoisomers (two pair of enantiomers) are possible. – For a molecule with three stereocenters, a maximum of 23 = 8 stereoisomers (four pairs of enantiomers) are possible, and so forth.

The Four Stereoisomers of a Simple sugar 2 R, 3 S 2 S, 3 R 2 R, 3 R 2 S, 3 S

Fischer Projections

Threose and Erythrose, Fischerized
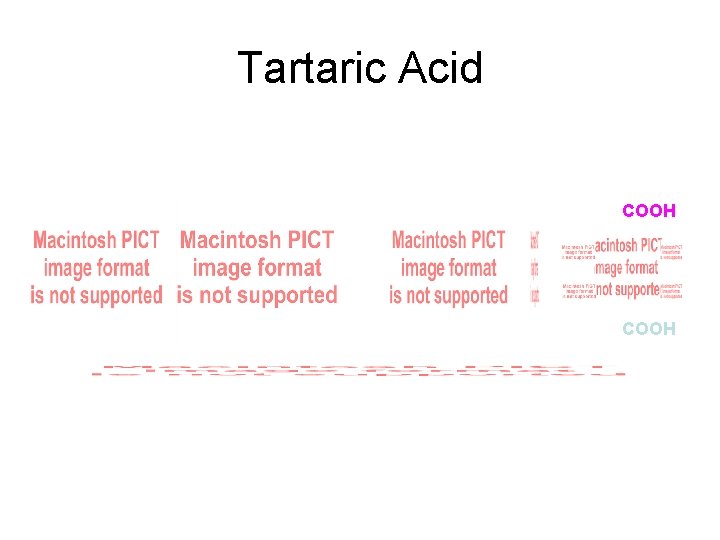
Tartaric Acid COOH meso
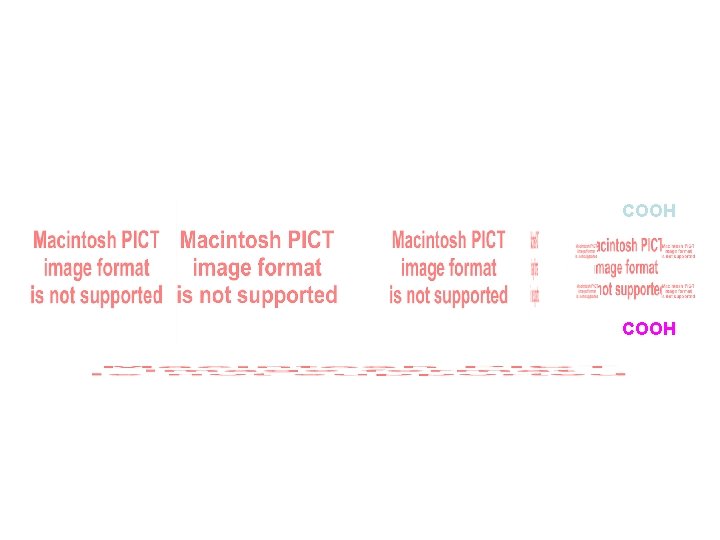
COOH meso
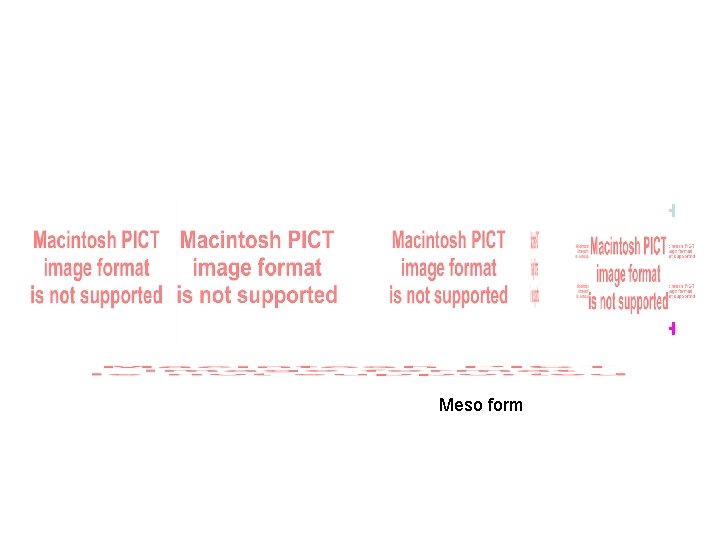
COOH Meso formmeso

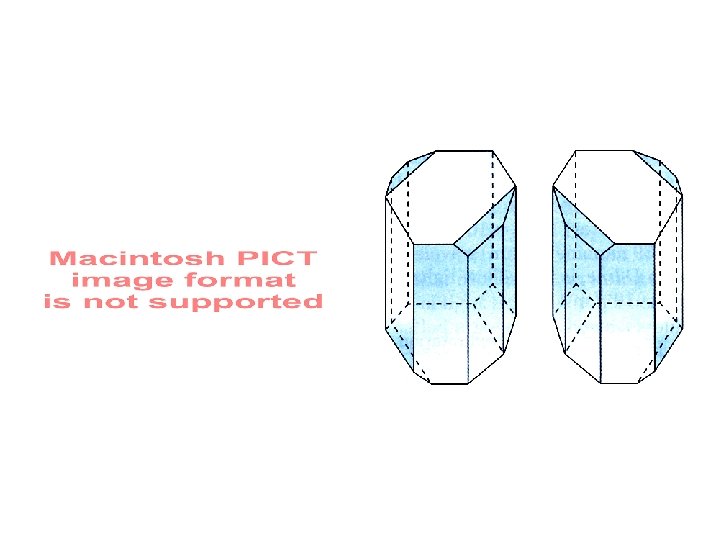

Stereochemical Reactions

* pyruvic acid lactic acid

* pyruvic acid lactic acid






But, Biologically… (+)





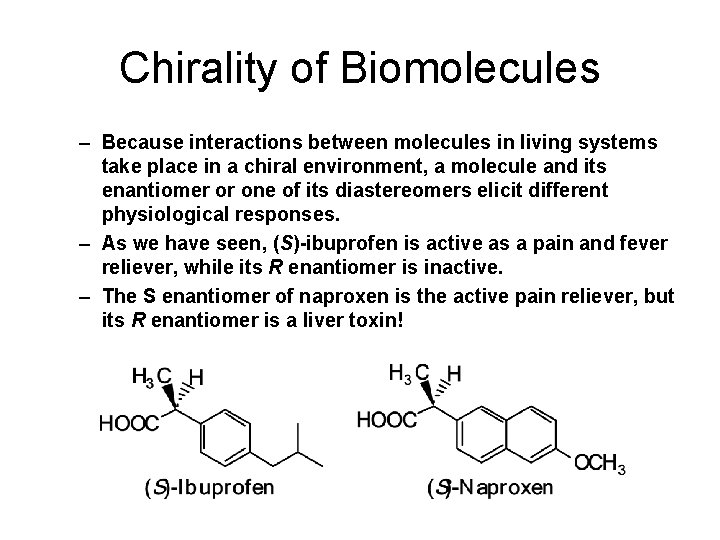
Chirality of Biomolecules – Because interactions between molecules in living systems take place in a chiral environment, a molecule and its enantiomer or one of its diastereomers elicit different physiological responses. – As we have seen, (S)-ibuprofen is active as a pain and fever reliever, while its R enantiomer is inactive. – The S enantiomer of naproxen is the active pain reliever, but its R enantiomer is a liver toxin!
- Slides: 48