Chemistry 125 Lecture 46 February 1 2010 E

Chemistry 125: Lecture 46 February 1, 2010 E 2, SN 1, E 1 This For copyright notice see final page of this file
![E 2 -Elimination Text sec. 7. 9 Rate influenced by: [base] attack occurs during E 2 -Elimination Text sec. 7. 9 Rate influenced by: [base] attack occurs during](http://slidetodoc.com/presentation_image_h2/b4911314cd1c43c6dce5e9b75fd3c72e/image-2.jpg)
E 2 -Elimination Text sec. 7. 9 Rate influenced by: [base] attack occurs during (or before) rate-determining-step nature of leaving group it leaves during (or before) rds H isotope (kinetic isotope effect) C-H broken during rds Heavier atom, lower ZPE see Lecture 8: frames 21 -22 ABN AON ABN k. H > k. D ZPE (kinetic) C H D but only. Fif bond CH 2 is weakened in rate-determining transition state CH 2 H : OH F CH 2 "E 2 Elimination" CCH 2 H O H D H OH D O

E 2 -Elimination Anti Stereochemistry sec 7. 9 c starting material is already eclipsed but not dogmatic Which How to should test experimentally? be better? Text sec. 7. 9 (S) (R) ABN (more strained) AON ABN R R R syn trans R F R R R anti cis R (anti hybrids overlap better) F CH 2 (S) H H OH : OH "E 2 Elimination"

E 2 -Elimination Text sec. 7. 9 Regiochemistry (Saytzeff/Hofmann) sec 7. 9 d “Saytzeff” “Hofmann”

E 2 -Elimination Text sec. 7. 9 E 2 vs. SN 2 (Sterics & Base Strength) sec 7. 9 b
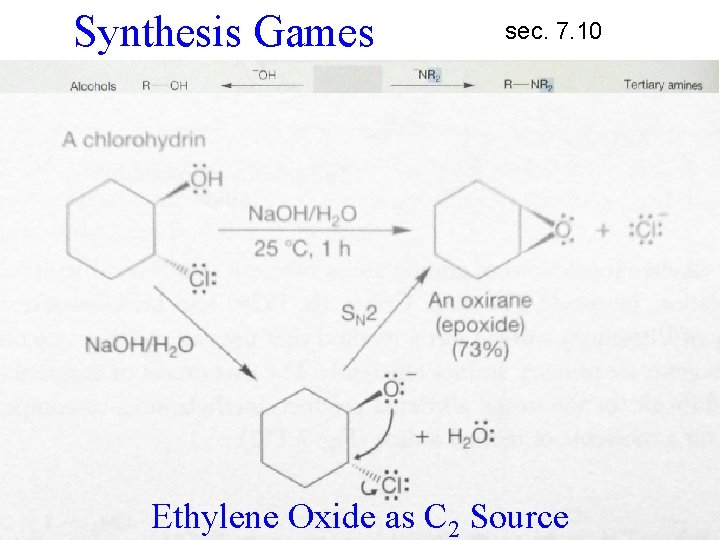
Synthesis Games sec. 7. 10 Ethylene Oxide as C 2 Source
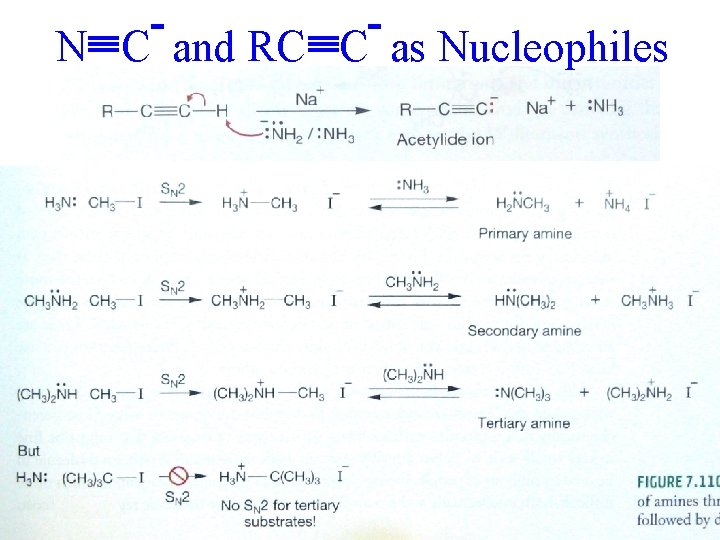
N C and RC C as Nucleophiles
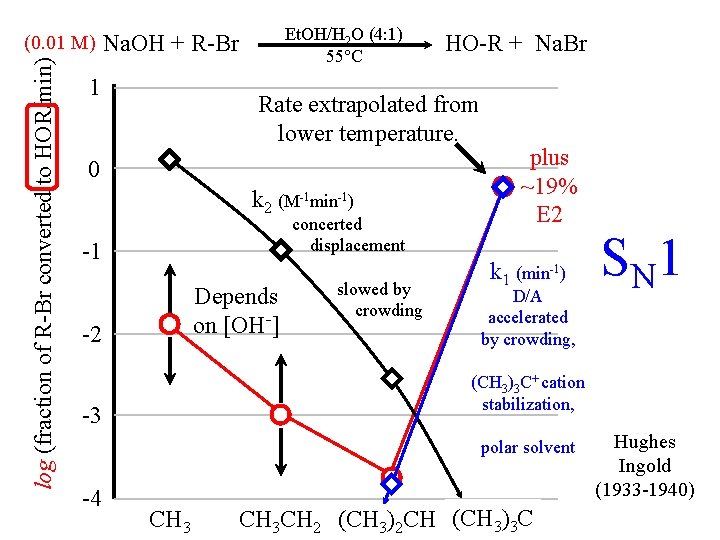
log (fraction of R-Br converted to HOR/min) (0. 01 M) Et. OH/H 2 O (4: 1) 55°C Na. OH + R-Br 1 HO-R + Na. Br Rate extrapolated from lower temperature. 0 k 2 (M-1 min-1) concerted displacement -1 Depends on [OH-] -2 slowed by crowding plus ~19% E 2 k 1 (min-1) D/A accelerated by crowding, (CH 3)3 C+ cation stabilization, -3 polar solvent -4 S N 1 CH 3 CH 2 (CH 3)2 CH (CH 3)3 C Hughes Ingold (1933 -1940)
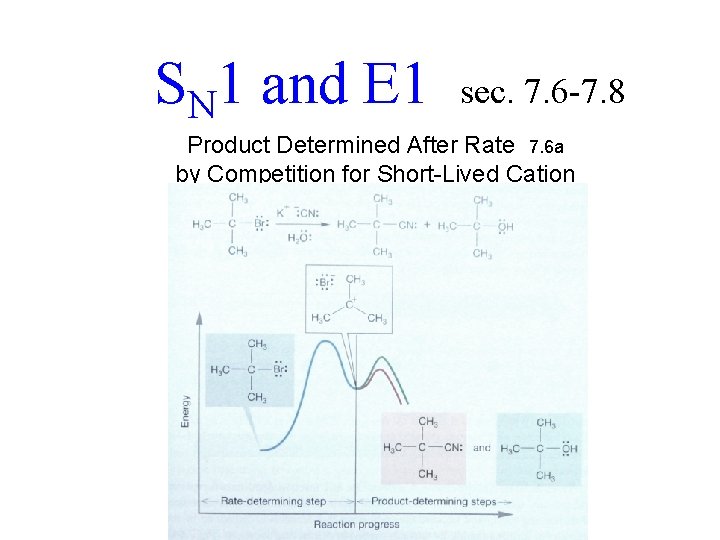
SN 1 and E 1 sec. 7. 6 -7. 8 Product Determined After Rate 7. 6 a by Competition for Short-Lived Cation
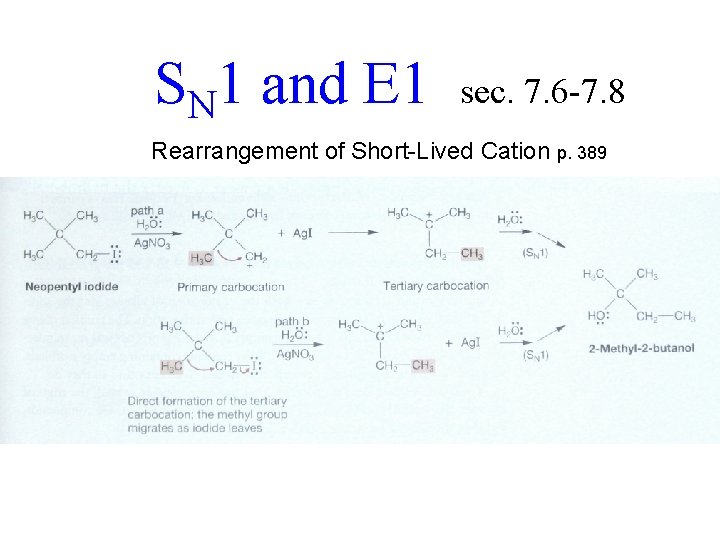
SN 1 and E 1 sec. 7. 6 -7. 8 Rearrangement of Short-Lived Cation p. 389

SN 1 and E 1 sec. 7. 6 -7. 8 Net Inversion from Short-Lived Ion Pair 7. 6 b

(0. 01 M) Na. OH + t. Bu-Br Et. OH/H 2 O (4: 1) 55°C HO-t-Bu + Na. Br + CH 2=C(CH 3)2 E 2 or E 1? How do you tell? Overall rate (and % alkene) depends on [OH-] Kinetic Isotope Effect shows whether H is being transferred in rate-determining step.

5. (5 min) Give a real example of the influence of a change of reactant structure on the ratio of SN 2 to E 2 products. Be as specific and quantitative as you can. (You will need to show the ratios for two different reactants. ) CH 3 -Br + OH (CH 3)3 C-Br + OH- Perspectives on Drastic Ratios Synthetic Organic Chemist : Reliable High-Yield Tool Physical-Organic Chemist : Definitive Ea Difference Unambiguous interpretation of cause e. g. Steric retardation of SN 2 / 105 acceleration for t-Butyl via SN 1

Perspectives on 50: 50 Product Ratios Synthetic Organic Chemist : Deadly Influence on 12 -Step Synthesis (1/2)12 = 0. 02% Yield (Might provide optimizable lead) Physical-Organic Chemist : Valuable “Borderline” Reference Allows Sensitive Tests of Subtle Influences. e. g. isotope effect by competition

A lesson from E 2 Elimination
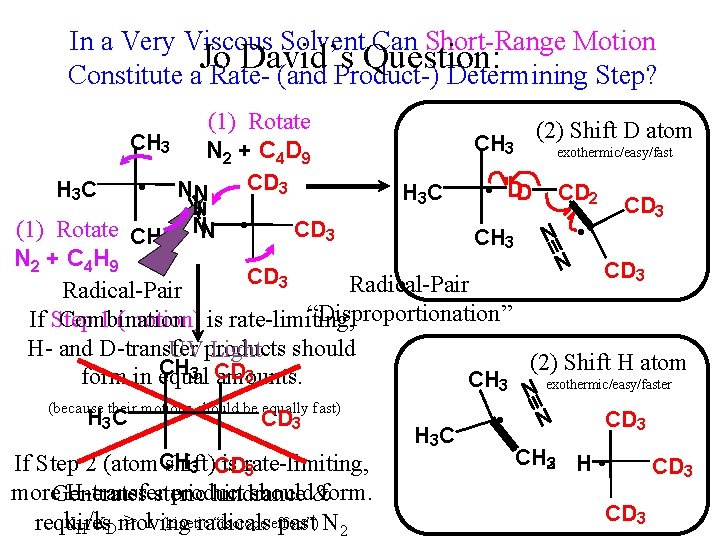
In a Very Viscous Solvent Can Short-Range Motion Jo David’s Question: Constitute a Rate- (and Product-) Determining Step? CH 3 H 3 C • (1) Rotate N 2 + C 4 D 9 NN (1) Rotate CH 3 NN CD 3 • (2) Shift D atom CH 3 H 3 C CD 3 exothermic/easy/fast • DD CH 3 CD 2 CD 3 N • N CD 3 N 2 + C 4 H 9 CD 3 Radical-Pair “Disproportionation” If Step 1 (motion) is rate-limiting, Combination H- and D-transfer UVproducts Light should (2) Shift H atom CH CD 3 3 form in equal amounts. CH 3 exothermic/easy/faster (because their motions should be equally fast) H 3 C CD 3 If Step 2 (atom. CH shift) is rate-limiting, 3 CD 3 more. Generates H-transfersteric product should&form. hindrance k. H/k. Dmoving > 1 (kinetic “isotope effect”) requires radicals past N 2 H 3 C N • N CD 3 CH 23 H • CD 3

t-Butylhydrazine (prepare from) CD 3 To do his project, Jo David needed to prepare this compound. E 2 >> SN 2 ?

Smith-Lakritz

It is very common to change a C=X double bond into C=O and H 2 X (we’ll be discussing this) C=N-R C=O + H N-R

t-Butylhydrazine CD 3 ? ? ? Jo David Fine April-October 1971 - + O

Happy Ending: Jo David Fine Jo David is now a respected professor of dermatology at Vanderbilt University, whose son has graduated from Yale. Jo David Fine Notebook p. 91 (October 1971)

Crucial Lesson (from S. Nelsen, U. Wisc. ) HCl salt easily purified by crystallization CD 3 When you need a compound, % yield isn’t everything! 5% SN 1 95% E 1 / E 2 Major product a gas, just “goes away” Cheap!

Happy Ending: Jo David Fine’s successor found that in fluid solvents, there was more H- than Dtransfer (atom transfer is rate-limiting), but that in very viscous solvents at low temperature this “kinetic isotope effect” disappeared (there were equal amounts of H- and D-transfer), because motion had indeed become rate-limiting.

End of Lecture 46 Feb. 1, 2010 Copyright © J. M. Mc. Bride 2010. Some rights reserved. Except for cited third-party materials, and those used by visiting speakers, all content is licensed under a Creative Commons License (Attribution-Non. Commercial-Share. Alike 3. 0). Use of this content constitutes your acceptance of the noted license and the terms and conditions of use. Materials from Wikimedia Commons are denoted by the symbol . Third party materials may be subject to additional intellectual property notices, information, or restrictions. The following attribution may be used when reusing material that is not identified as third-party content: J. M. Mc. Bride, Chem 125. License: Creative Commons BY-NC-SA 3. 0
- Slides: 24