Chemistry 125 Lecture 52 February 17 2010 This

Chemistry 125: Lecture 52 February 17, 2010 This Additions by Radicals & Electrophilic Carbon; Isoprenoids; and Polymer Properties For copyright notice see final page of this file
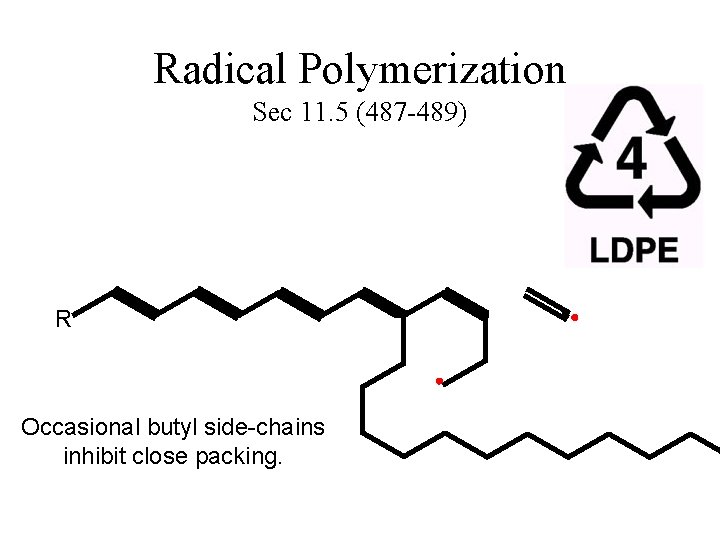
Radical Polymerization Sec 11. 5 (487 -489) R H Occasional butyl side-chains inhibit close packing.
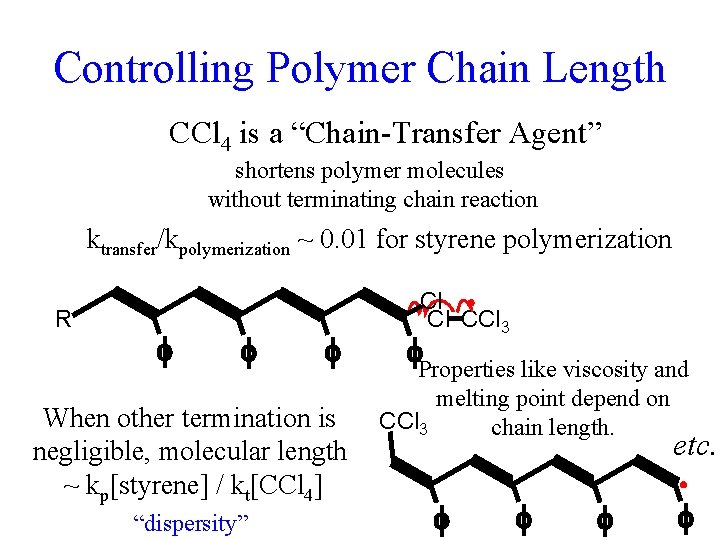
Controlling Polymer Chain Length CCl 4 is a “Chain-Transfer Agent” shortens polymer molecules without terminating chain reaction ktransfer/kpolymerization ~ 0. 01 for styrene polymerization R When other termination is negligible, molecular length ~ kp[styrene] / kt[CCl 4] “dispersity” Cl Cl CCl 3 Properties like viscosity and melting point depend on CCl 3 chain length. etc.

(“oligo”, a few) Alkene Oligomerization and Polymerization Using Carbon Electrophiles R-L (SN 2) + R (SN 1) *
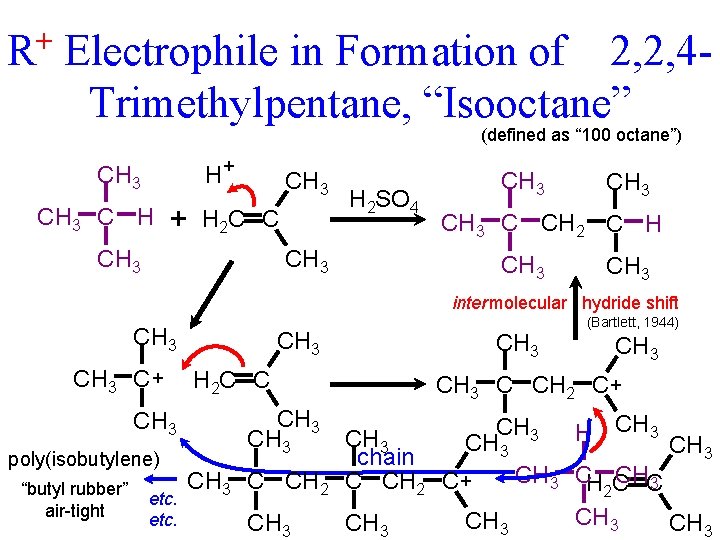
+ R Electrophile in Formation of 2, 2, 4 Trimethylpentane, “Isooctane” (defined as “ 100 octane”) H+ CH 3 C H + CH 3 H 2 C C CH 3 H 2 SO 4 CH 3 C CH 2 C H CH 3 inter molecular hydride shift CH 3 C + CH 3 poly(isobutylene) “butyl rubber” air-tight etc. CH 3 H 2 C C CH 3 C CH 2 CH 3 (Bartlett, 1944) CH 3 C CH 2 C+ CH 3 H CH 3 chain CH 3 CH C CHC C CH 2 C+ 3 2 CH 3

R-L and * + R Electrophiles in Terpene/Steroid Biogenesis Sec. 12. 13 pp. 554 -562
![Isopentenyl Pyrophosphate krel for rxn with I- in acetone n-propyl allyl benzyl Cl [1] Isopentenyl Pyrophosphate krel for rxn with I- in acetone n-propyl allyl benzyl Cl [1]](http://slidetodoc.com/presentation_image_h2/aabbfbd4bb14751e2b381547d90137ff/image-7.jpg)
Isopentenyl Pyrophosphate krel for rxn with I- in acetone n-propyl allyl benzyl Cl [1] Cl 90 Cl 250 I Dimethylallyl Pyrophosphate Cl Adjacent unsaturation apparently speeds SN 2 (as well as SN 1)
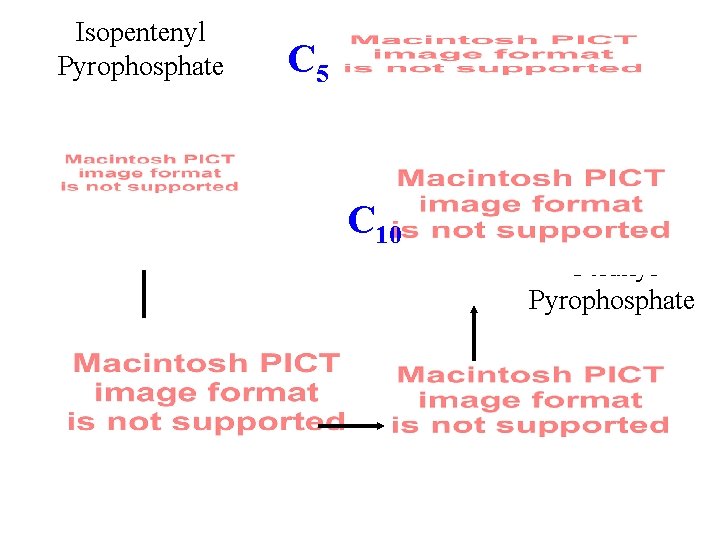
Isopentenyl Pyrophosphate C 5 C 10 Geranyl Pyrophosphate
![Geranyl Pyrophosphate trans cis Neryl Pyrophosphate Camphor [Ox] +H 2 O Limonene -H+ anti-Markovnikov Geranyl Pyrophosphate trans cis Neryl Pyrophosphate Camphor [Ox] +H 2 O Limonene -H+ anti-Markovnikov](http://slidetodoc.com/presentation_image_h2/aabbfbd4bb14751e2b381547d90137ff/image-9.jpg)
Geranyl Pyrophosphate trans cis Neryl Pyrophosphate Camphor [Ox] +H 2 O Limonene -H+ anti-Markovnikov -H+ b-Pinene "Terpene" essential oils -H+ C 10
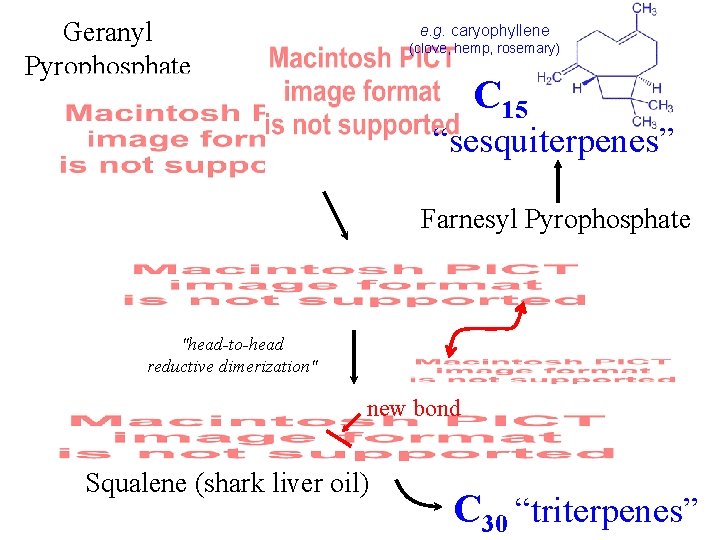
Geranyl Pyrophosphate e. g. caryophyllene (clove, hemp, rosemary) C 15 “sesquiterpenes” Farnesyl Pyrophosphate "head-to-head reductive dimerization" new bond Squalene (shark liver oil) C 30 “triterpenes”
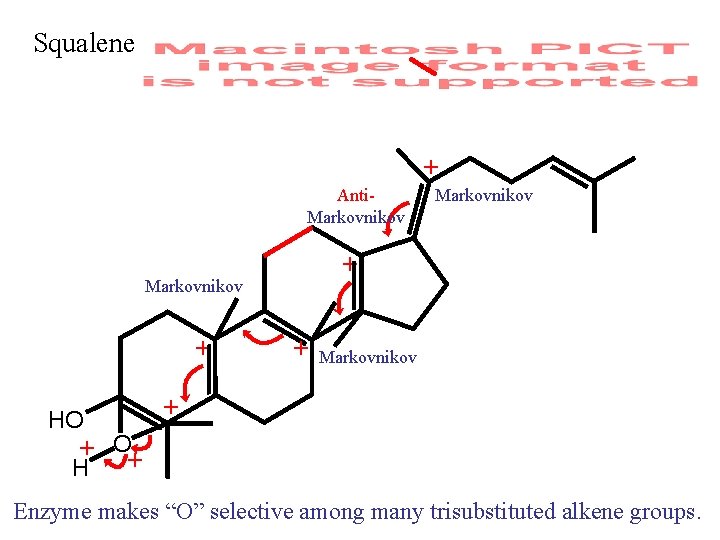
Squalene + Anti. Markovnikov + HO Markovnikov + + O+ H Enzyme makes “O” selective among many trisubstituted alkene groups.
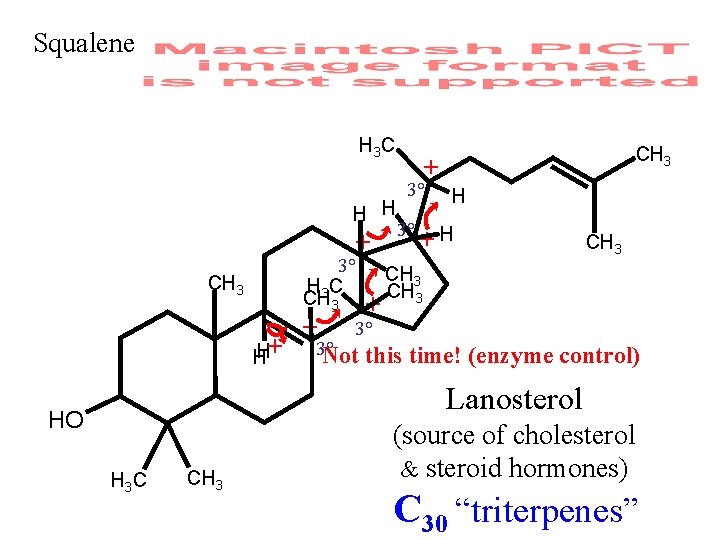
Squalene H 3 C H H + CH 3 H+ H CH 3 + 3° 3° H +H 3° CH 3 C CH 3 + + 3° 3° Not this time! (enzyme CH 3 control) Lanosterol HO H 3 C CH 3 (source of cholesterol & steroid hormones) C 30 “triterpenes”

Squalene H 3 C Cute Story H H + CH 3 + 3° 3° H +H 3° CH 3 C CH 3 + + 3° 3° Not this time! (enzyme CH 3 Is it True? CH 3 H+ H control) Lanosterol (Wait for NMR) HO H 3 C CH 3 (source of cholesterol & steroid hormones) C 30 “triterpenes”
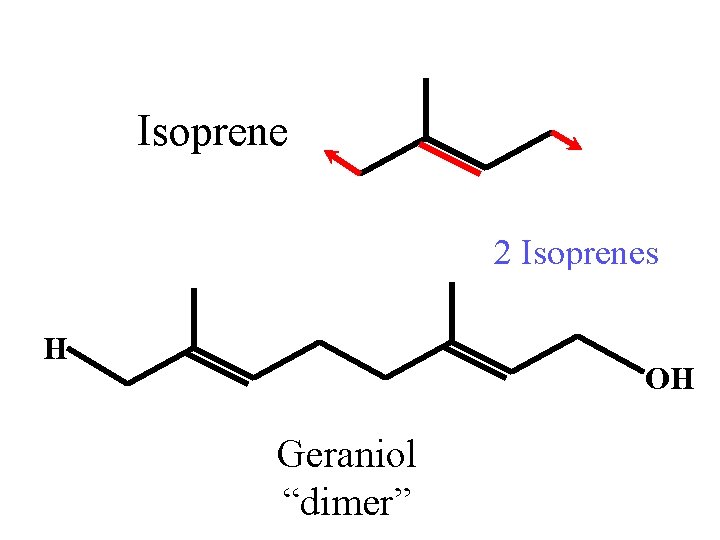
Isoprene 2 Isoprenes H OH Geraniol “dimer”
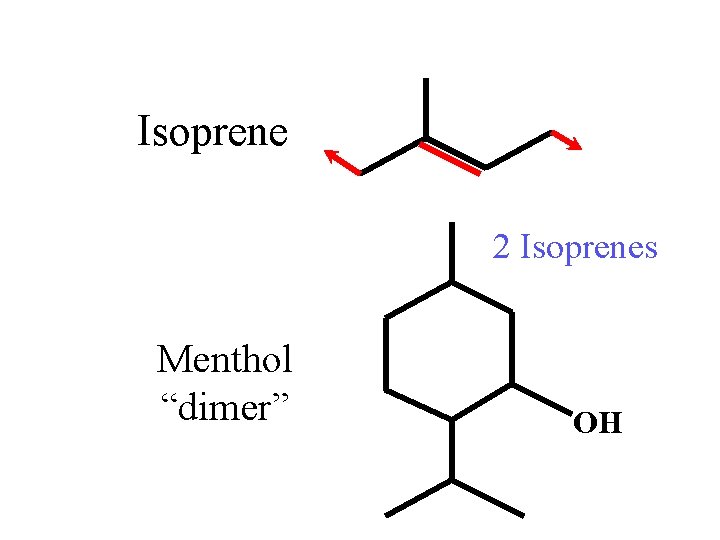
Isoprene 2 Isoprenes Menthol “dimer” OH
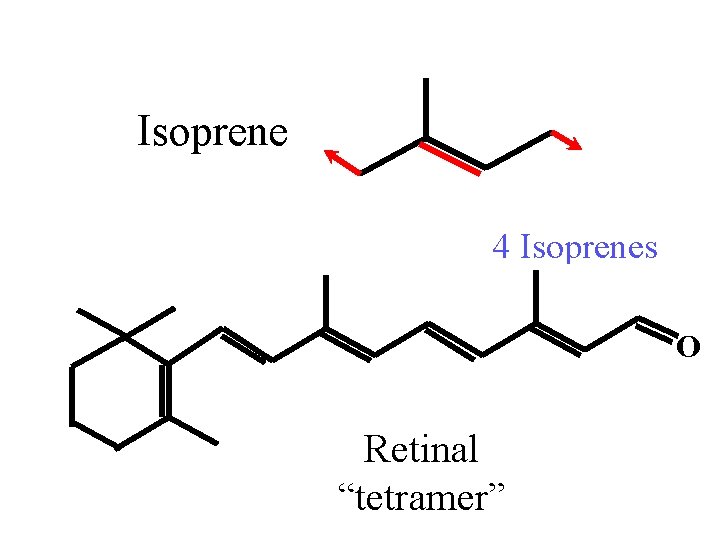
Isoprene 4 Isoprenes O Retinal “tetramer”
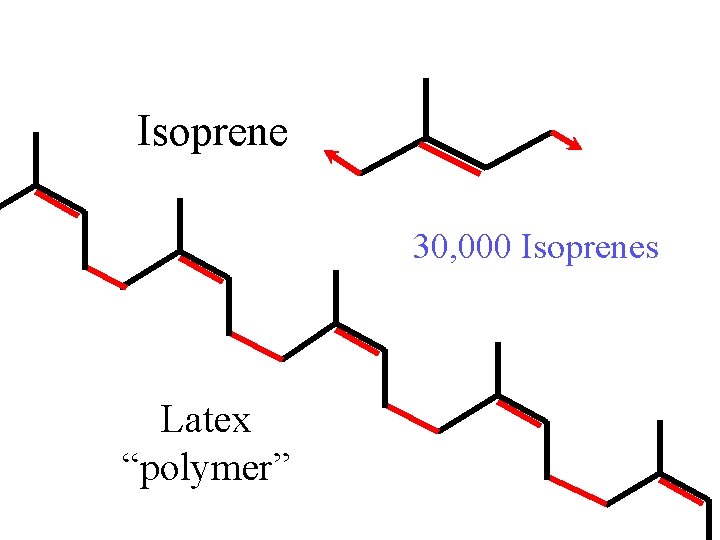
Isoprene 30, 000 Isoprenes Latex “polymer”

Hevea braziliensis

Latex to Caoutchouc

Thomas Hancock (England -1820) “Masticator” Charles Macintosh (Scotland - 1823) Sandwiched rubber between cloth layers for waterproof garments Gooey in heat Goodyear (1839) Brittle in cold Vulcanization

Discovery of Vulcanization 1839

Discovery of Vulcanization from Goodyear’s Autobiographical “Gum-Elastic” (1855) He endeavoured to call the attention of his The occurrence did not at the time seem to brother, as wellsurprised as some other individuals “He to find thatconsidered them to be worthy of notice; it was who were present, and whobrought were acquainted specimen, being carelessly into as one of the frequent appeals that he was in with thewith manufacture of gum-elastic, to this contact a hot stove, charred like leather. the habit of making, in behalf of some new effect, as remarkable, and unlike any before experiment. ” known, since gum-elastic always melted when exposed to a high degree of heat.

Silliman consult “Having seen experiments made, and also performed them myself, with the India rubber prepared by Mr. Charles Goodyear, I can state that it does not melt, but rather chars, by heat, and that it does not stiffen by cold, but retains its flexibility with cold, even when laid between cakes of ice. ” B. Silliman October 14, 1839


U. S. Pavilion Crystal Palace (1851)

India Rubber Desk Goodyear’s Vulcanite Court Mattatuck Museum, Waterbury
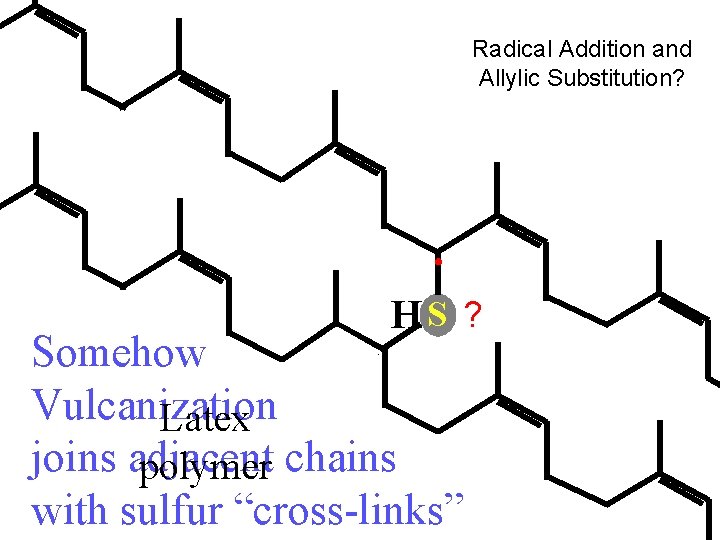
Radical Addition and Allylic Substitution? HS ? Somehow Vulcanization Latex joins adjacent polymer chains with sulfur “cross-links”

Vulcanization and the Physical Properties of Polymers

Gough

wordsworth (1757 -1825) “No floweret blooms Throughout the lofty range of these rough hills, Nor in the woods, that could from him conceal Its birth-place; none whose figure did not live Upon his touch. ” Wordsworth “Excursion” (1813)

Heating rubber makes it expand (more than H 2 O). John Gough


Heating tightly stretched rubber makes it contract!

If stretching rubber generates heat, what should letting it contract do? A) If heat comes from internal friction, B)contraction should also cause C)friction and generate heat. B) If heat comes from some other cause, contraction may do the opposite and absorb heat (“generate cold”).

Why?

Goodyear Plot

Goodyear Inventor

Goodyear to Gibbs

Gibbs Mathematical Physics

Gibbs to Onsager
Kirkwood & Onsager

Polymer Statistical Mechanics
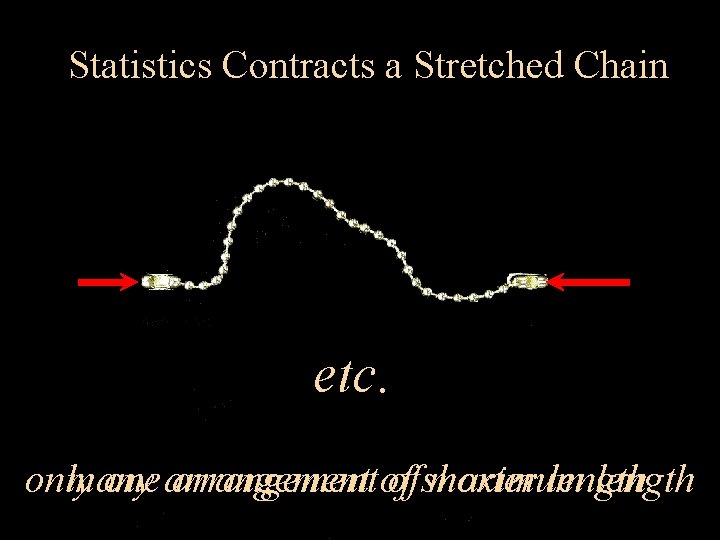
Statistics Contracts a Stretched Chain etc. only many one arrangement of of shorter maximum length

Fixed, Absorbing Warming irregular heat “melts” cross-links “melts” thethe crystalline between crystalline adjacent regions, chains Stretching prevents and allows crystallization statistics to (and makebrittleness) the materialincontract. the cold. Near maximum extension there is local Crystallization Contributes Rigidity Releases Heat

Lengthwise Motion by “Reptation” How to make a tangle flow? Change shape by snaking along a tunnel through the tangled neighbors.
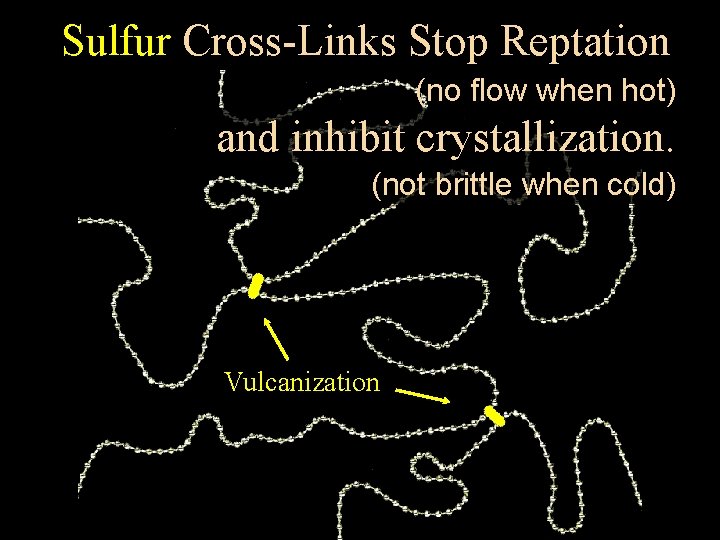
Sulfur Cross-Links Stop Reptation (no flow when hot) and inhibit crystallization. (not brittle when cold) Vulcanization

Vulcanization in the Home

End of Lecture 52 Feb. 17, 2010 Copyright © J. M. Mc. Bride 2010. Some rights reserved. Except for cited third-party materials, and those used by visiting speakers, all content is licensed under a Creative Commons License (Attribution-Non. Commercial-Share. Alike 3. 0). Use of this content constitutes your acceptance of the noted license and the terms and conditions of use. Materials from Wikimedia Commons are denoted by the symbol . Third party materials may be subject to additional intellectual property notices, information, or restrictions. The following attribution may be used when reusing material that is not identified as third-party content: J. M. Mc. Bride, Chem 125. License: Creative Commons BY-NC-SA 3. 0
- Slides: 48