Chemistry 18 5 Slide 1 of 21 18









![Using experimental data to determine reaction order [reactants] (in mol/L) Reaction rate (in mol/ Using experimental data to determine reaction order [reactants] (in mol/L) Reaction rate (in mol/](https://slidetodoc.com/presentation_image_h2/82b6bc2ea25aa62fa762b950045d4e31/image-10.jpg)













- Slides: 24

Chemistry 18. 5 Slide 1 of 21

18. 5 The Progress of Chemical Reactions During the course of the Tour de France, cyclists travel almost 4000 kilometers with steep mountains at 1600 meters or more. Crossing these high mountains requires extra energy. Chemical reactions can be thought of as processes involving peaks and valleys also. You will learn about the energy of a chemical reaction. Slide 2 of 21 © Copyright Pearson Prentice Hall

18. 5 The Progress of Chemical Reactions > Rate Laws What is the general relationship between the value of the specific rate constant, k, and the speed of a chemical reaction? Slide 3 of 21 © Copyright Pearson Prentice Hall

18. 5 The Progress of Chemical Reactions > Rate Laws The rate law is an expression for the rate of a reaction in terms of the concentration of reactants. The specific rate constant (k) for a reaction is a proportionality constant relating the concentrations of reactants to the rate of the reaction. Slide 4 of 21 © Copyright Pearson Prentice Hall

18. 5 The Progress of Chemical Reactions > Rate Laws The value of the specific rate constant, k, is large if the products form quickly; the value is small if the products form slowly. Slide 5 of 21 © Copyright Pearson Prentice Hall

The Progress of Chemical Reactions > Rate Laws Animation 24 Observe the characteristics of a first-order reaction. Slide 6 of 21 © Copyright Pearson Prentice Hall

18. 5 The Progress of Chemical Reactions > Rate Laws In a first-order reaction, the reaction rate is directly proportional to the concentration of only one reactant. As the concentration of that reactant decreases, the rate of the reaction also decreases by the same factor. Ex: The decomposition of Ca. CO 3(s): Ca. CO 3(s) Ca(s) + CO 2(g) + ½ O 2(g) Rate = k [Ca. CO 3(s)] Slide 7 of 21 © Copyright Pearson Prentice Hall

18. 5 The Progress of Chemical Reactions > Rate Laws Slide 8 of 21 © Copyright Pearson Prentice Hall

Reaction Order is the power to which the reactant(s) must be raised to give the experimentally observed relationship between concentration and rate. NOTE: Reaction order cannot be determined from the balanced chemical equation – it must be determined experimentally by changing concentration of the reactants by a known factor and measuring the factor by which the reaction rate changes Slide 9 of 21 © Copyright Pearson Prentice Hall
![Using experimental data to determine reaction order reactants in molL Reaction rate in mol Using experimental data to determine reaction order [reactants] (in mol/L) Reaction rate (in mol/](https://slidetodoc.com/presentation_image_h2/82b6bc2ea25aa62fa762b950045d4e31/image-10.jpg)
Using experimental data to determine reaction order [reactants] (in mol/L) Reaction rate (in mol/ L s) 0. 50 0. 0040 1. 00 0. 0080 [reactants] (in mol/L) Conclusion Reaction is first order as reaction rate increased to 2 x the original rate when [reactants] was 2 x the original amount Reaction rate (in mol/ L s) Conclusion 0. 50 0. 0020 1. 00 0. 0080 Reaction is second order as reaction rate increased to 4 x the original rate when [reactants] was 2 x the original amount Reaction rate (in mol/ L s) Conclusion [reactants] (in mol/L) 0. 50 0. 0010 1. 00 0. 0080 Reaction is third order as reaction rate increased to 8 x the original rate when [reactants] was 2 x the original amount © Copyright Pearson Prentice Hall Slide 10 of 21

Conceptual Problem 18. 2 Slide 11 of 21 © Copyright Pearson Prentice Hall

Conceptual Problem 18. 2 Slide 12 of 21 © Copyright Pearson Prentice Hall

Conceptual Problem 18. 2 Slide 13 of 21 © Copyright Pearson Prentice Hall

Practice Problems for Conceptual Problem 18. 2 Problem Solving 18. 36 Solve Problem 36 with the help of an interactive guided tutorial. Slide 14 of 21 © Copyright Pearson Prentice Hall

18. 5 The Progress of Chemical Reactions > Reaction Mechanisms What do the hills and valleys in a reaction progress curve represent? Slide 15 of 21 © Copyright Pearson Prentice Hall

18. 5 The Progress of Chemical Reactions > Reaction Mechanisms An elementary reaction is a reaction in which reactants are converted to products in a single step. The series of elementary reactions or steps that take place during the course of a complex reaction is called a reaction mechanism. An intermediate is a product of one of the steps in the reaction mechanism. Slide 16 of 21 © Copyright Pearson Prentice Hall

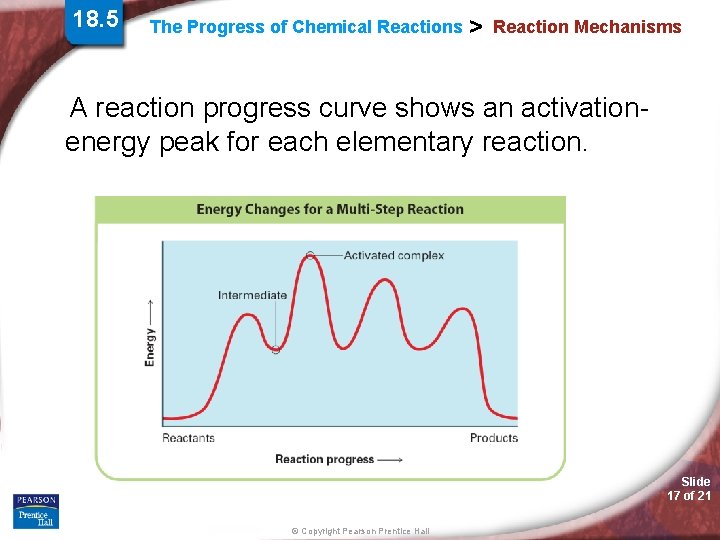
18. 5 The Progress of Chemical Reactions > Reaction Mechanisms A reaction progress curve shows an activationenergy peak for each elementary reaction. Slide 17 of 21 © Copyright Pearson Prentice Hall

18. 5 The Progress of Chemical Reactions > Reaction Mechanisms The peaks correspond to the energies of the activated complexes. Each valley corresponds to the energy of an intermediate. Slide 18 of 21 © Copyright Pearson Prentice Hall

18. 5 Section Quiz. Assess students’ understanding of the concepts in Section 18. 5. Continue to: -or- Launch: Section Quiz Slide 19 of 21 © Copyright Pearson Prentice Hall

18. 5 Section Quiz. 1. Choose the correct words for the spaces. If the specific rate constant, k, is _____, then products form _____. a. large, slowly b. large, quickly c. small, quickly d. small, a first order reaction Slide 20 of 21 © Copyright Pearson Prentice Hall

18. 5 Section Quiz. 2. Choose the correct words for the spaces. In a graph that shows the progress of a reaction, the peaks in the curve correspond to the energies of the _______ and the valleys correspond to the energies of the ____. a. activated complexes, reactants b. intermediates, activated complexes c. reactants, products d. activated complexes, intermediates © Copyright Pearson Prentice Hall Slide 21 of 21

18. 5 Section Quiz. 3. Which of the following statements is true? a. Most reactions are elementary reactions. b. In most reactions, reactants are converted to products in a single step. c. Most reactions take place in a series of steps called intermediates. d. Most reactions take place in a series of steps called a reaction mechanism. Slide 22 of 21 © Copyright Pearson Prentice Hall

18. 5 Section Quiz. 4. The reaction mechanism can best be determined from a. the specific rate constant. b. the overall chemical reaction. c. the rate law. d. experimentation. Slide 23 of 21 © Copyright Pearson Prentice Hall

END OF SHOW