Chemistry 125 Lecture 68 April 14 2010 Mitsunobu

Chemistry 125: Lecture 68 April 14, 2010 Mitsunobu Reaction Acids and Acid Derivatives This For copyright notice see final page of this file

Teleology Lectures 69 -70 (4/16, 19) Topics from Chapters 18 -19 - Acid Derivatives and Condensations Lecture 71 (4/21) Topics from Ch. 22 - Carbohydrates Lecture 72 (4/23, 26? ) guest lecture(s) by Prof. Ziegler Carbohydrates - Fischer's Proof of the Configuration of Glucose Lecture 73 (4/28) Synthesis of an Unnatural Product (Review) (Anti-Aromatic Cyclobutadiene in a Clamshell) Lecture 74 (4/30) Synthesis of a Natural Product (Review) (Woodward's Synthesis of Cortisone)

“Key green chemistry research areas - a perspective from pharmaceutical manufacturers” Green Chemistry, 2007, 9, 411 -420 Table 1 Reactions companies use now but would strongly prefer better reagents Research Area Number of roundtable companies voting for this research area as a priority area Amide formation avoiding poor atom economy reagents 6 votes OH activation for nucleophilic substitution 5 votes Reduction of amides without hydride reagents 4 votes Oxidation/Epoxidation methods without the use of chlorinated solvents 4 votes Safer and more environmentally friendly Mitsunobu reactions 3 votes Friedel-Crafts reaction on unactivated systems 2 votes Nitrations 2 votes
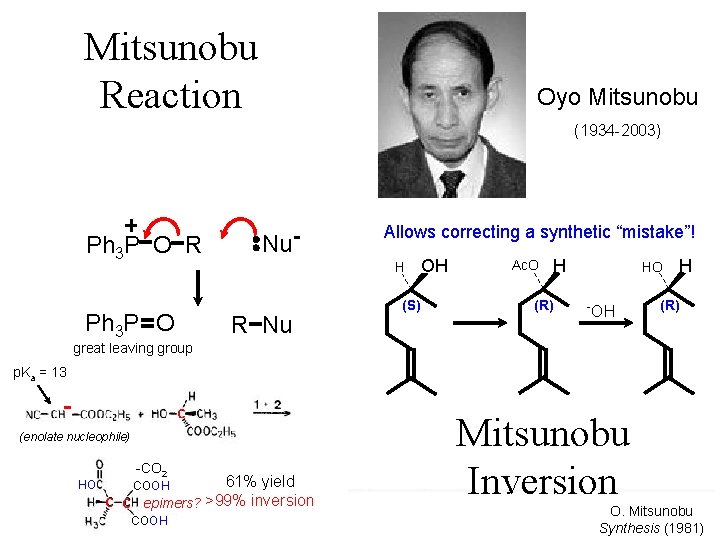
Mitsunobu Reaction Oyo Mitsunobu (1934 -2003) Ph 3 P O R Ph 3 P O Nu. R Nu p. Ka = 13 C (enolate nucleophile) HO 61% yield C epimers? >99% inversion COOH C C COOH Very general for Ac. O H OH acidic Nu-H H great leaving group -CO 2 Allows correcting a synthetic “mistake”! (S) (p. Ka <(R)15) HO -OH H (R) e. g. R-CO 2(RO)2 PO 2 - Mitsunobu N “active methylene compounds” Inversion (RCO)2 N 3 - O. Mitsunobu Synthesis (1981)
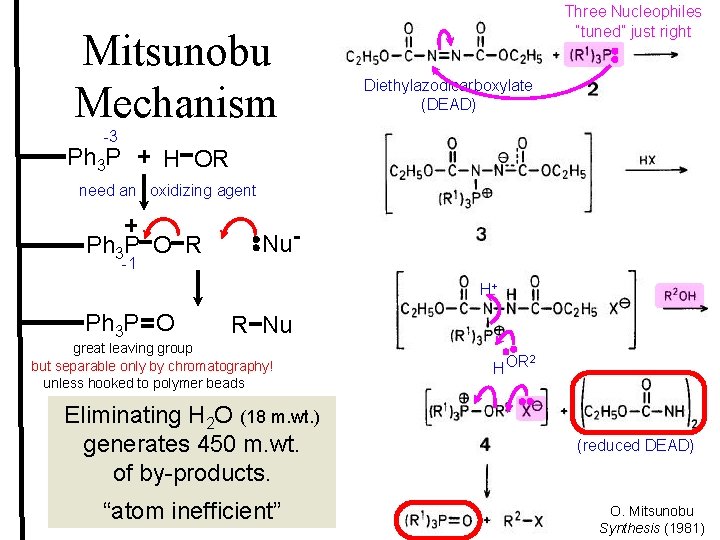
Mitsunobu Mechanism -3 Ph 3 P Three Nucleophiles “tuned” just right Diethylazodicarboxylate (DEAD) H OR need an oxidizing agent Ph 3 P O R -1 Nu. H+ Ph 3 P O R Nu great leaving group but separable only by chromatography! unless hooked to polymer beads Eliminating H 2 O (18 m. wt. ) generates 450 m. wt. of by-products. “atom inefficient” HOR 2 (reduced DEAD) O. Mitsunobu Synthesis (1981)

Acidity of RCO 2 H (p. 836) Making RCO 2 H by Oxidation and Reduction (sec. 17. 6)
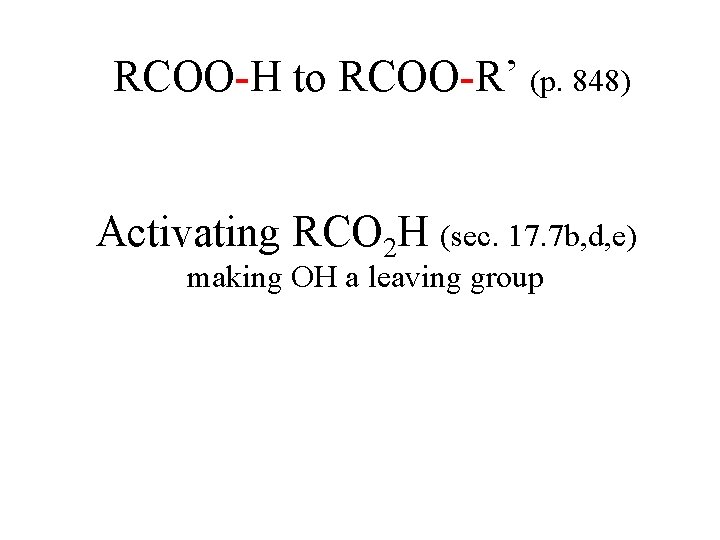
RCOO-H to RCOO-R’ (p. 848) Activating RCO 2 H (sec. 17. 7 b, d, e) making OH a leaving group
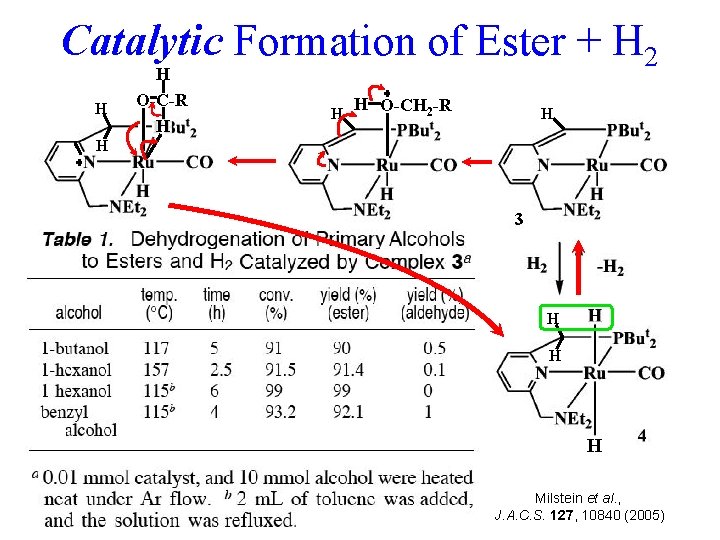
Catalytic Formation of Ester + H 2 H H O-C-R H H H O-CH 2 -R H H 3 Another oxidation involving removal of an H from RCHO and one from another RCH 2 OH, plus C-O coupling, completes 2 R-CH 2 -OH R-CO 2 -CH 2 R + 2 H 2 with no other activation! GREEN H H H Milstein et al. , J. A. C. S. 127, 10840 (2005)

Catalytic Formation of Ester + H 2 Thermochemistry of 2 Et. OH Ac. OEt + 2 H 2 Hf HOEt x 2 -66. 1± 0. 5 -132. 2± 1. 0 Ac. OEt -114. 8± 0. 2 H 2 0 Hrxn 17. 4 endothermic! K 3/2 Rm. T 10 -1/2 17. 4 10 -9 need p. H 2 > 10 -9 atm Milstein et al. , J. A. C. S. 127, 10840 (2005)
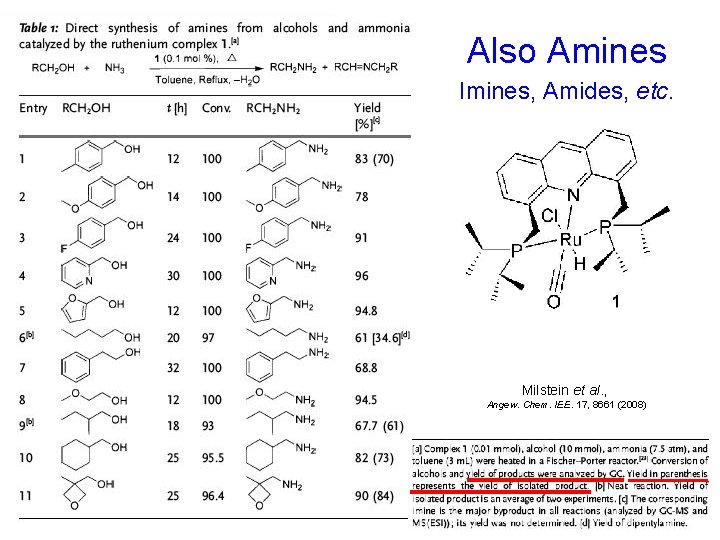
Also Amines Imines, Amides, etc. Milstein et al. , Angew. Chem. IEE. 17, 8661 (2008)

Benzoic Acid O 2 Oil of Bitter Almonds

Air Oxidation of Benzaldehyde Cf. sec. 18. 12 a

R-Li & Li. Al. H 4 (sec. 17. 7 f) stop at C=O?

End of Lecture 68 April 14, 2010 Copyright © J. M. Mc. Bride 2010. Some rights reserved. Except for cited third-party materials, and those used by visiting speakers, all content is licensed under a Creative Commons License (Attribution-Non. Commercial-Share. Alike 3. 0). Use of this content constitutes your acceptance of the noted license and the terms and conditions of use. Materials from Wikimedia Commons are denoted by the symbol . Third party materials may be subject to additional intellectual property notices, information, or restrictions. The following attribution may be used when reusing material that is not identified as third-party content: J. M. Mc. Bride, Chem 125. License: Creative Commons BY-NC-SA 3. 0
- Slides: 14