Chemistry 125 Lecture 73 April 28 2010 Benzoin

Chemistry 125: Lecture 73 April 28, 2010 Benzoin, Claisen, Robinson (Ch. 19) Two Un-Natural Products This For copyright notice see final page of this file
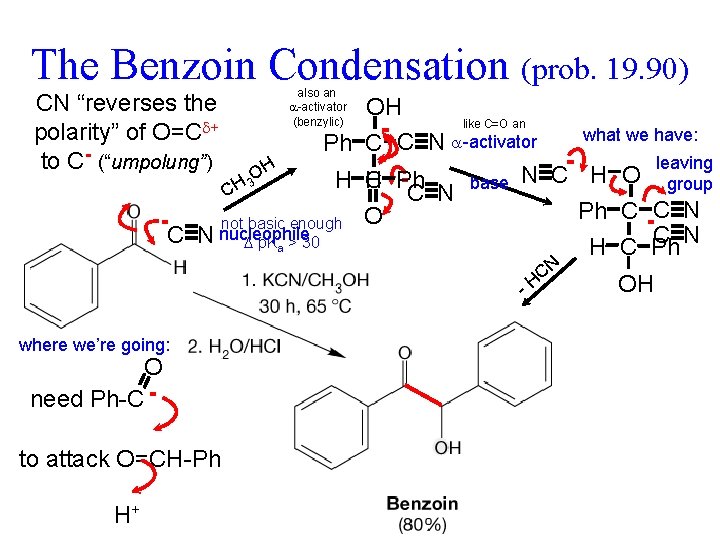
The Benzoin Condensation (prob. 19. 90) CN “reverses the polarity” of O=C + to C- (“umpolung”) also an -activator (benzylic) OH like C=O an what we have: Ph C C N -activator leaving H O N C H O H C 3 H Ph group CH C N base Ph C C N not basic enough O C N nucleophile C N p. Ka > 30 H C Ph - where we’re going: O need Ph-C to attack O=CH-Ph H+ N C H OH
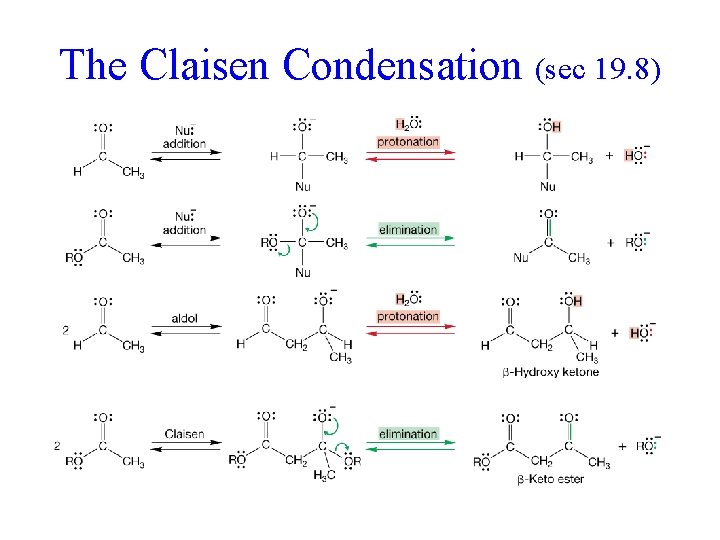
The Claisen Condensation (sec 19. 8)
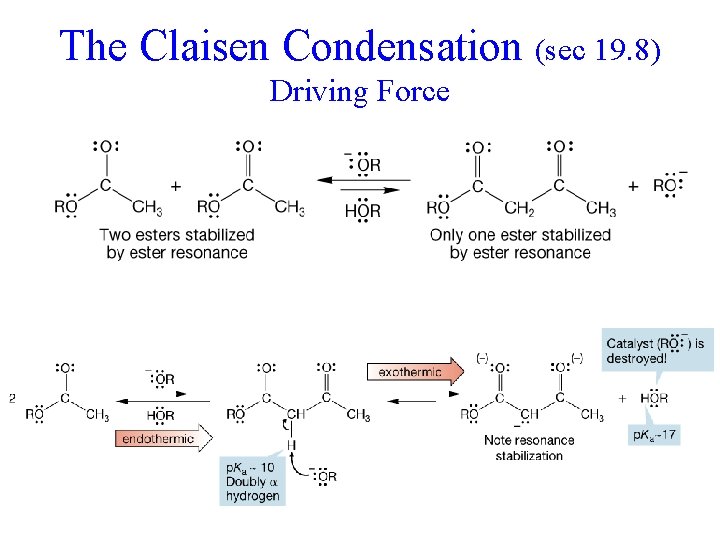
The Claisen Condensation (sec 19. 8) Driving Force
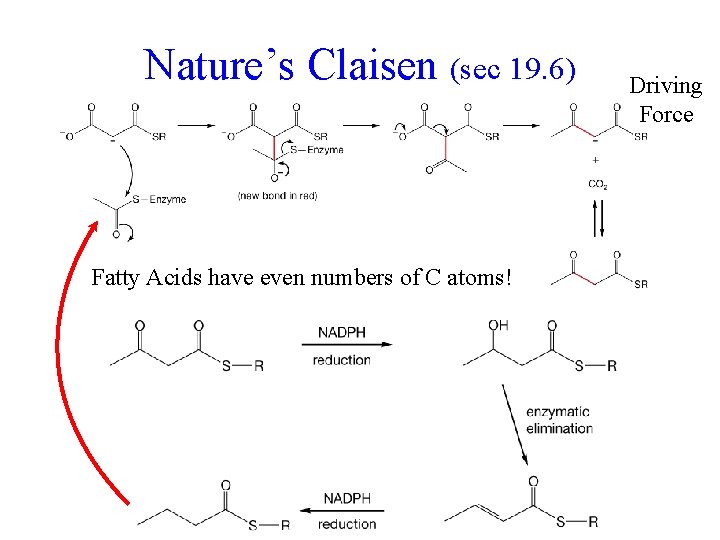
Nature’s Claisen (sec 19. 6) Fatty Acids have even numbers of C atoms! Driving Force
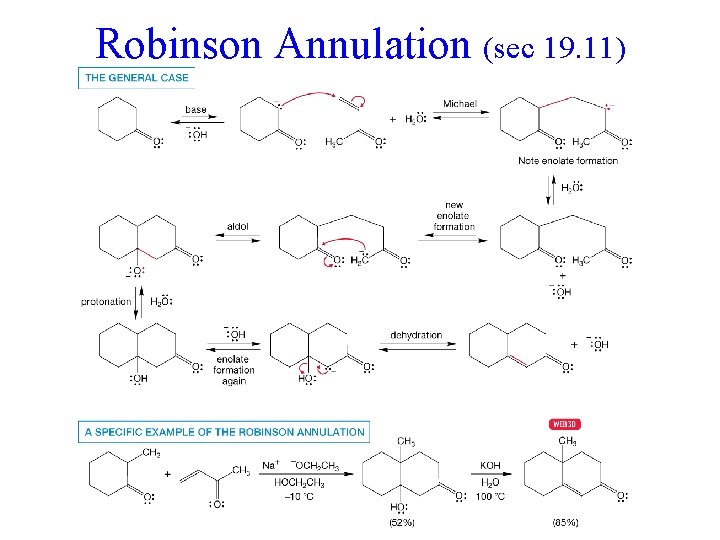
Robinson Annulation (sec 19. 11)

REVIEW 1: The Synthesis of Two Unnatural Products (in order to settle a question in theory of organic chemistry)
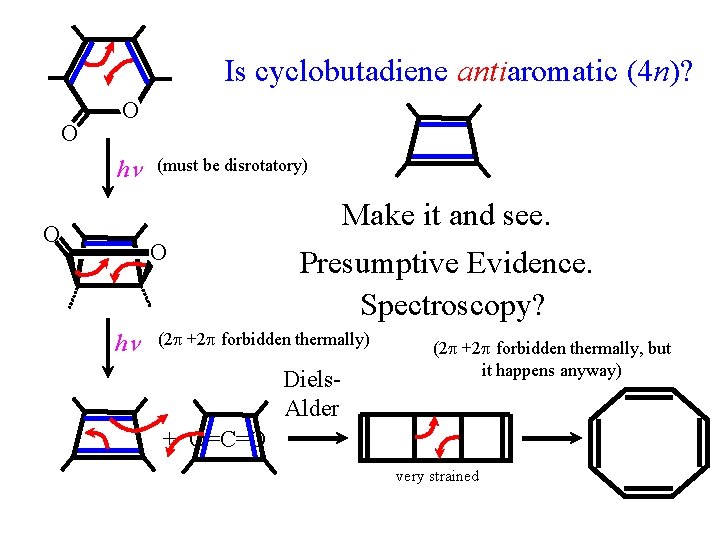
Is cyclobutadiene antiaromatic (4 n)? O O h (must be disrotatory) Make it and see. O O h Presumptive Evidence. Spectroscopy? (2 +2 forbidden thermally) Diels. Alder (2 +2 forbidden thermally, but it happens anyway) + O=C=O very strained
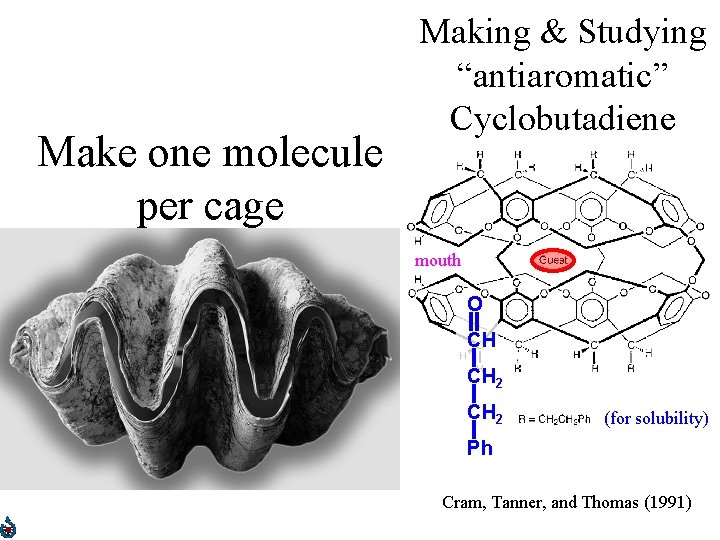
Make one molecule per cage Making & Studying “antiaromatic” Cyclobutadiene mouth O CH CH 2 (for solubility) Ph Cram, Tanner, and Thomas (1991)
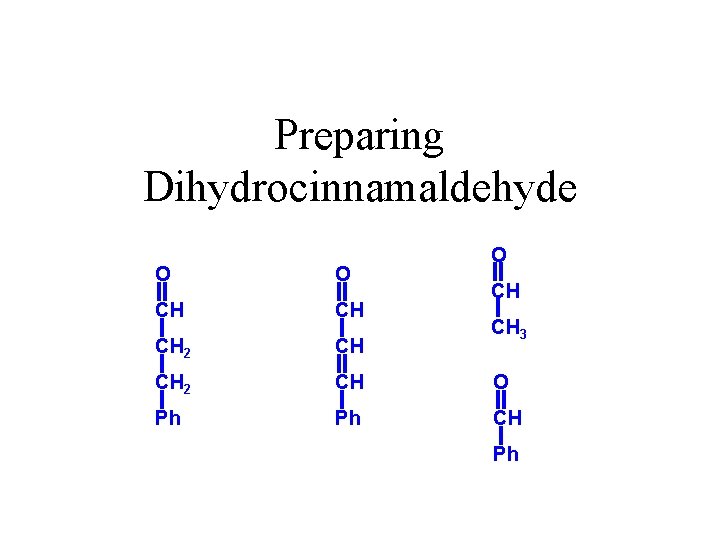
Preparing Dihydrocinnamaldehyde O O O CH CH CH 2 CH O Ph Ph CH CH CH 3 Ph
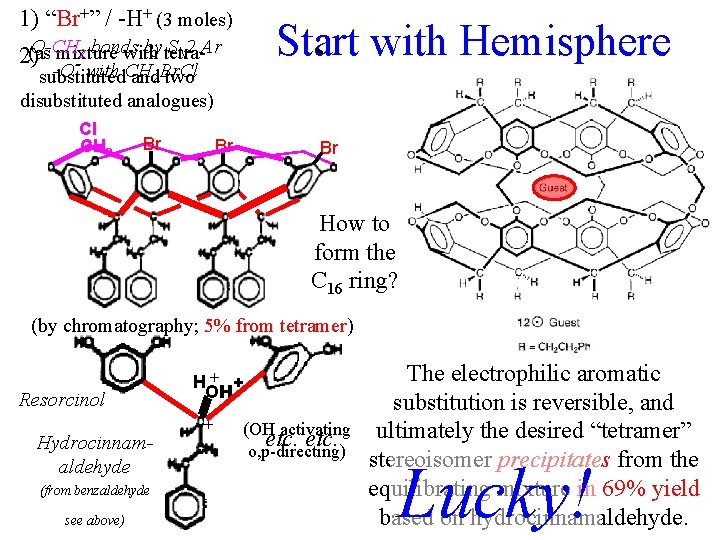
1) “Br+” / -H+ (3 moles) Start with Hemisphere O-CH by tetra. SN 2 Ar 2 bonds mixture with 2)(as -O with CH substituted and 2 Br. Cl two disubstituted analogues) - Cl Br. CH 2 - Br Br Br How to form the C 16 ring? (by chromatography; 5% from tetramer) Resorcinol Hydrocinnamaldehyde (from benzaldehyde see above) H+ + H OH + (OH activating etc. o, p-directing) The electrophilic aromatic substitution is reversible, and ultimately the desired “tetramer” stereoisomer precipitates from the equilibrating mixture in 69% yield based on hydrocinnamaldehyde. Lucky!
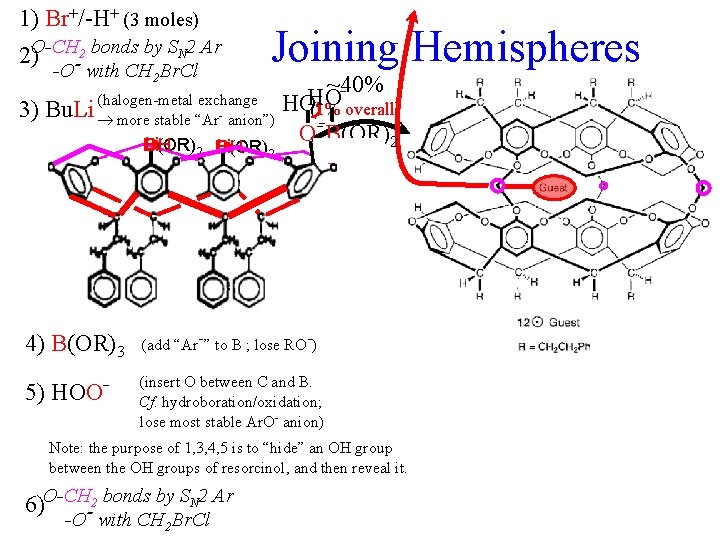
1) Br+/-H+ (3 moles) 2)O-CH-2 bonds by SN 2 Ar -O with CH 2 Br. Cl 3) Joining Hemispheres exchange Bu. Li (halogen-metal more stable “Ar- anion”) - - Li Br OH B(OR)2 Br Li OH B(OR)2 ~40% HO HO(1% overall) -O O-B(OR) 2 Li Br OH B(OR)2 4) B(OR)3 (add “Ar- ” to B ; lose RO-) 5) HOO- (insert O between C and B. Cf. hydroboration/oxidation; lose most stable Ar. O- anion) Note: the purpose of 1, 3, 4, 5 is to “hide” an OH group between the OH groups of resorcinol, and then reveal it. 6)O-CH-2 bonds by SN 2 Ar -O with CH 2 Br. Cl
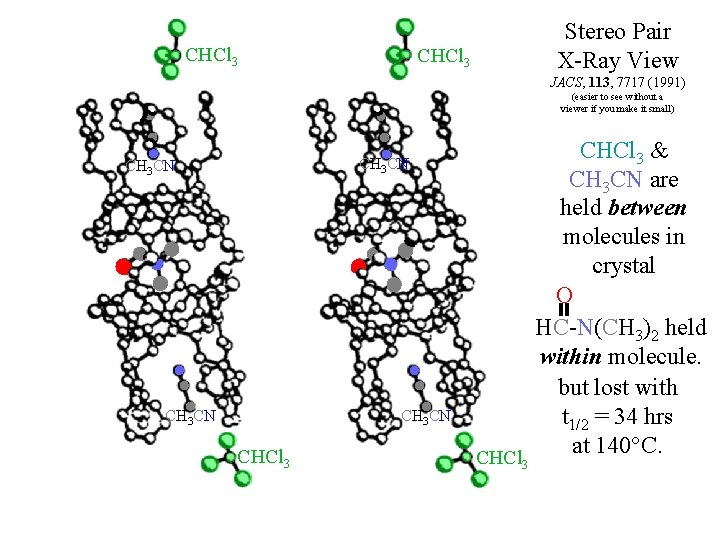
CHCl 3 Stereo Pair X-Ray View JACS, 113, 7717 (1991) (easier to see without a viewer if you make it small) CH 3 CN CHCl 3 & CH 3 CN are held between molecules in crystal O HC-N(CH 3)2 held within molecule. but lost with t 1/2 = 34 hrs at 140°C. CHCl 3
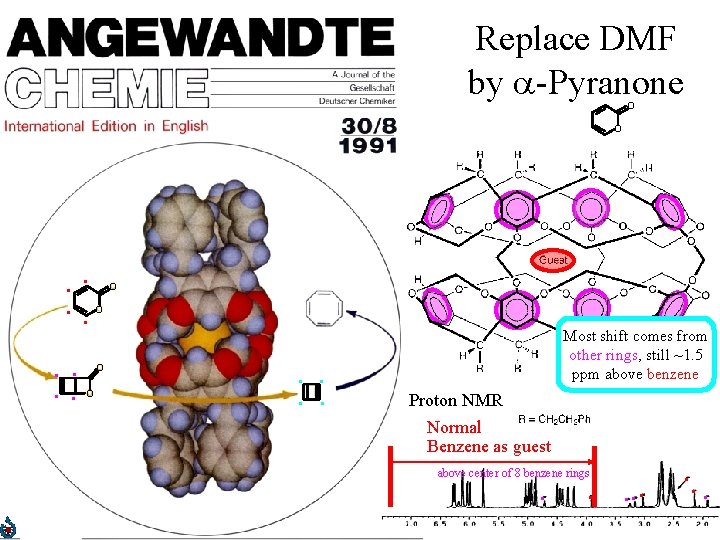
Replace DMF by -Pyranone O O . . O. . O O . . Most shift comes from Antiaromatic other rings, still ~1. 5 upfield ppm above shift? benzene . Proton NMR Normal Benzene as guest above center of 8 benzene rings . . . .

End of Lecture 73 April 28, 2010 Copyright © J. M. Mc. Bride 2010. Some rights reserved. Except for cited third-party materials, and those used by visiting speakers, all content is licensed under a Creative Commons License (Attribution-Non. Commercial-Share. Alike 3. 0). Use of this content constitutes your acceptance of the noted license and the terms and conditions of use. Materials from Wikimedia Commons are denoted by the symbol . Third party materials may be subject to additional intellectual property notices, information, or restrictions. The following attribution may be used when reusing material that is not identified as third-party content: J. M. Mc. Bride, Chem 125. License: Creative Commons BY-NC-SA 3. 0

These last six frames are left over from a previous year (and a different textbook). Instead of discarding them, I left them here in case they might be useful as you review.
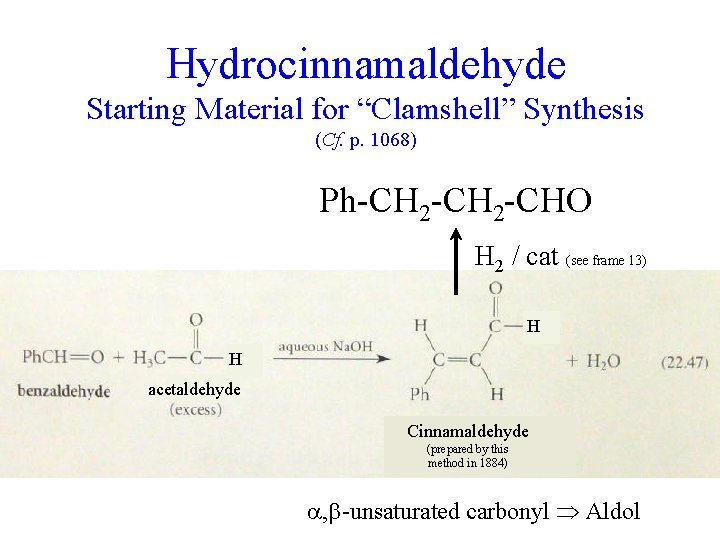
Hydrocinnamaldehyde Starting Material for “Clamshell” Synthesis (Cf. p. 1068) Ph-CH 2 -CHO H 2 / cat (see frame 13) H H acetaldehyde Cinnamaldehyde (prepared by this method in 1884) , -unsaturated carbonyl Aldol
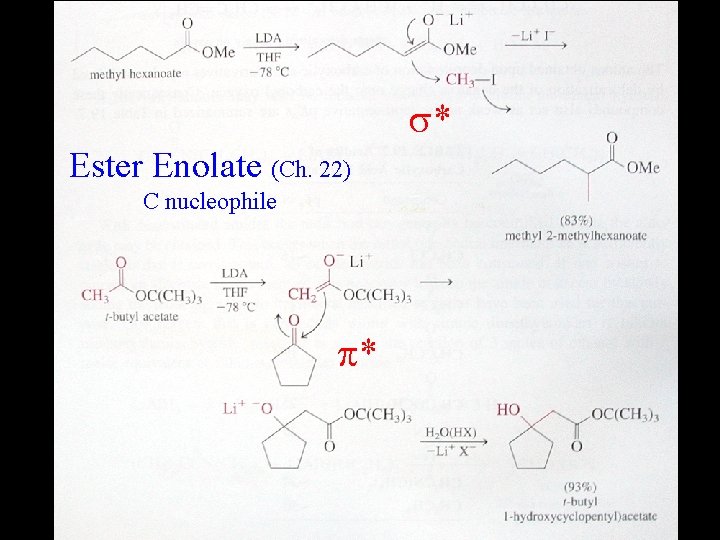
* Ester Enolate (Ch. 22) C nucleophile *

“Claisen” or “Acetoacetic Ester” Condensation (pp. 1072 -1075) Why not use OH-? substitution at ester Equilibrium position?
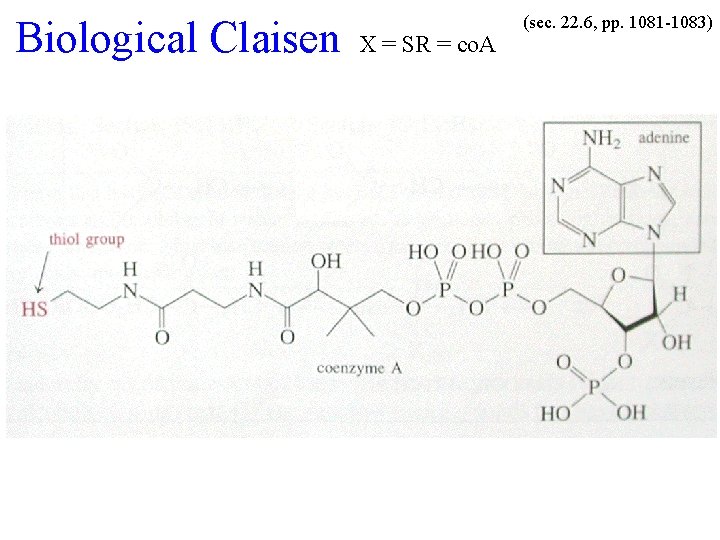
Biological Claisen X = SR = co. A (sec. 22. 6, pp. 1081 -1083)
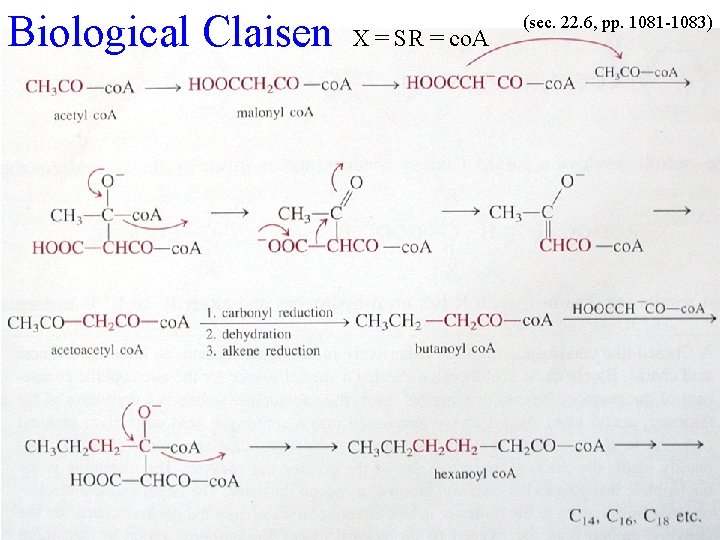
Biological Claisen X = SR = co. A (sec. 22. 6, pp. 1081 -1083)
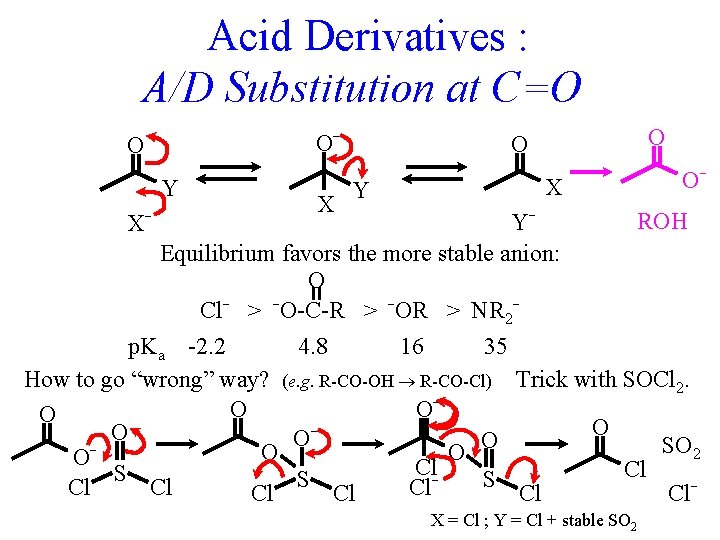
Acid Derivatives : A/D Substitution at C=O O- O Y X O- ROH YEquilibrium favors the more stable anion: O Cl- > -O-C-R > -OR > NR 2 p. Ka -2. 2 4. 8 16 35 How to go “wrong” way? (e. g. R-CO-OH R-CO-Cl) Trick with SOCl 2. O O O O SO 2 O O OCl. Cl S S S Cl Cl. X- X = Cl ; Y = Cl + stable SO 2
- Slides: 22