Chemistry 125 Lecture 8 OneDimensional Wave Functions The












- Slides: 29

Chemistry 125: Lecture 8 One-Dimensional Wave Functions The magnitude of the curvature of a wave function relates to the kinetic energy of the system, and the square of the wave function relates to probability density. The requirement that the wavefunction not diverge in areas of negative kinetic energy can constrain total energies to certain values, a property which is explored for the harmonic oscillator, the Morse potential, and the Columbic potential. Consideration of the influence of mass reveals an “isotope effect” on dynamics and on the energy, vibration frequency, and length of bonds. Synchronize when the speaker finishes saying “last time we saw the Jeopardy approach” Synchrony can be adjusted by using the pause(||) and run(>) controls. For copyright notice see final page of this file

Exam 1 - Friday, Sept. 26 ! Covers Lectures through Wednesday Including: Functional Groups X-Ray Diffraction 1 -Dimensional Quantum Mechanics & 1 -Electron Atoms (Sections I-V of quantum webpage & Erwin Meets Goldilocks) Exam Review 8 -10 pm Wednesday, Sept. 24, Room WLH ? ? ? Come with questions.

From “Jeopardy” Approach to Recipe for Solution of Schrödinger Equation Using Guessed Energies

Rearranging Schrödinger to give a formula for curve tracing. C Curvature of m Curvature of y y y = y +V=E m (VE) C Curves away from 0 for V>E; toward 0 for V<E. Since m, C, V(x) are given, this recipe allows tracing (x) in steps, from initial (0) [= 1], with initial slope [0], and a guessed E.

100 kcal/mole Too Cold Erwin. Nodes Meets and Quantization Goldilocks in One Dimension Danger Negative Just Right! from Kinetic Erwin Meets Goldilocks Energy Guess 21 kcal/mole (for Wiki see 20. 74 kcal/mole (Curve Away Guess 20 kcal/mole from Baseline) 0 Monday Problem Set) (Curve Away from Baseline) Too Hot 2. 5Å

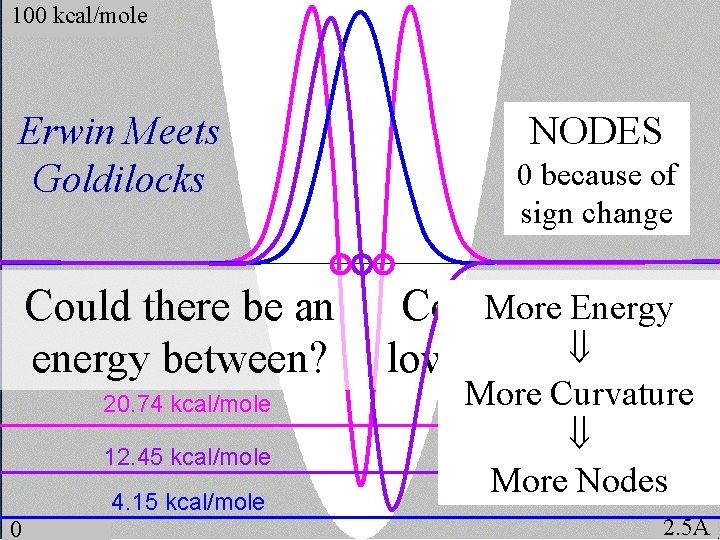
100 kcal/mole Erwin Meets Goldilocks Could there be an energy between? 20. 74 kcal/mole 12. 45 kcal/mole 4. 15 kcal/mole 0 NODES 0 because of sign change More Energy Could there be a Psi? lower-energy More Curvature More Nodes 2. 5Å
Much Harder for Many Particles Is it worth our effort?

Reward for Finding Knowledge of Everything e. g. Allowed Energies Structure Dynamics Bonding Reactivity

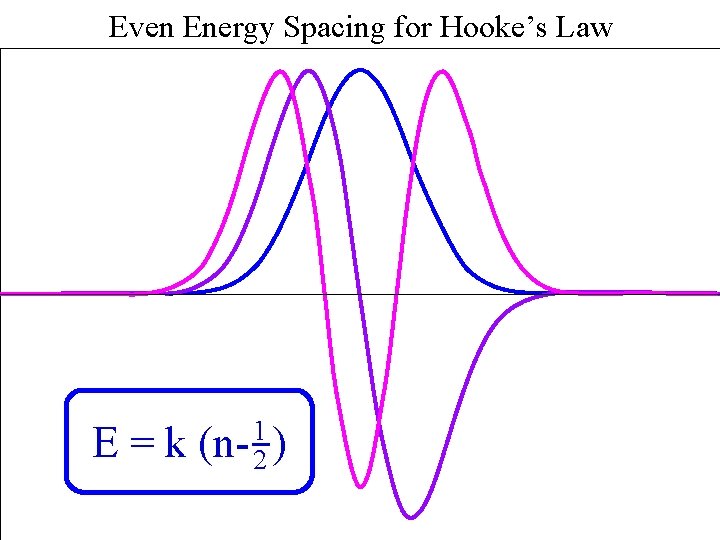
Even Energy Spacing for Hooke’s Law Harmonic Spacing E=k 1 (n- 2 )

“We only wish that we could glean an inkling of what could mean. ”

Structure: Y 2 Probability Density Oops! If one wishes to translate this result into physical terms, only one interpretation is possible, signifies the probability 1) [of the structure] 1) Correction in proof: more careful consideration shows that the probability is proportional to the square of the size of . Max Born (June 25, 1926)

Structure: Y 2 Probability Density Albert Einstein to Max Born December 4, 1926 Aber eine innere Stimme sagt mir, dass das doch nicht der wahre Jakob ist. Die Theorie liefert viel, aber dem Geheimnis des Alten bringt sie uns kaum näher. Jedenfalls bin ich überzeugt, dass der nicht würfelt. But an inner voice tells me, that this is not the real thing. The theory yields a great deal, but it brings us no nearer to the secret of the Old One. Anyway I am convinced that He does not play dice.

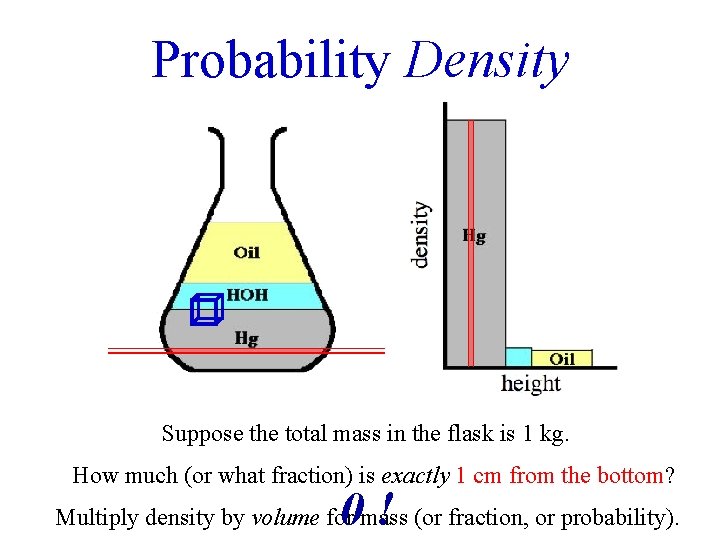
Probability Density Suppose the total mass in the flask is 1 kg. How much (or what fraction) is exactly 1 cm from the bottom? 0! Multiply density by volume for mass (or fraction, or probability).

“Normalization” Scale so that total (integral of) 2 volume = 1

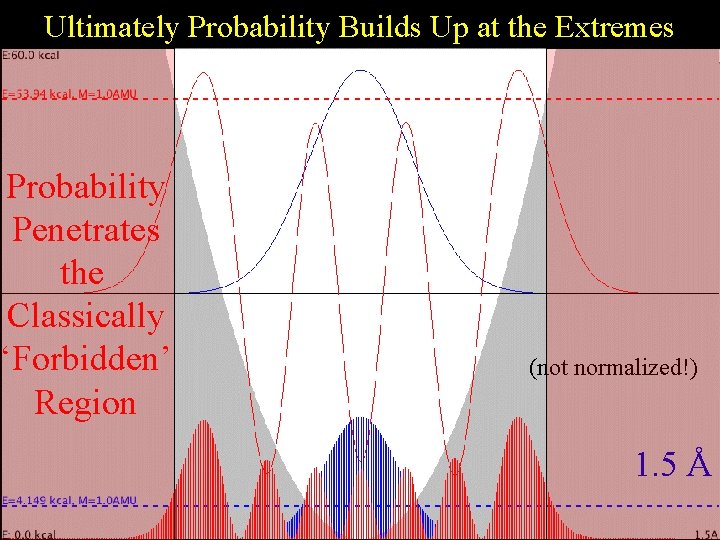
Ultimately Probability Builds Up at the Extremes Harmonic Probability Penetrates the Classically ‘Forbidden’ Region (not normalized!) 1. 5 Å

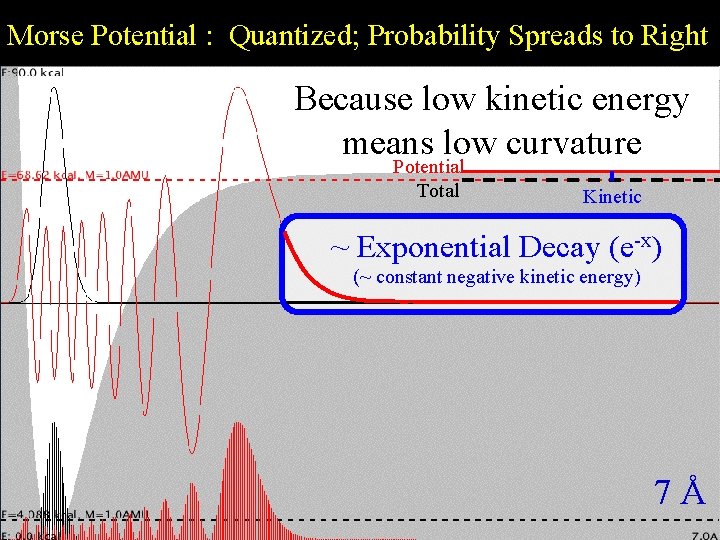
Morse Potential : Quantized; Probability Spreads to Right Morse. Because Quantization low kinetic energy means low curvature Potential Total Kinetic ~ Exponential Decay (e-x) (~ constant negative kinetic energy) 7Å

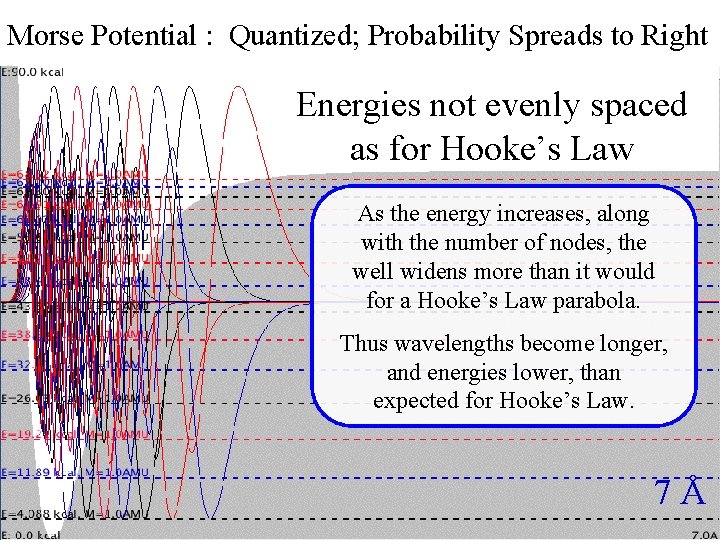
Morse Potential : Quantized; Probability Spreads to Right Morse. Energies Quantization not evenly spaced as for Hooke’s Law As the energy increases, along with the number of nodes, the well widens more than it would for a Hooke’s Law parabola. Thus wavelengths become longer, and energies lower, than expected for Hooke’s Law. 7Å

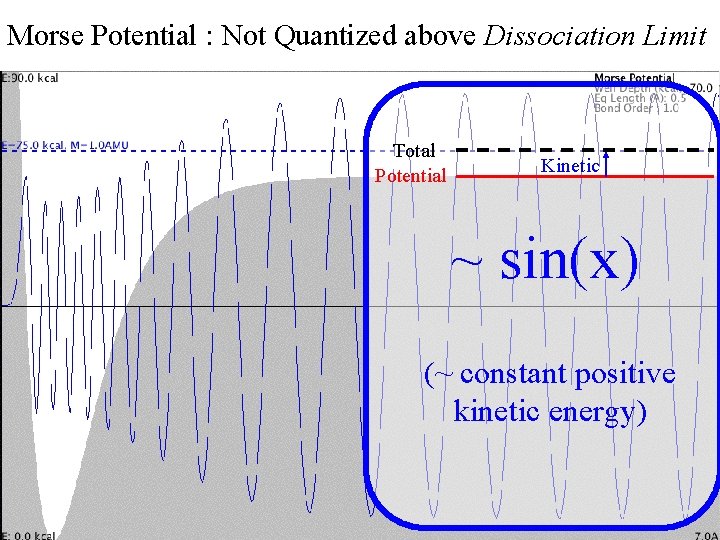
Morse Potential : Not Quantized above Dissociation Limit Morse Continuum Total Potential Kinetic ~ sin(x) (~ constant positive kinetic energy)

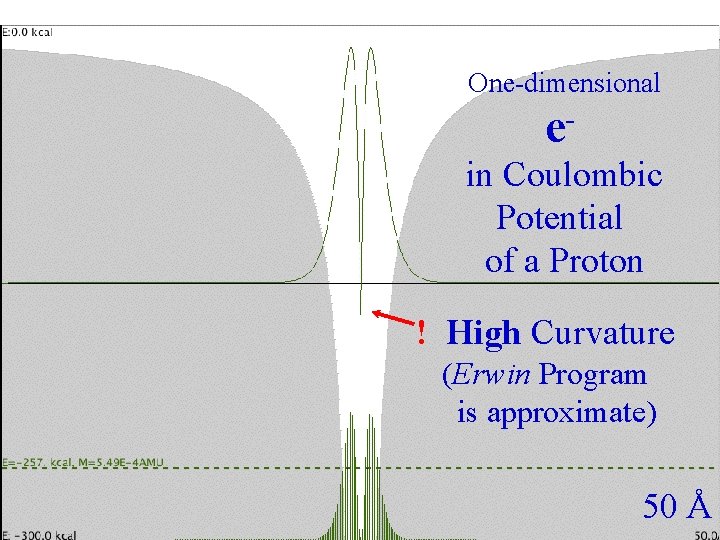
Coulombic One-dimensional Spacing e in Coulombic Potential of a Proton ! High Curvature (Erwin Program is approximate) 50 Å

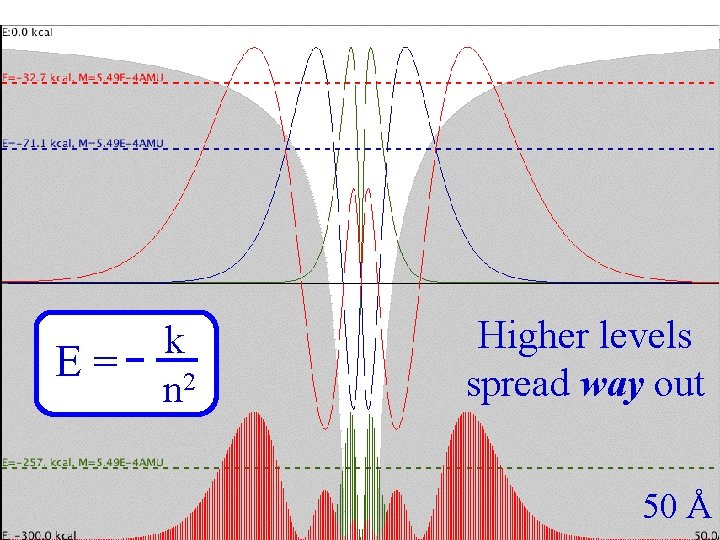
Coulomb Three E= k n 2 Higher levels spread way out 50 Å

Reward for Finding Knowledge of Everything e. g. Allowed Energies Structure Dynamics Bonding Reactivity

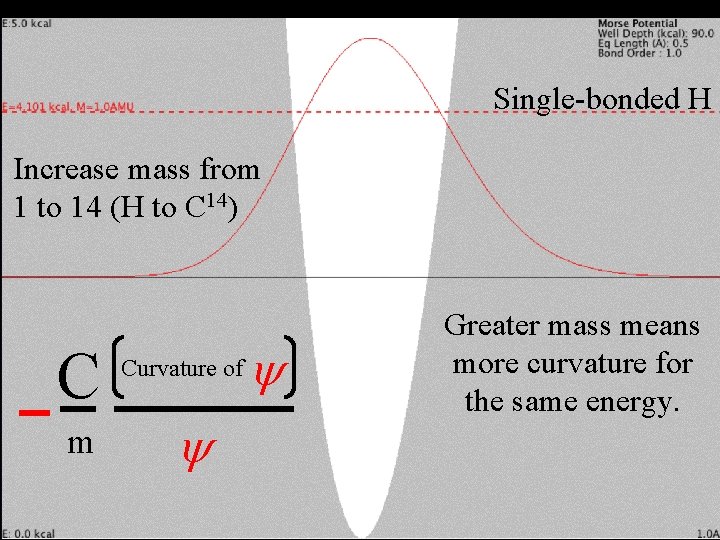
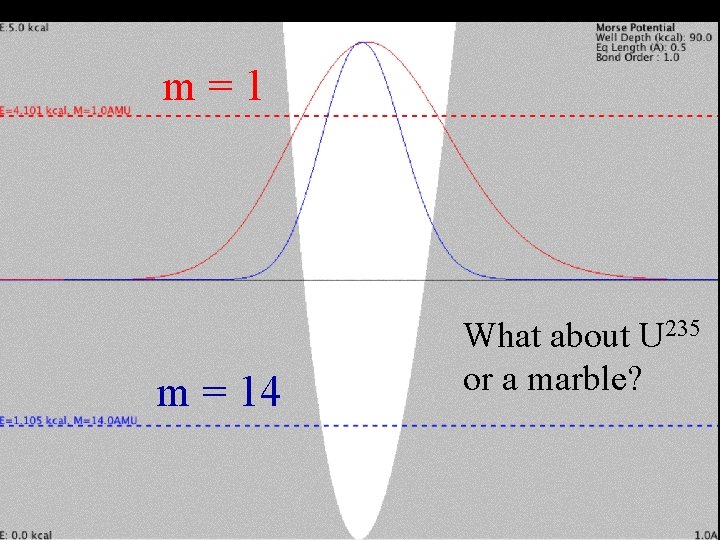
Change mass Single-bonded H Increase mass from 1 to 14 (H to C 14) C m Curvature of y y Greater mass means more curvature for the same energy.

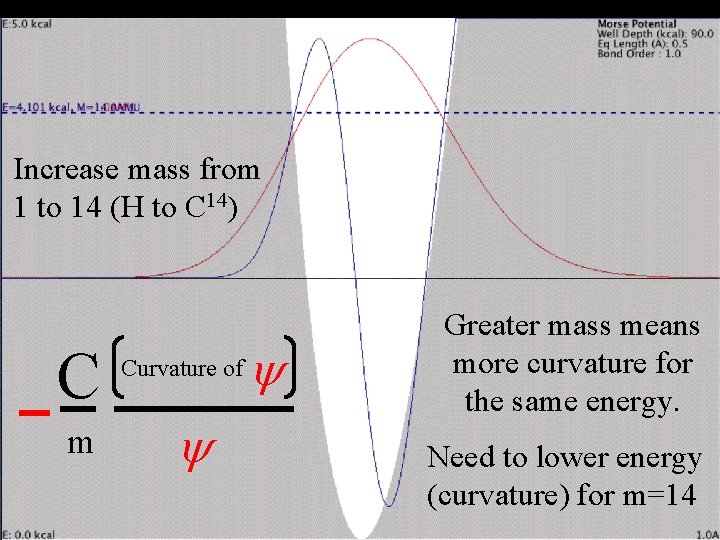
Change mass Single-bonded H Increase mass from 1 to 14 (H to C 14) C m Curvature of y y Greater mass means more curvature for the same energy. Need to lower energy (curvature) for m=14

m = 1 Mass Effect m = 14 What about U 235 or a marble?

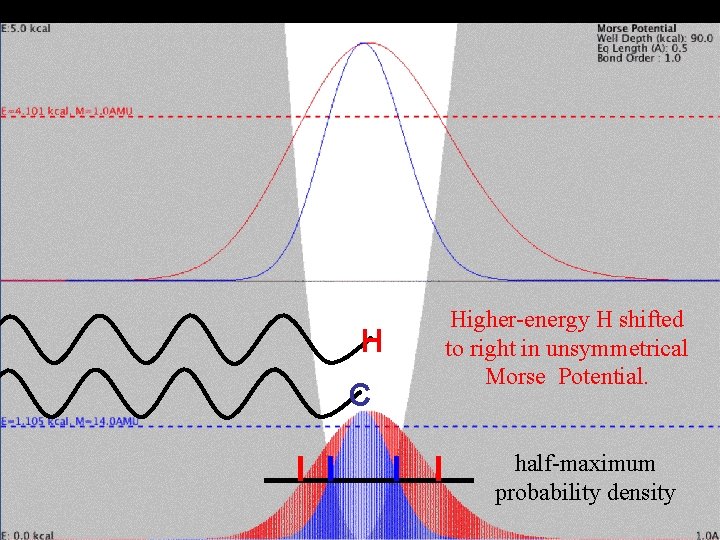
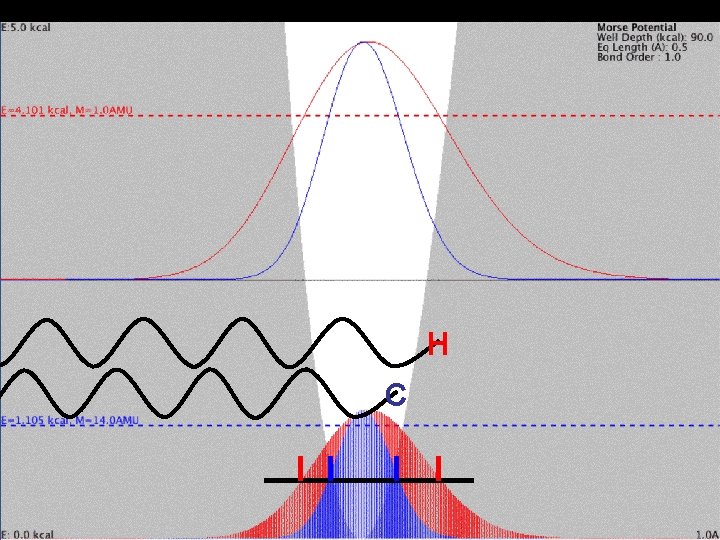
Mass Effect and Vibration H C Higher-energy H shifted to right in unsymmetrical Morse Potential. half-maximum probability density

Mass Effect and Vibration H C

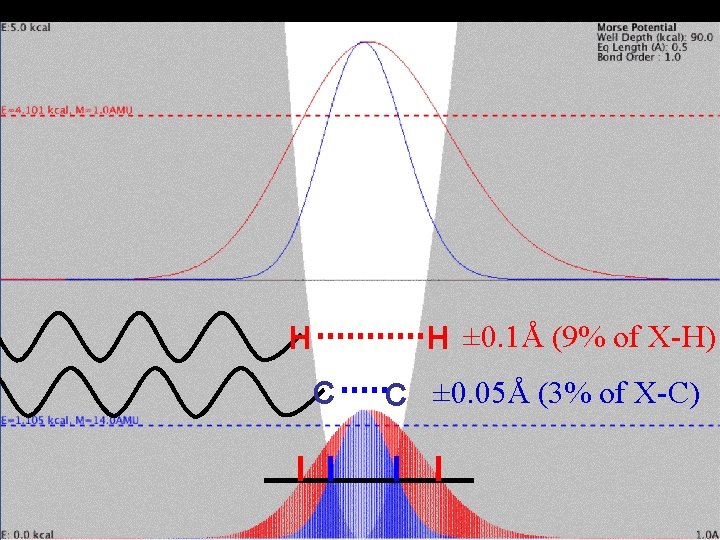
Mass Effect and Vibration H ± 0. 1Å (9% of X-H) H C C ± 0. 05Å (3% of X-C)

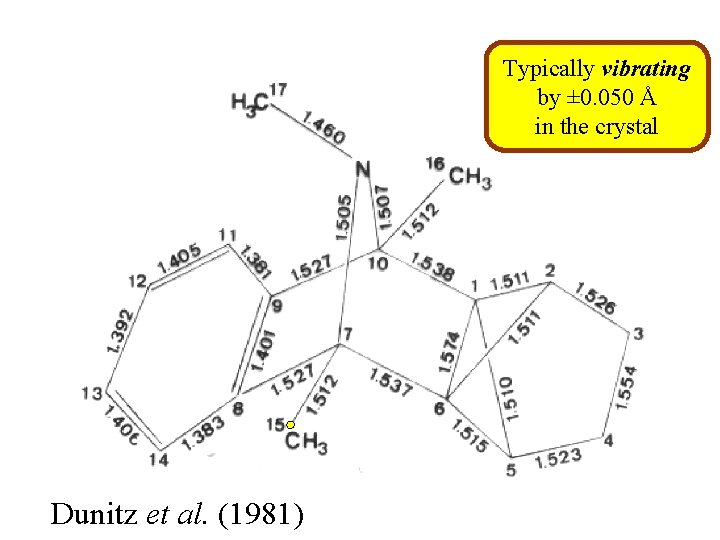
Typically vibrating by ± 0. 050 Å in the crystal Dunitz et al. (1981)

End of Lecture 8 Sept 19, 2008 Copyright © J. M. Mc. Bride 2009. Some rights reserved. Except for cited third-party materials, and those used by visiting speakers, all content is licensed under a Creative Commons License (Attribution-Non. Commercial-Share. Alike 3. 0). Use of this content constitutes your acceptance of the noted license and the terms and conditions of use. Materials from Wikimedia Commons are denoted by the symbol . Third party materials may be subject to additional intellectual property notices, information, or restrictions. The following attribution may be used when reusing material that is not identified as third-party content: J. M. Mc. Bride, Chem 125. License: Creative Commons BY-NC-SA 3. 0