Chemistry 139 General Chemistry Prep Lets begin with

Chemistry& 139 General Chemistry Prep

Lets begin with a quote from your text: “The message should be clear. The contents of this chapter (and Chapter 3) must be taken very seriously. Skimming over this material is a sure invitation to frustration and struggle with the chemical topics that follow. ” If you are having difficulties seek help early and frequently.

Unit Systems and Dimensional Analysis 3. 1 The Metric System of Units 3. 2 Metric Units of Length 3. 3 Metric Units of Mass 3. 4 Metric Units of Volume 3. 5 Units in Mathematical Operations 3. 6 Conversion Factors 3. 7 Dimensional Analysis 3. 8 Density 3. 9 Equivalence Conversion Factors Other than Density 3. 10 Percentage and Percentage Error 3. 11 Temperature Scales

Unit Systems and Dimensional Analysis 3. 1 The Metric System of Units

All measurements have at least three parts a number (the magnitude), an error that produces an uncertainty and units. e. g. “today’s maximum temperature was 27. 5 ± 0. 2 o. C” The units are a label that tells us what is being measured: e. g. Gallons tells us volume is being measured, miles tells us length is being measured.

In science we use the international system of units (Le Système international d'unités, SI). This is a modern version of the metric system. Metric system is preferred over the English system of units as the relationships between units are always powers of ten. e. g. Metric English 1 cm = 10 -2 m 1 foot = 12 inches 1 m. L = 10 -3 L 1 gallon = 4 quarts
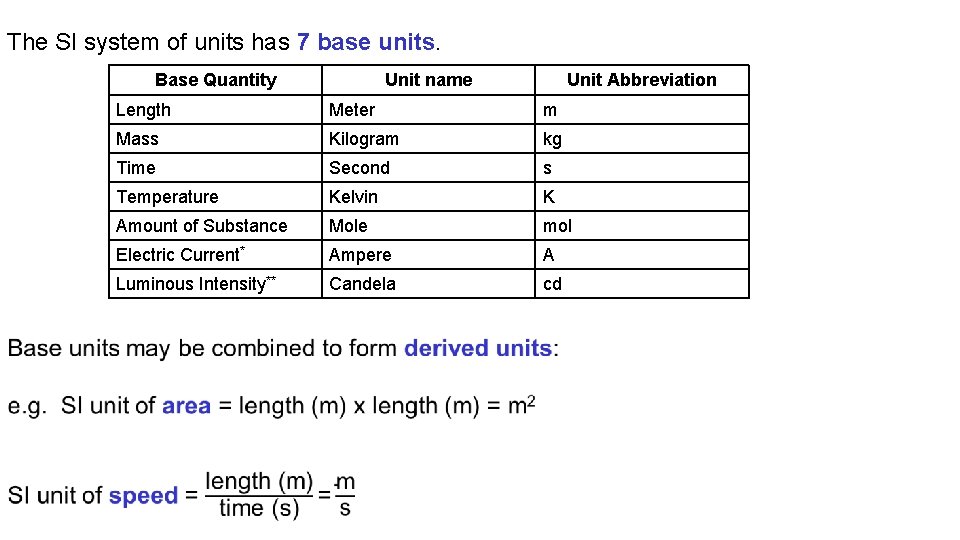
The SI system of units has 7 base units. Base Quantity Unit name Unit Abbreviation Length Meter m Mass Kilogram kg Time Second s Temperature Kelvin K Amount of Substance Mole mol Electric Current* Ampere A Luminous Intensity** Candela cd
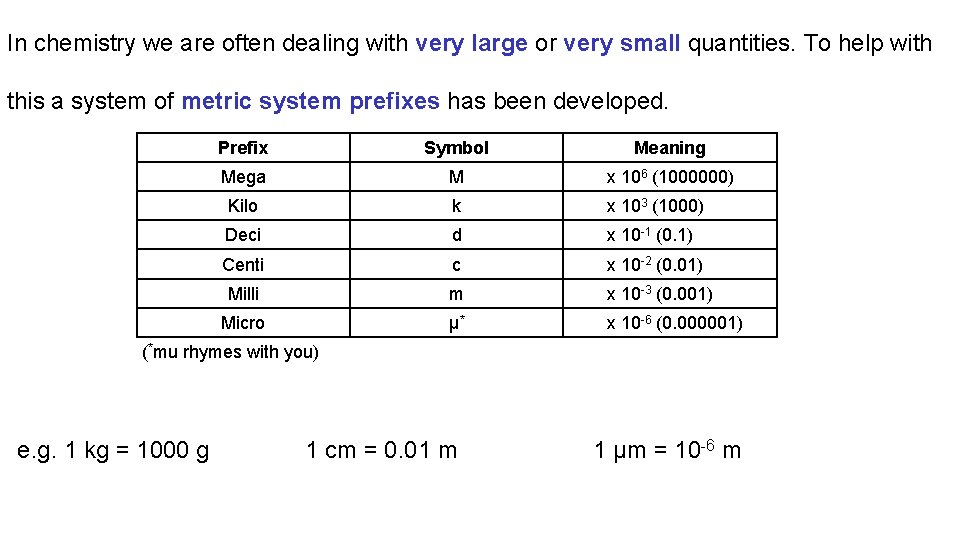
In chemistry we are often dealing with very large or very small quantities. To help with this a system of metric system prefixes has been developed. Prefix Symbol Meaning Mega M x 106 (1000000) Kilo k x 103 (1000) Deci d x 10 -1 (0. 1) Centi c x 10 -2 (0. 01) Milli m x 10 -3 (0. 001) Micro μ* x 10 -6 (0. 000001) (*mu rhymes with you) e. g. 1 kg = 1000 g 1 cm = 0. 01 m 1 μm = 10 -6 m

Unit Systems and Dimensional Analysis 3. 1 The Metric System of Units All measurements contain a number (size), some uncertainty, and units indicating what is being measured. Scientists use a modern version of the metric system called the SI system of units. The SI system of units has 7 base units which can be combined to form derived units. To assist in writing very small and very large quantities metric system prefixes are used, these act as multipliers.

Unit Systems and Dimensional Analysis 3. 2 Metric Units of Length

The base unit of length in the SI system is the metre (meter). A meter is slightly longer than a yard. 1 meter = 1. 09 yards ~ 1 meter 1 km = 1000 m 1 cm = 10 -2 m 1 mm = 10 -3 m 1 nanometer = 10 -9 m

Unit Systems and Dimensional Analysis 3. 2 Metric Units of Length The SI unit of length is the meter, however, in the chemistry laboratory we are most commonly measuring the length of objects with sizes of a few mm or cm. Kilometers is often used to measure the distance between geographical locations.

Unit Systems and Dimensional Analysis 3. 3 Metric Units of Mass

The base unit of mass in the SI system is the kilogram. A kilogram is slightly more than two pounds. 1 kilogram = 2. 20 pounds Commonly in chemistry we will measure the mass of objects in grams or milligrams. 1 kg = 1000 g 1 mg = 10 -3 g A quart of milk has a mass of ~ 1 kg

Unit Systems and Dimensional Analysis 3. 3 Metric Units of Mass The base unit of mass in the SI system of units is the kilogram. Mass is most conveniently measured in the chemistry laboratory in grams or occasionally milligrams.

Unit Systems and Dimensional Analysis 3. 4 Metric Units of Volume
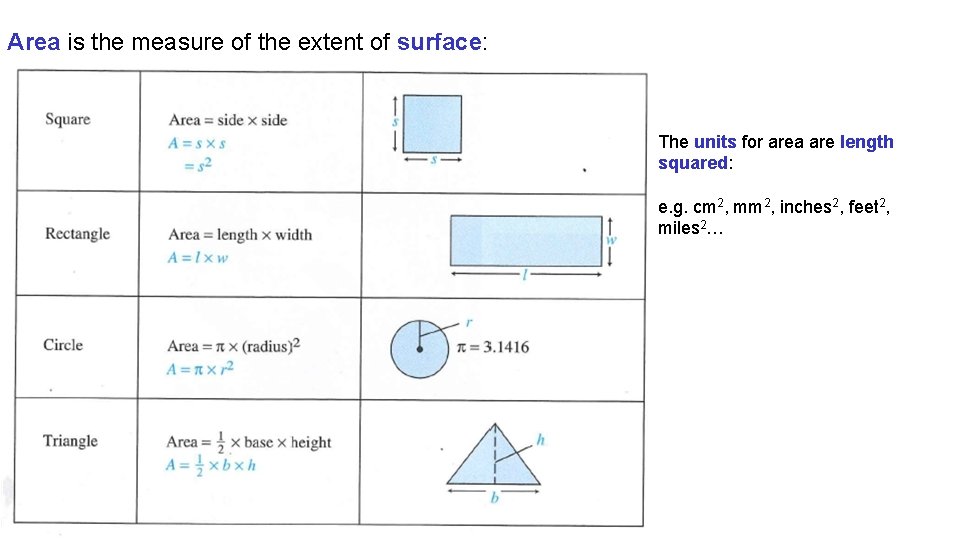
Area is the measure of the extent of surface: The units for area are length squared: e. g. cm 2, mm 2, inches 2, feet 2, miles 2…

Volume is the measure of the amount of space an object occupies: The units for volume are length cubed: e. g. cm 3, mm 3, inches 3, feet 3, miles 3…

The cubic meter is the SI unit of volume it corresponds to the volume occupied by a cube with sides of 1 meter. Volume = 1 m x 1 m = 1 m 3 1 m The cubic meter is too large a unit for most chemistry applications. More commonly we will use dm 3 or cm 3.
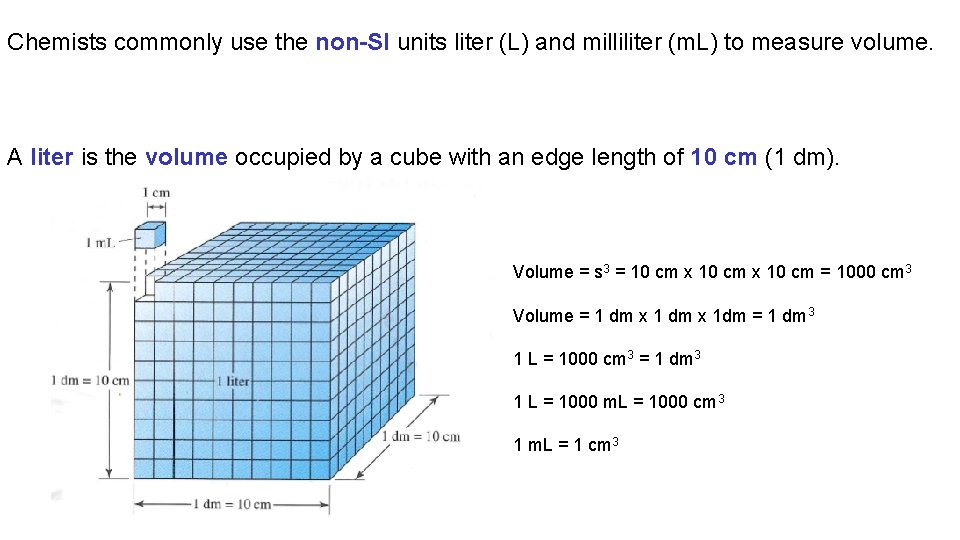
Chemists commonly use the non-SI units liter (L) and milliliter (m. L) to measure volume. A liter is the volume occupied by a cube with an edge length of 10 cm (1 dm). Volume = s 3 = 10 cm x 10 cm = 1000 cm 3 Volume = 1 dm x 1 dm = 1 dm 3 1 L = 1000 cm 3 = 1 dm 3 1 L = 1000 m. L = 1000 cm 3 1 m. L = 1 cm 3

Unit Systems and Dimensional Analysis 3. 4 Metric Units of Volume Area has units of length squared. Formulas are used to calculate the area of common shapes. Volume has units of length cubed. Formulas are used to calculate the volume of common shapes. The SI unit of volume is the m 3. Commonly in chemistry we use the non-SI units of volume the L and the m. L. One liter is the volume occupied by a cube with edges of 10 cm. 1 L is equivalent to 1 dm 3 or 1000 cm 3. There are one thousand m. L in a liter. The m. L and the cm 3 are equal in volume.

Unit Systems and Dimensional Analysis 3. 5 Units in Mathematical Operations


Unit Systems and Dimensional Analysis 3. 5 Units in Mathematical Operations In mathematical operations on measurements the exponents of units are treated in the same way we treat powers of ten. When multiplying measurements we add the exponents of the units, when dividing measurements we subtract the exponent of the units in the denominator from the exponent of the units in the numerator.

Unit Systems and Dimensional Analysis 3. 6 Conversion Factors

In chemistry we use the international system of units (Le Système international d'unités, SI). This is a modern version of the metric system. Unfortunately this system of units is not widely used in everyday life in the USA. Being able to use conversion factors and formulas to transform measurements between systems of units is extremely important.

There have been many serious incidents that have resulted from errors in converting between systems of units. Air Canada Flight 143 (You. Tube has the “air crash investigation” episode about this)

$193 million Mars Climate Orbiter. Lost in Space.

An equality can be written that describes the relationship between two units. e. g. 60 minutes = 1 hr We can re-arrange this equality to get two conversion factors: Multiplying by either of these conversion factors is the same as multiplying by one, but will result in a change in units.

We use a conversion factor that divides by the units we wish to cancel and multiplies by the units we want in our answer. e. g. Q. Express 1400 millimeters in meters. A.

Conversion factors can be used to convert between units within the metric system. The conversion factors we will use for metric-metric unit conversions are based on the prefixes we discussed earlier. These conversion factors are exact numbers (powers of 10) as they arise from unit definitions.

Q. Express 3. 65 km in meters. A.
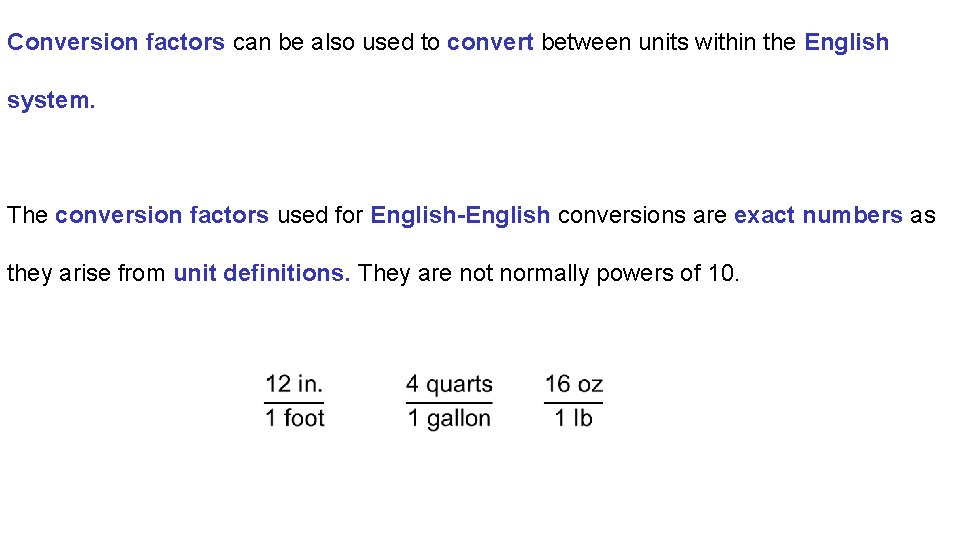
Conversion factors can be also used to convert between units within the English system. The conversion factors used for English-English conversions are exact numbers as they arise from unit definitions. They are not normally powers of 10.

Q. Express 3. 42 yards in feet. A.

Conversion factors can be also used to convert between the English and metric systems The conversion factors used for metric-English and English-metric conversions are not exact numbers as they arise from measurements. Table 3. 4 on page 71 has some conversion factors that relate English and metric units.

Q. Express 3. 42 yards in meters. A.

Unit Systems and Dimensional Analysis 3. 6 Conversion Factors Conversion factors are fractions derived from the equality describing the relationship between two units. We use the conversion factor that multiplies by the desired unit and divides by the unit to be eliminated. English-English conversions use exact conversion factors based on unit definitions. Similarly metric-metric conversions use exact conversion factors based on the metric prefixes. Conversions between the metric and English system use inexact conversion factors determined through measurement.
- Slides: 37