Chemistry 2100 Chapter 28 Carbohydrate Catabolism glycolysis glucose

Chemistry 2100 Chapter 28
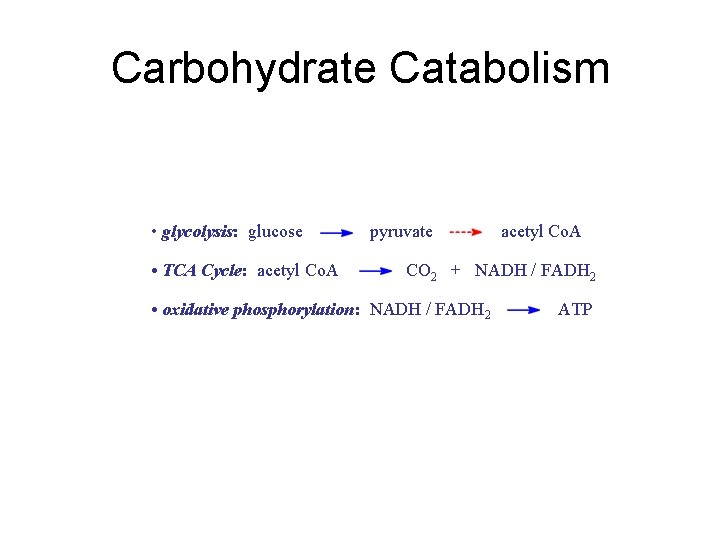
Carbohydrate Catabolism • glycolysis: glucose • TCA Cycle: acetyl Co. A pyruvate acetyl Co. A CO 2 + NADH / FADH 2 • oxidative phosphorylation: NADH / FADH 2 ATP

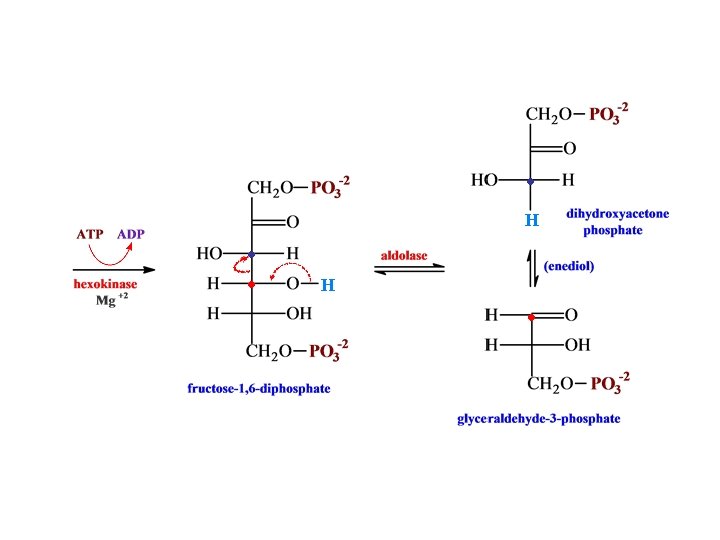
Glycolysis

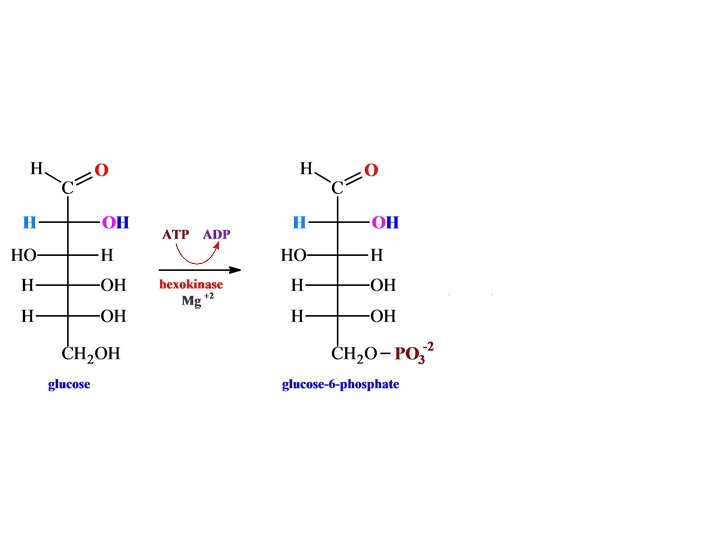
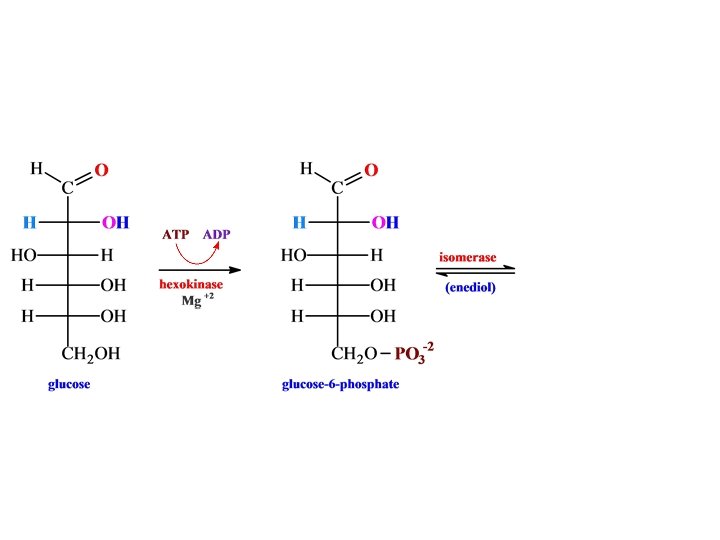
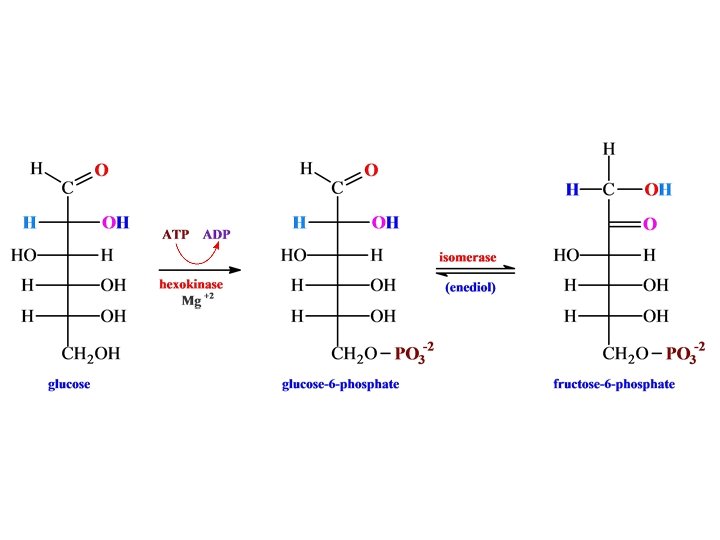

• H phosphofructokinase •
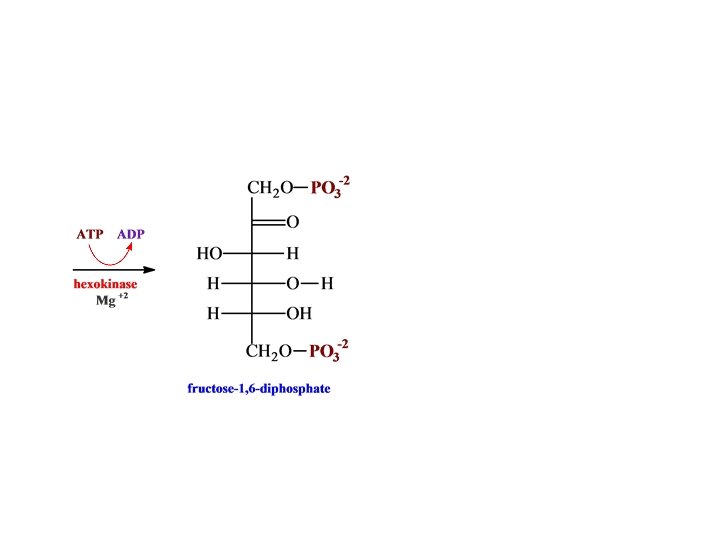
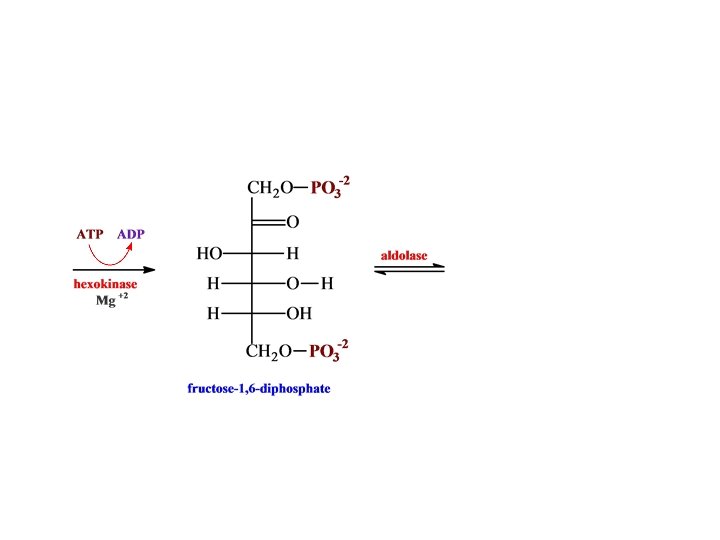

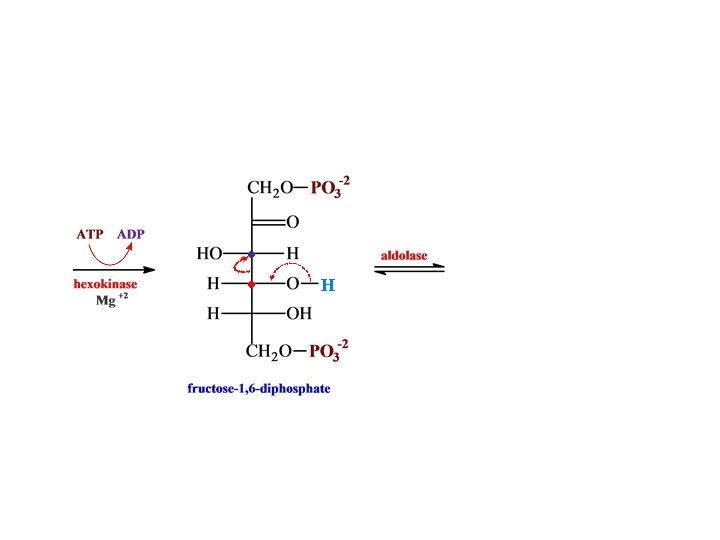
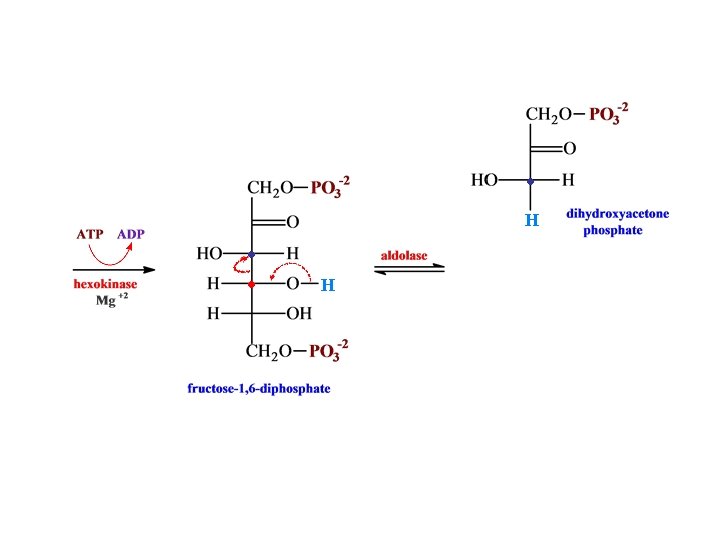

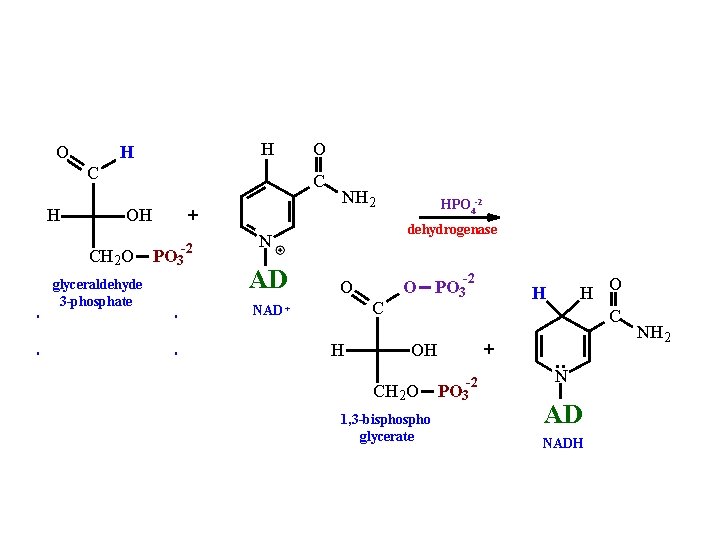
O H H C H O C OH CH 2 O glyceraldehyde 3 -phosphate + PO 3 -2 NH 2 dehydrogenase N AD HPO 4 -2 -2 O C NAD + H O PO 3 H O C H + OH • • CH 2 O 1, 3 -bispho glycerate -2 PO 3 N AD NADH NH 2

1, 3 bispho glycerate
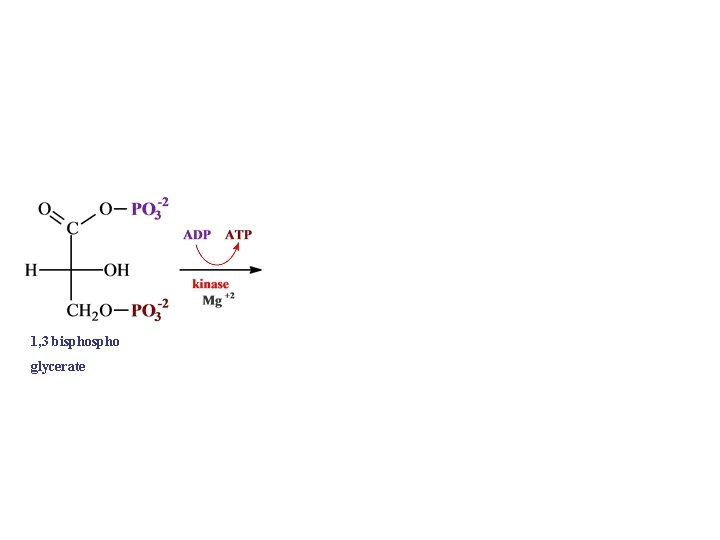
1, 3 bispho glycerate
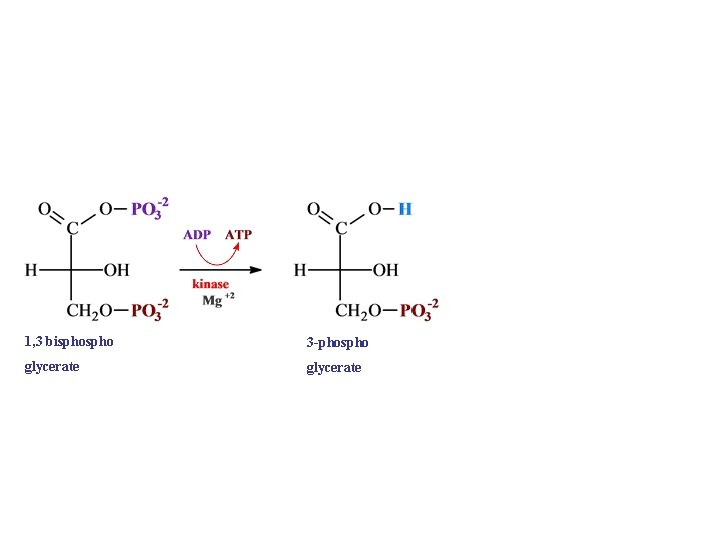
1, 3 bispho 3 -phospho glycerate
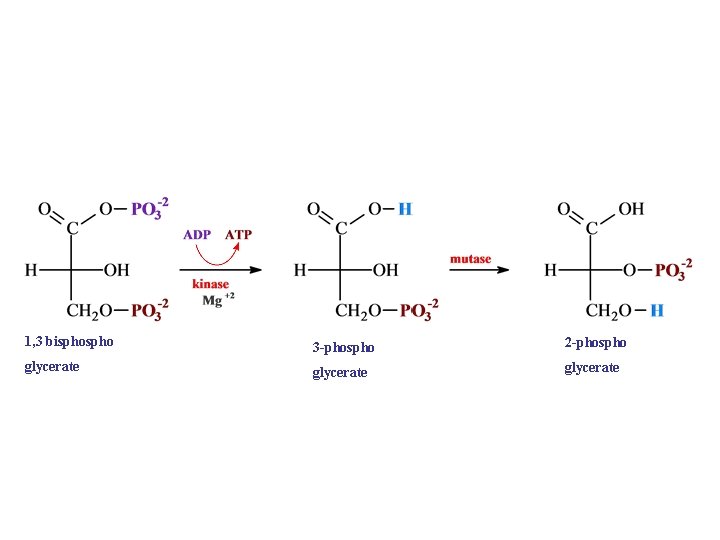
1, 3 bispho 3 -phospho 2 -phospho glycerate
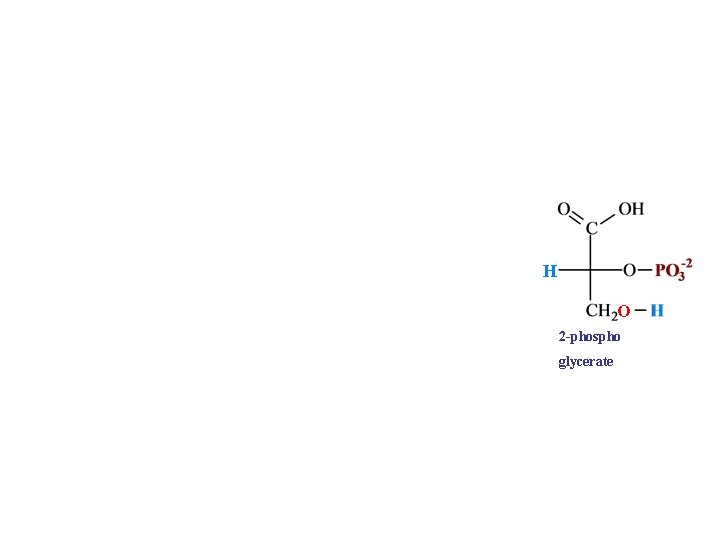
H O 2 -phospho glycerate
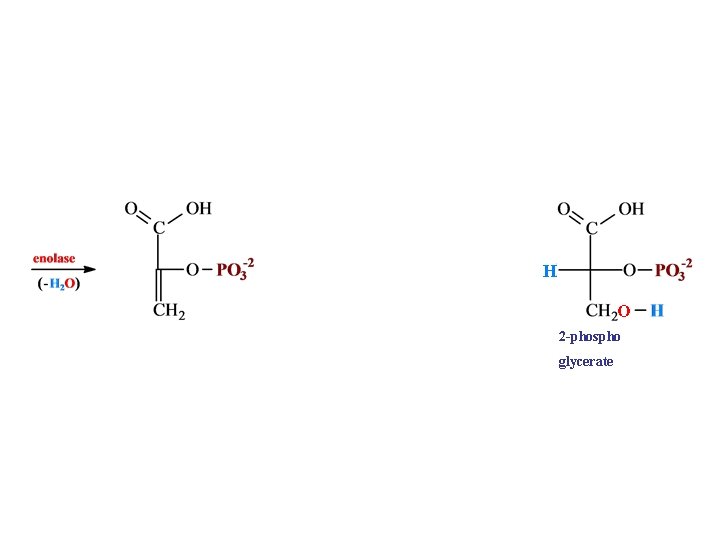
H O 2 -phospho glycerate



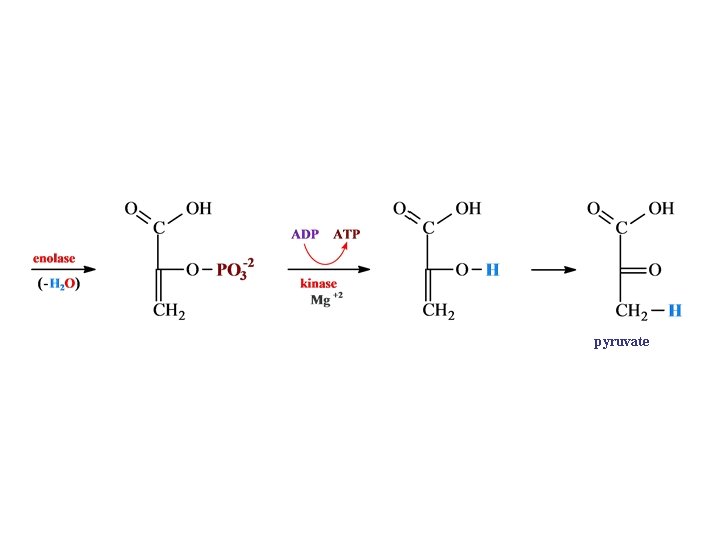
pyruvate
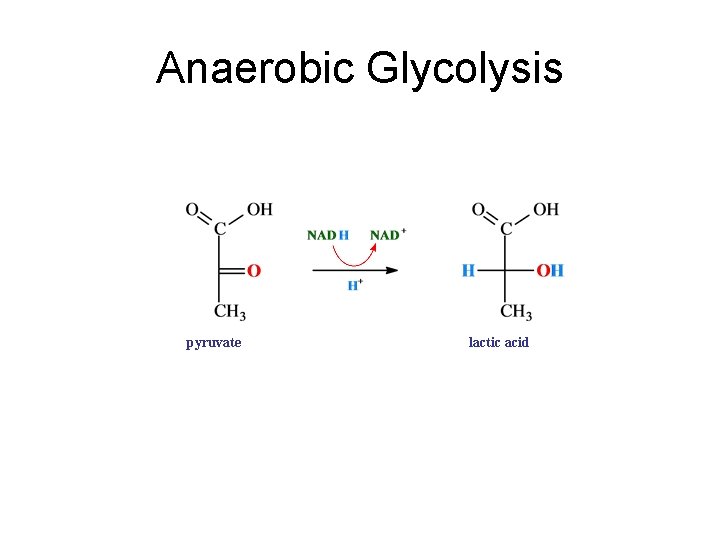
Anaerobic Glycolysis pyruvic acid pyruvate (–)-lactic acid
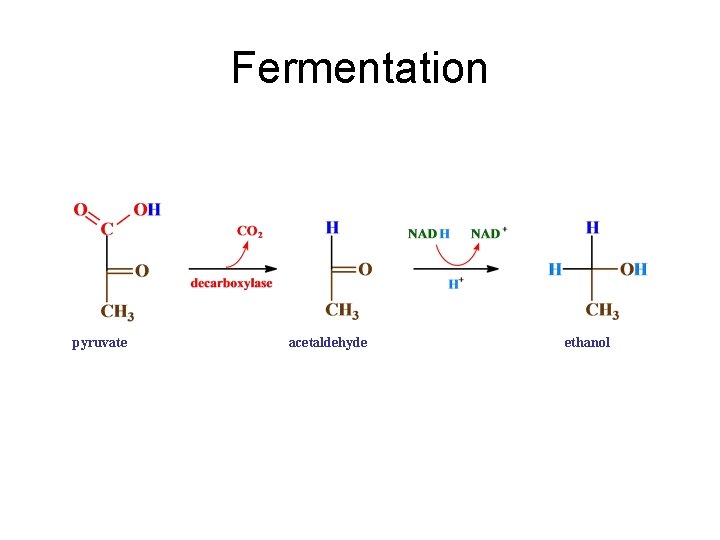
Fermentation pyruvic acid pyruvate acetaldehyde ethanol
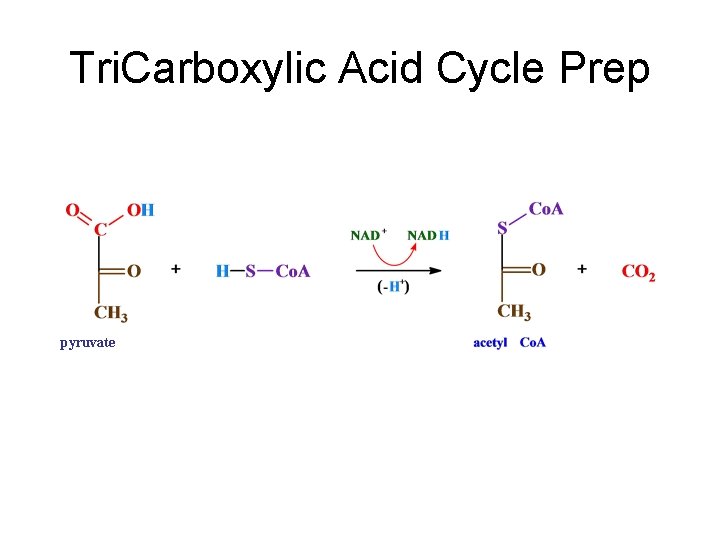
Tri. Carboxylic Acid Cycle Prep pyruvate

• http: //highered. mcgrawhill. com/sites/0072507470/student_view 0/ chapter 25/animation__how_glycolysis_wo rks. html

Fatty Acids and Energy Fatty acids in triglycerides are the principal storage form of energy for most organisms. – Hydrocarbon chains are a highly reduced form of carbon. – The energy yield per gram of fatty acid oxidized is greater than that per gram of carbohydrate oxidized.
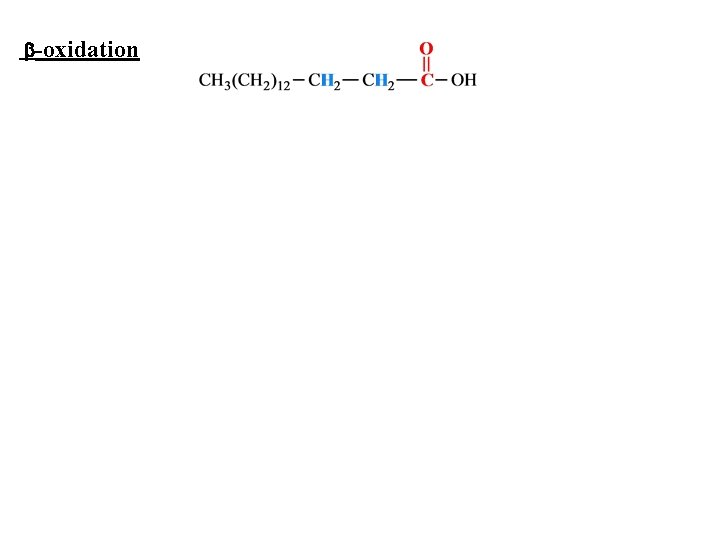
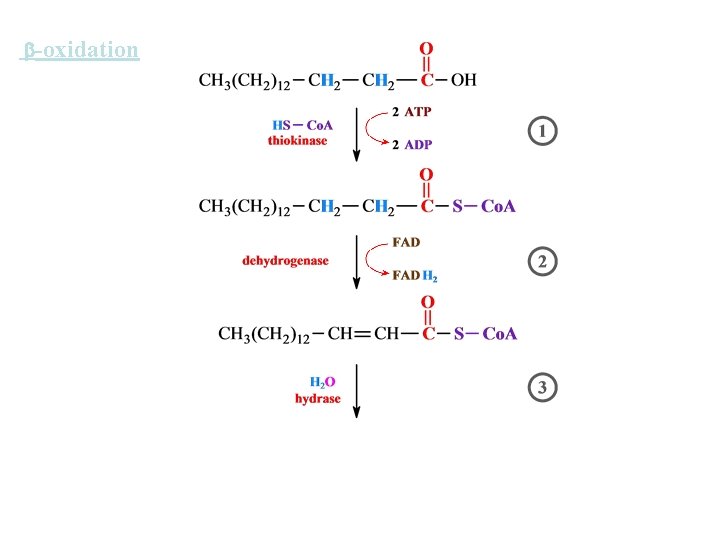
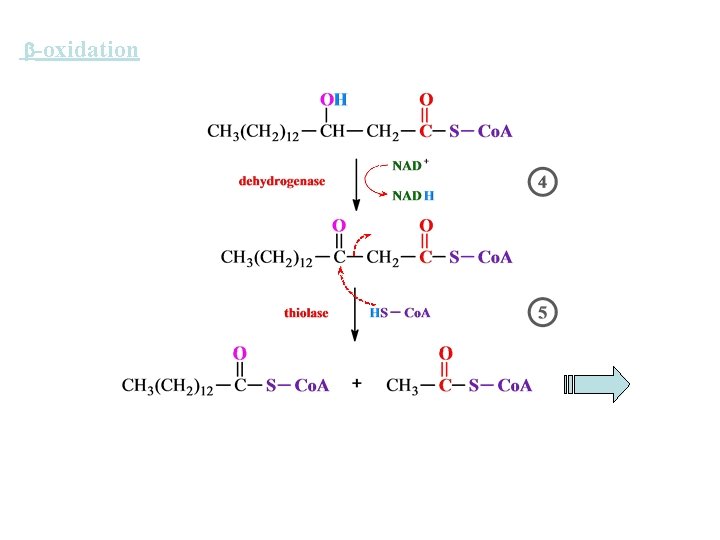
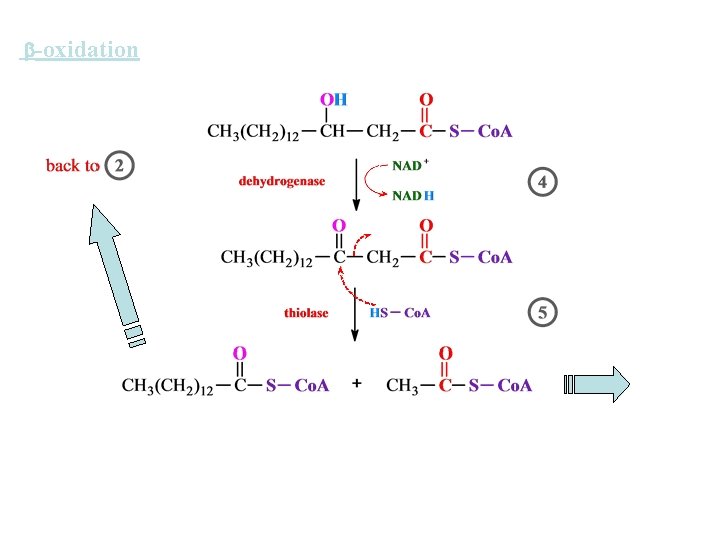
-oxidation
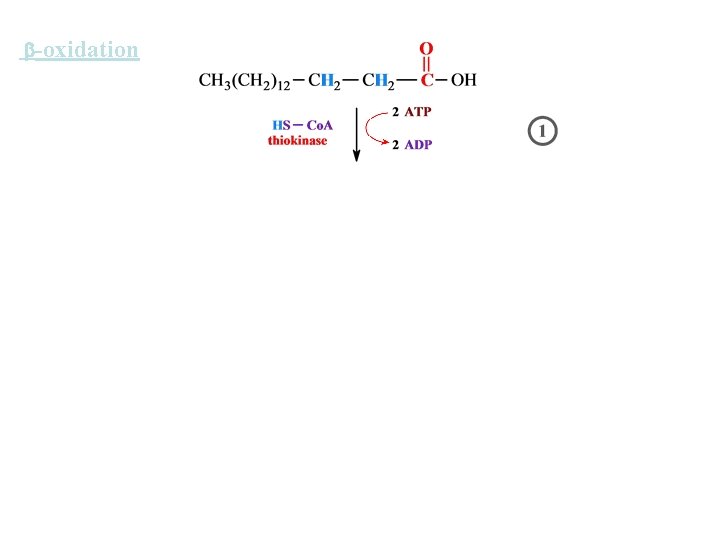
-oxidation
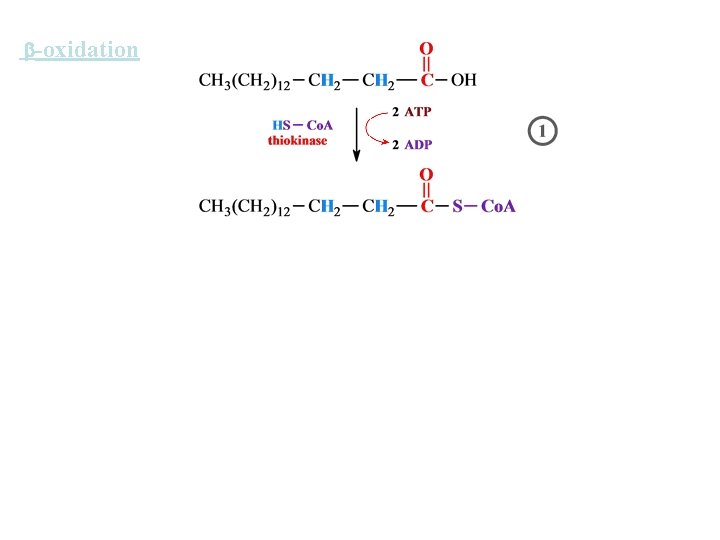
-oxidation
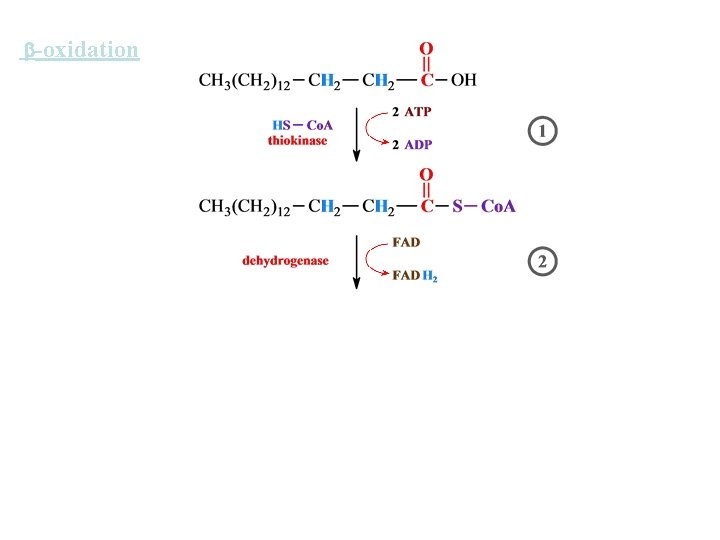
-oxidation
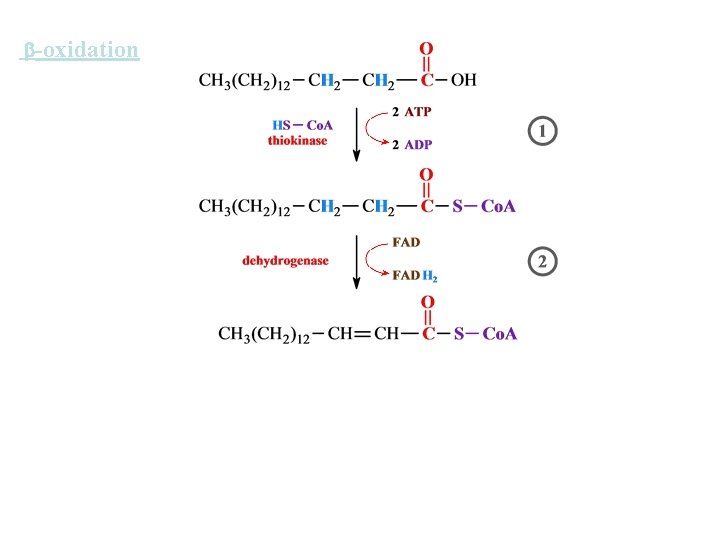
-oxidation
-oxidation
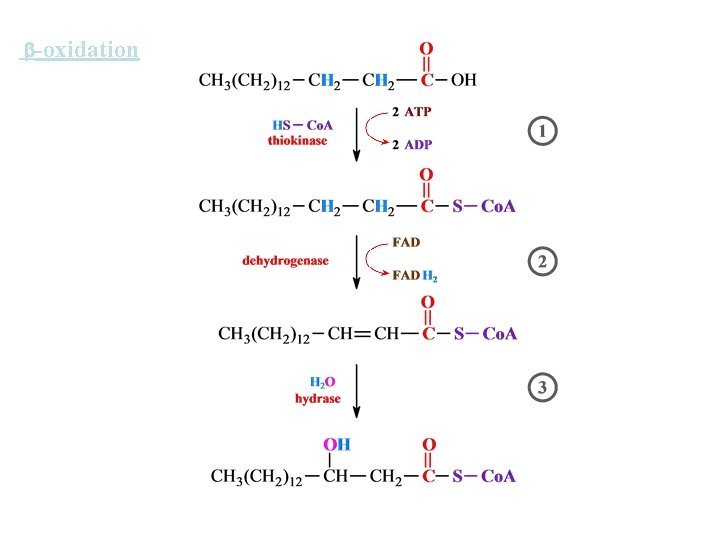
-oxidation
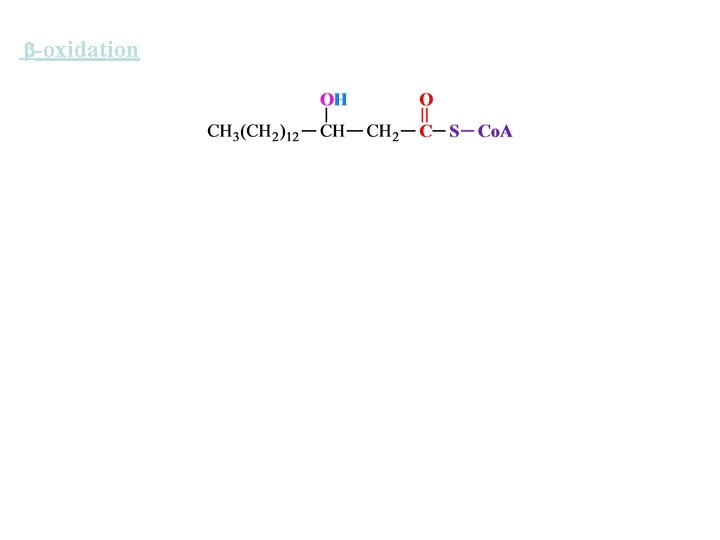
-oxidation
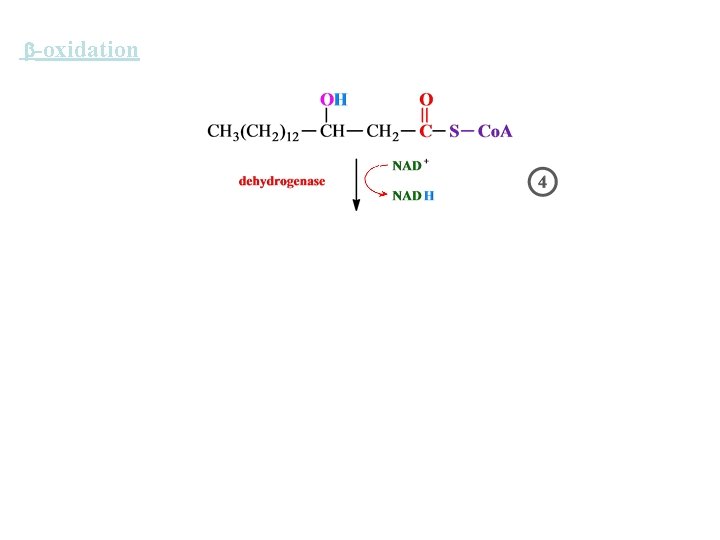
-oxidation
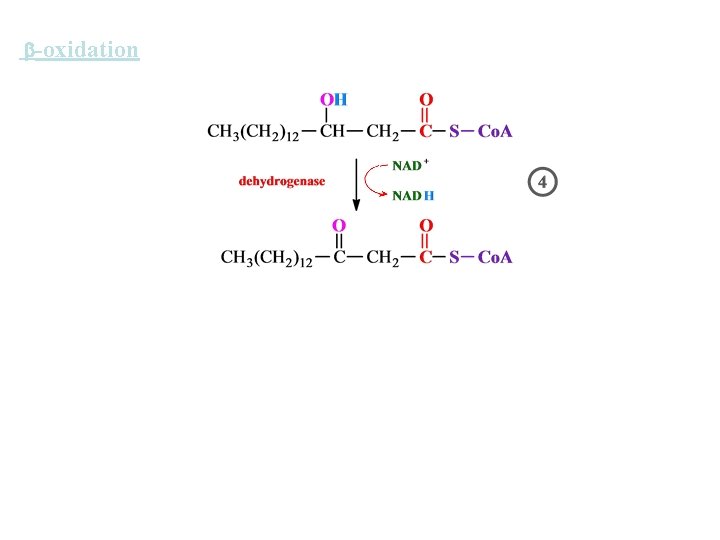
-oxidation
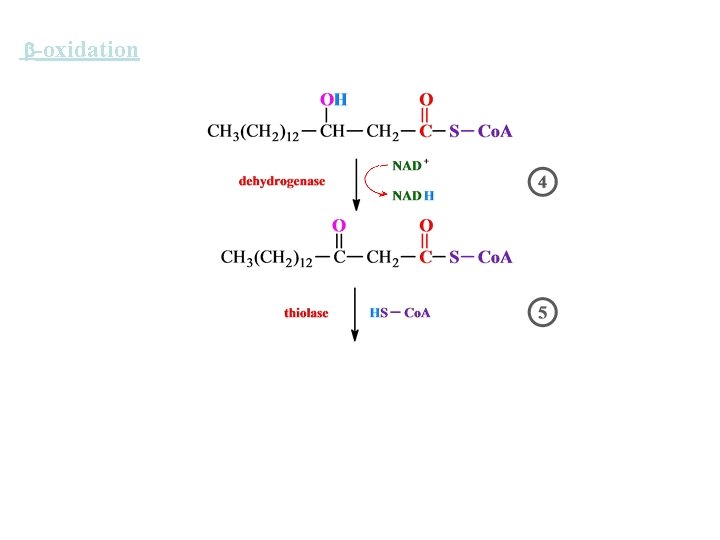
-oxidation
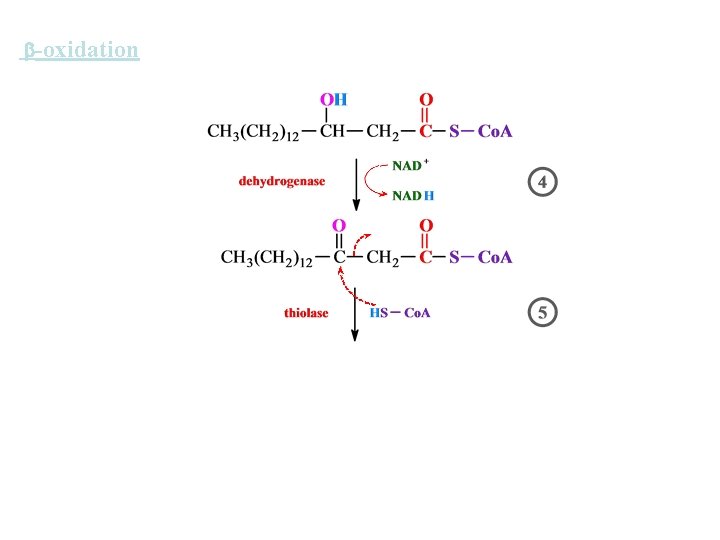
-oxidation
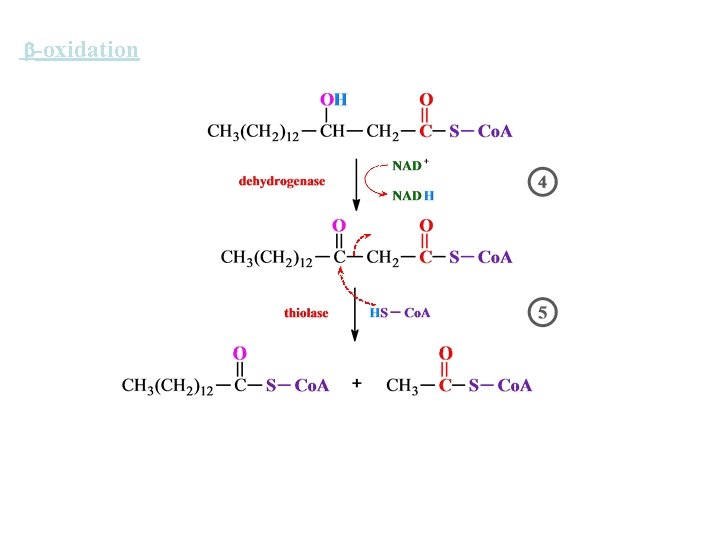
-oxidation
-oxidation
-oxidation

-oxidation


Energy Yield on -Oxidation • Yield of ATP per mole of stearic acid (C 18).

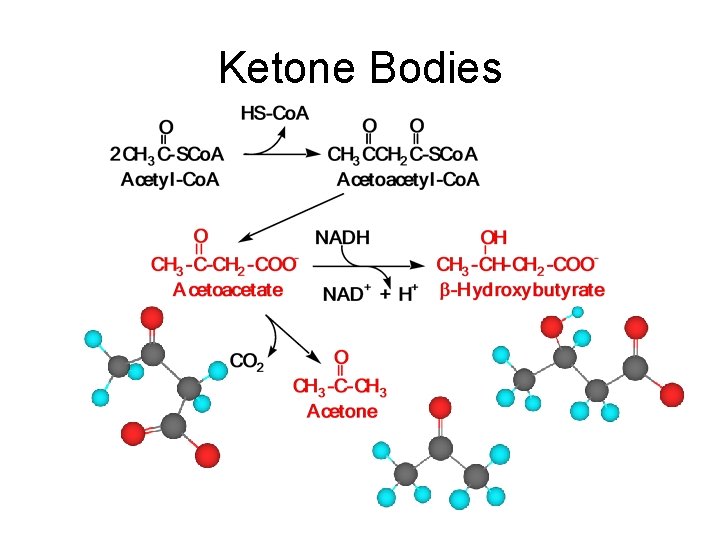
Ketone Bodies • Ketone bodies: bodies Acetone, -hydroxybutyrate, and acetoacetate; – Are formed principally in liver mitochondria. – Can be used as a fuel in most tissues and organs. • Formation occurs when the amount of acetyl Co. A produced is excessive compared to the amount of oxaloacetate available to react with it and take it into the TCA; for example: – Dietary intake is high in lipids and low in carbohydrates. – Diabetes is not suitably controlled. – Starvation.
Ketone Bodies
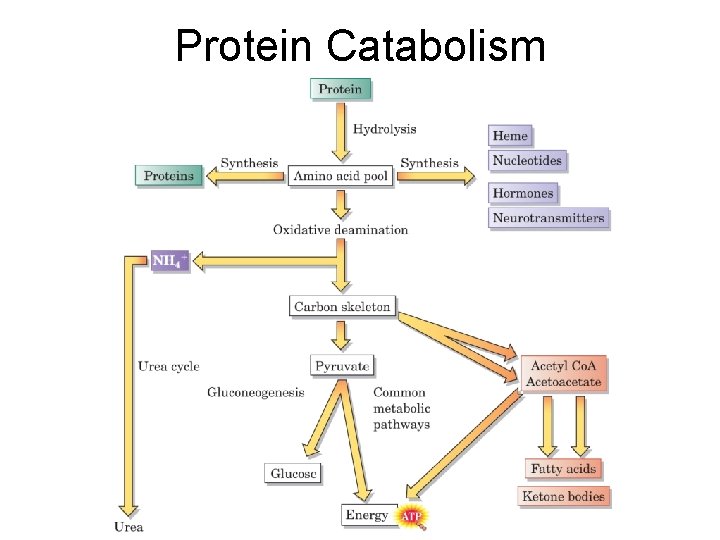
Protein Catabolism
- Slides: 52