Chemistry 125 Lecture 20 October 21 2009 Berzelius

Chemistry 125: Lecture 20 October 21, 2009 Berzelius, Gay-Lussac, Davy, & Wöhler (1805 -1830) Isomerism The most prominent chemist in the generation following Lavoisier was Sweden’s J. J. Berzelius. Together with Gay-Lussac in Paris and Davy in London, he discovered new elements and improved atomic weights and combustion analysis for organic compounds. Invention of electrolysis led not only to new elements but also to theory of dualism, in which elements were united by electrostatic attraction. Wöhler’s report on the synthesis of urea revealed not only isomerism but also a persistent naiveté about treating quantitative data. It was now clear that there was more to chemical substances than which atoms they contained and in what ratios. For copyright notice see final page of this file

Jöns-Jakob Berzelius BERZELIUS (1779 -1848) Organic & Mineral Analysis 2000 compounds in 6 years! Good Atomic Weights Textbook (1808) for 50 elements! Electrolysis Dualism (double decomposition) Teaching & Writing Notation for Composition

Atomic Weights and Equivalents
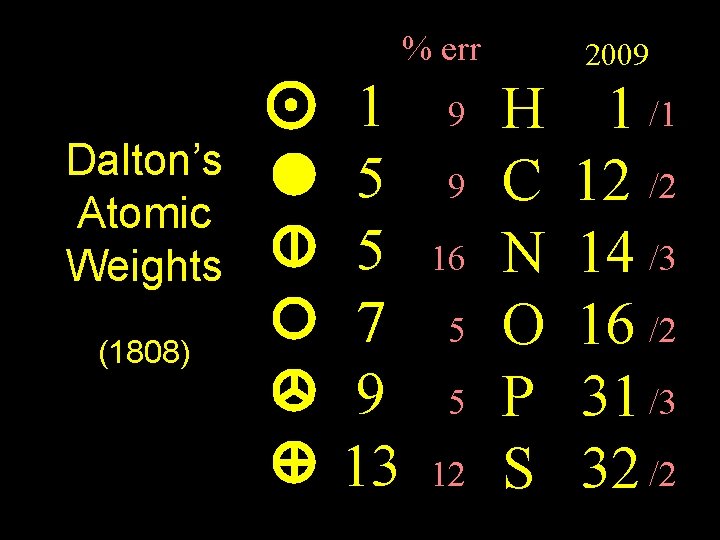
% err Dalton’s Atomic Weights (1808) Weights 1 5 5 7 9 13 9 9 16 5 5 12 2009 H C N O P S 1 /1 12 /2 14 /3 16 /2 31 /3 32 /2

Silicon Chloride ? Si. Cl T. Thomson Si. Cl 2 L. Gmelin Si. Cl 3 J. J. Berzelius Si. Cl 4 Wm. Odling
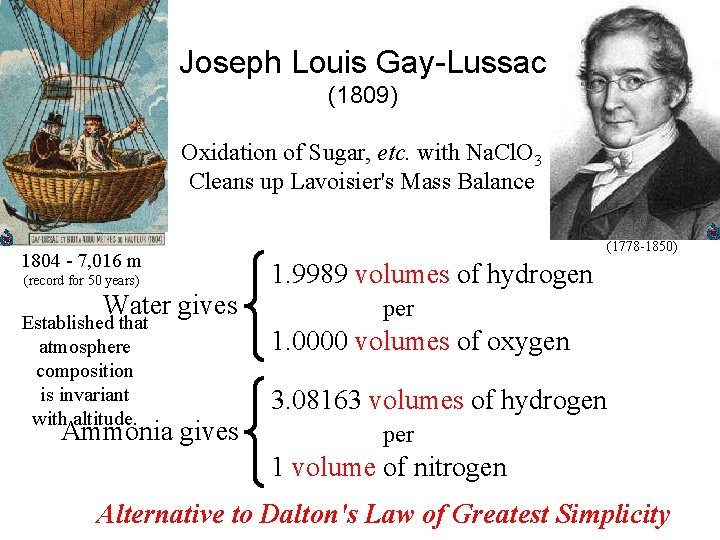
Joseph Louis Gay-Lussac (1809) Oxidation of Sugar, etc. with Na. Cl. O 3 Cleans up Lavoisier's Mass Balance 1804 - 7, 016 m (record for 50 years) Water gives Established that atmosphere composition is invariant with altitude. Ammonia gives (1778 -1850) 1. 9989 volumes of hydrogen per 1. 0000 volumes of oxygen 3. 08163 volumes of hydrogen per 1 volume of nitrogen Alternative to Dalton's Law of Greatest Simplicity

Eilhard Mitscherlich (1794 -1863) Goniometer measures angles between crystal faces.

Relating Crystal Shape to Chemical Proportions (formula)
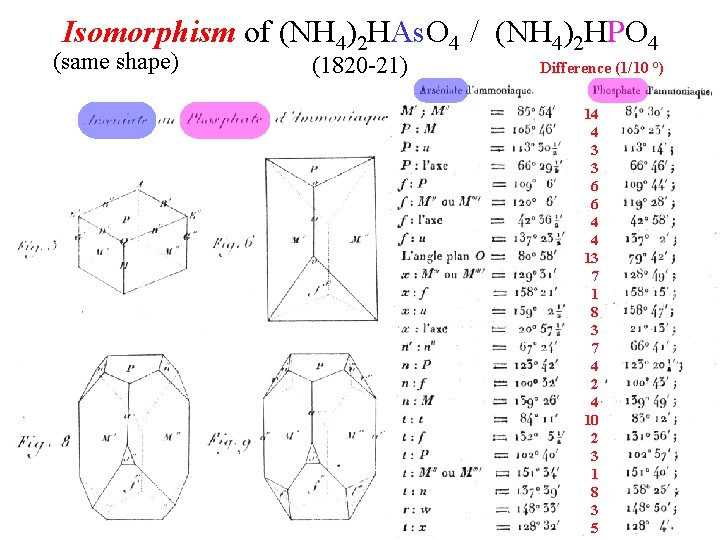
Isomorphism of (NH 4)2 HAs. O 4 / (NH 4)2 HPO 4 (same shape) (1820 -21) Difference (1/10 °) 14 4 3 3 6 6 4 4 13 7 1 8 3 7 4 2 4 10 2 3 1 8 3 5

Need for relative atomic weights: Is water HO (Dalton) or H 2 O (Gay-Lussac)? Isomorphism provided definitive atomic weights ratios for some atom pairs. As atoms must play exactly the same role in arsenates that P atoms play in phosphates. 100 g of N, O, H combines with 30. 64 g P, or 78. 11 g As atom 2. 55 times as heavy as P atom (neither 1. 275 nor 5. 10 times as heavy)

Berzelius’s Table of atomic & molecular weights (1831) Weights
![Scaled to modern O = 15. 9994 [15. 999] 0. 998 (-1. 0%) Bars Scaled to modern O = 15. 9994 [15. 999] 0. 998 (-1. 0%) Bars](http://slidetodoc.com/presentation_image/2761d17ea3ed8ce902cab781130f9fd4/image-12.jpg)
Scaled to modern O = 15. 9994 [15. 999] 0. 998 (-1. 0%) Bars denote doubled atoms 14. 162 (-1. 0) 32. 185 (0. 4) 30. 974 (1. 3) discrepancy from modern value 10 -fold increase in accuracy from Dalton! Based on O = 100 or H 2 = 1

Combustion Analysis

Improved Method of Combustion Analysis Berzelius Analysis (1) (4)Tube Assembled 1/2” diameter (Fig 6) so(Fig that 1) charged gases (Owith ) that powder exit drying containing: tube 2, CO 2 dried ~0. 5 will. Joined gbubble of organic into substance bell-jar containing to be analyzed, Hg (Fig 3) (Fig 4) to water-collecting bulb (2) Neck of tube heated and drawn out (Fig 2) with floating 3 g. Ca. Cl Na. Cl. O bulb 2 (Fig (Gay-Lussac’s holding KOH Orubber (to absorb CO 2) and drying tube with tubing 2 source), 3 5) and closed 50 with g Na. Cl permeable (to moderate glove reaction leather by dilution). (to keep out Hg).
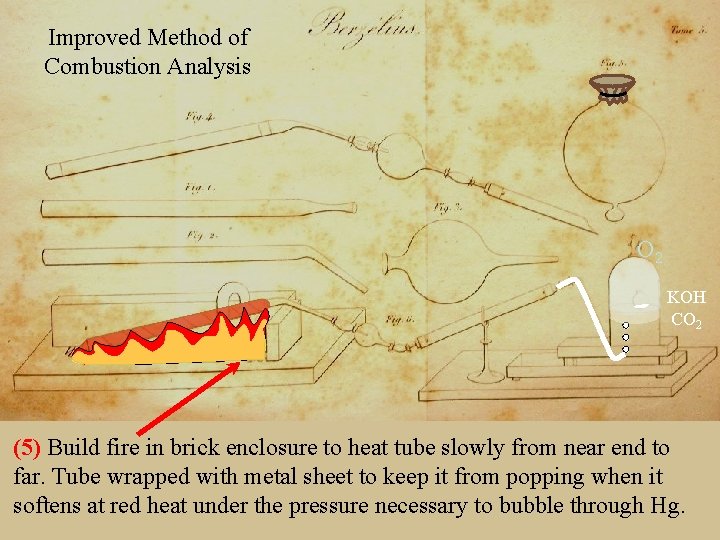
Improved Method of Combustion Analysis Berzelius Analysis O 2 KOH CO 2 (5)(6) Build firecertain in brick to heatalltube slowly nearthe end to To be theenclosure KOH absorbs of the CO 2 from through far. glove Tubeleather, wrapped with keep it from popping wait 12 metal hourssheet aftertothe mercury stops risingwhen in theit softens at before red heatdisassembling under the pressure necessary to bubble through Hg. bell jar and weighing.

Electricity

N 2 O at the Royal Institution London, 1802 Humphry Davy New York Public Library http: //seeing. nypl. org/152 t. html
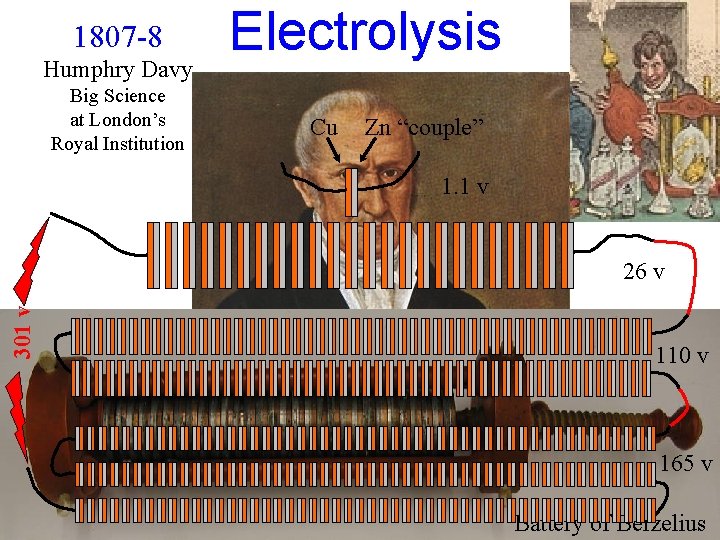
1807 -8 Humphry Davy Big Science at London’s Royal Institution Electrolysis Cu Zn “couple” 1. 1 v 301 v 26 v Pile 110 v 1799 Volta 165 v Battery of Berzelius

Davy (1808) I acted upon aqueous solutions of potash and soda, saturated at common temperatures, by the highest electrical power I could command, and which was produced by a combination of Voltaic batteries belonging to the Royal Institution, containing 24 plates of copper and zinc of 12 inches square, 100 plates of 6 inches, and 150 of 4 inches square… …though there was a high intensity of action, the water of the solutions alone was affected, and hydrogen and oxygen disengaged with the production of much heat and violent effervescence.

Davy (1808) The presence of water appearing thus to prevent any decomposition, I used potash in igneous fusion. By means of a stream of oxygen gas from a gasometer applied to the flame of a spirit lamp, which was thrown on a platina spoon containing potash, this alkali was kept for some minutes in a strong red heat, and in a state of perfect fluidity. The spoon was preserved in communication with the positive side of the battery of the power of 100 of 6 inches, highly charged; and the connection from the negative side was made by a platina wire. By this arrangement some brilliant phenomena were produced. The potash appeared a conductor in a high degree, and as long as the communication was preserved, a most intense light was exhibited at the negative wire, and a column of flame, which seemed to be owing to the development of combustible matter, arose from the point of contact.

Davy (1808) A small piece of pure potash, which had been exposed for a few seconds to the atmosphere, so as to give conducting power to the surface, was placed upon an insulated disc of platina, connected with the negative side of the battery of the power of 250 of 6 and 4, in a state of intense activity; and a platina wire, communicating with the positive side, was brought in contact with the upper surface of the alkali. …small globules having a high metallic lustre, and being precisely similar in visible characters to quick-silver, appeared, some of which burnt with explosion and bright flame, as soon as they were formed, and others remained, and were merely tarnished, and finally covered by a white film which formed on their surfaces.

1807 -8 Humphry Davy Big Science at London’s Royal Institution Li Be B Na Mg Al K Ca Ga Rb Sr In ~650 v Cs Ba Te Napoleon’s Response (via Gay-Lussac) “with that rapidity which characterised all his motions, and before the attendants could interpose any precaution, he thrust the extreme wires of the battery under his tongue, and received a shock which nearly deprived him of sensation. After recovering from its effects, he quitted the laboratory without making any 600 1 kg Cu plates remark, and was never afterwards heard to refer to 2, 200 3 kg Zn plates v the subject. ” from: “The Life of 600 Sir Humphry Davy, Bart. ” 2. 6 tons of metal

Electricity supplied more than new elements. . . It also supplied the organizing principle for Dualism

1774 Symbols Chemical Symbols of 1774 (Sweden) Mix with Caustic Soda
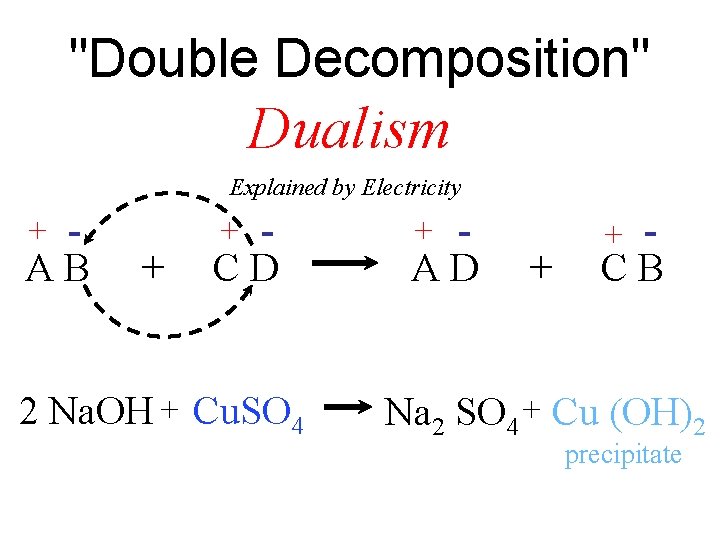
"Double Decomposition" Dualism Explained by Electricity + - AB + + - CD 2 Na. OH + Cu. SO 4 + - AD + + - CB Na 2 SO 4 + Cu (OH)2 precipitate

“Electronegativity” First use in English (according to O. E. D. ) 1837 J. D. Dana Syst. Mineral. 82 When chemistry has so far advanced, that the relative electro-negativity, (if I may so call it, ) or electro-positivity, of the several elements, is fully known, . . we shall probably be able to construct a natural arrangement of minerals on chemical principles. J. D. Dana 1813 -1895 son-in-law of Benjamin Silliman Dana House 1849
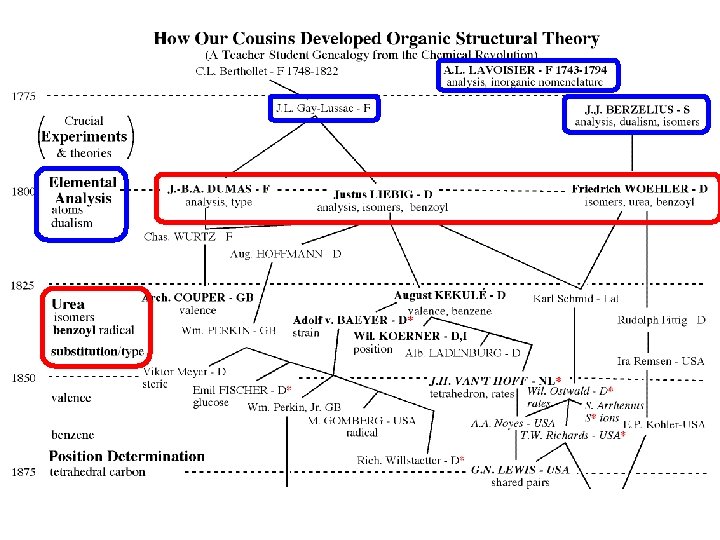
Genealogy Top

Wöhler/Liebig

to J. J. Berzelius Heidelberg, 17 July, 1823 “Having developed the greatest respect for you through studying your writings, I have always thought it would be my greatest good fortune to be able to practice this science under the direction of such a man, which has always been my fondest desire. Although I earlier had planned to become a physician…”

With the greatest respect F. Wöhler from Frankfurt am Main
![Letter to Berzelius (1837) “To see this old friend [Palmstedt] again, especially here [in Letter to Berzelius (1837) “To see this old friend [Palmstedt] again, especially here [in](http://slidetodoc.com/presentation_image/2761d17ea3ed8ce902cab781130f9fd4/image-31.jpg)
Letter to Berzelius (1837) “To see this old friend [Palmstedt] again, especially here [in Göttingen], was a real delight. He was just the same old guy, with the sole exception that he no longer wears the little toupee swept up over his forehead as he used to do. ” Friedrich Wöhler (1800 -1882)

1828 Urea & Isomerism (problems for Monday, Oct. 26 - see web readings)

Wöhler to Berzelius (1828) "Perhaps you still remember the experiment I carried out in that fortunate time when I was working with you, in which I found that whenever one tries to react cyanic acid with ammonia, a crystalline substance appears which is inert, behaving neither like cyanate nor like ammonia. "

"Ammonium Cyanate" from Double Decomposition NH 3 Not like an ammonium salt! Na. OH + - NH 4 Cl + Ag OCN HNO 3 - + (Just like those from Urea + HNO 3) - NH 4 OCN + Ag Cl H+ OCNH Brilliant Crystal Flakes + Pb++ Pb(OCN)2 Not like a cyanate salt!

Berzelius to Wöhler: "It is a unique situation that the salt nature so entirely disappears when the acid and ammonia combine, one that will certainly be most enlightening for future theory…" Might Ammonium Cyanate Be Urea? Wöhler to Berzelius: "I recently performed a small experiment, appropriate to the limited time I have available, which I quickly completed and which, thank God, did not require a single analysis. "

Urea had already been Analyzed Paragon of accuracy and honesty Dr. Prout Atoms Modern (1817) NH 4 OCN (calculated by Wöhler from Berzelius atomic weights) Recalc. (from Berzelius) 1 46. 650 46. 65 46. 781 1 19. 975 20. 00 20. 198 2 6. 670 6. 71 6. 59 06. 595 Discrepancies <2% 1 26. 650 26. 64 26. 24 26. 425 identical Total 99. 875 100. 00 99. 80? 100. 000 Prout 99. 945 did dry-lab by making an approximate analysis truncated Dislectic Error Experimental Candor? (added wrong) Prout’s Experiment better than Berzelius’s and reporting results “corrected” by theory! of Theory (instead & his he didn't notice!) Prout’s N 2 theory by volume: was better "6. 3 cubic than Berzelius’s inches" Just experiments! Lucky? rounding up Moral: Don’t dry-lab! (like Lavoisier) Prout’s Atomic Weight Theory: H = 1 ("protyle" ulh rwth) C = 6 ; O = 8 ; N = 14 N C H O
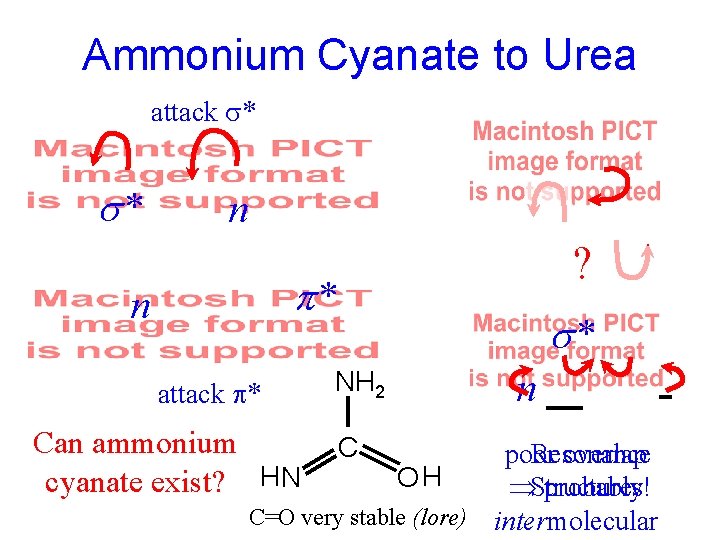
Ammonium Cyanate to Urea attack * s* n ? * n attack * Can ammonium cyanate exist? HH 2 N s* NH 2 C n poor overlap Resonance OH Structures! probably C=O very stable (lore) intermolecular
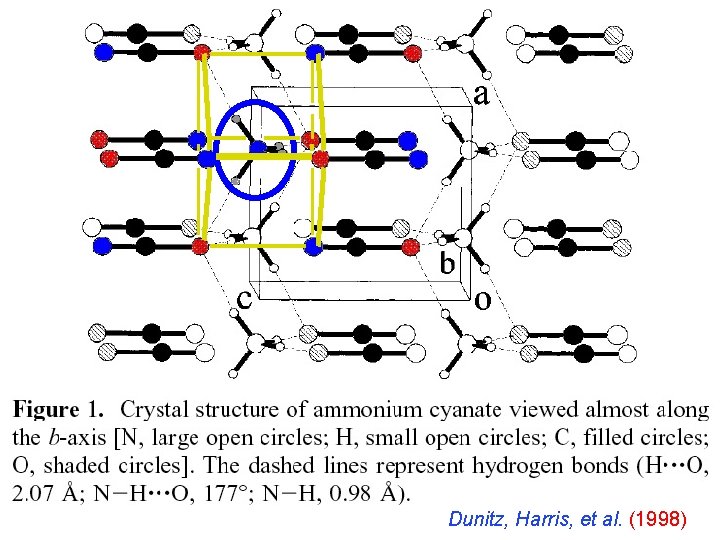
Dunitz, Harris, et al. (1998)

Wöhler on Isomerism (1828) "I refrain from all the considerations which so naturally suggest themselves from this fact, especially in respect to the composition ratios of organic substances and in respect to similar elemental and quantitative compositions among compounds with very different properties, as may be supposed, among others, of fulminic acid and cyanic acid and of a liquid hydrocarbon and the olefiant gas, and it must be left to further investigations of many similar cases to decide what general laws can be derived therefrom. "

On the Composition of Tartaric Acid and Racemic Acid (John's Acid from the Vosges Mountains), on the Atomic Weight of Lead Oxide, together with General Remarks on those Substances with have the Same Composition but Different Properties. by J. J. Berzelius (1830) "I have thought it necessary to choose between the words : homosynthetic and isomeric substances. The former is built from homos, equivalent, and synthetos, put together; the latter from isomeres has the same meaning, although it only properly says put together from the same pieces. The latter has the advantage with respect to brevity and euphony, and thus. X-Ray I haveshowed decidedthat to these crystals are choose it. " Calcium Tartrate • 4 H O "By isomeric substances I understand those which possess 2 the same chemical composition and the same atomic [molecular] weight, but different properties. "

There is more to chemistry than analytical COMPOSITION! Now we know the importance of atomic arrangement, or STRUCTURE: CONSTITUTION CONFIGURATION CONFORMATION but we need to be patient.

End of Lecture 20 October 21, 2009 Copyright © J. M. Mc. Bride 2009. Some rights reserved. Except for cited third-party materials, and those used by visiting speakers, all content is licensed under a Creative Commons License (Attribution-Non. Commercial-Share. Alike 3. 0). Use of this content constitutes your acceptance of the noted license and the terms and conditions of use. Materials from Wikimedia Commons are denoted by the symbol . Third party materials may be subject to additional intellectual property notices, information, or restrictions. The following attribution may be used when reusing material that is not identified as third-party content: J. M. Mc. Bride, Chem 125. License: Creative Commons BY-NC-SA 3. 0
- Slides: 42