Chemistry 125 Lecture 11 Orbital Correction and PlumPudding

Chemistry 125: Lecture 11 Orbital Correction and Plum-Pudding Molecules Several tricks (“Z-effective” and “Self Consistent Field”) allow one to correct approximately for the error in using orbitals when there is electron-electron repulsion. Residual error is hidden by naming it “Correlation energy. ” J. J. Thomson’s Plum-Pudding model of the atom can be modified to visualize the form of molecular orbitals. There is a close analogy in form between the molecular orbitals of CH 4 and NH 3 and the atomic orbitals of neon, which has the same number of protons and electrons. The underlying form, dictated by kinetic energy, is distorted by pulling protons out of the Ne nucleus to play the role of H atoms. Synchronize when the speaker finishes saying “we’ve been looking at atoms. ” Synchrony can be adjusted by using the pause(||) and run(>) controls. For copyright notice see final page of this file

What's Coming for Next Exam? Atoms Orbitals for Many-Electron Atoms (Wrong!) Recovering from the Orbital Approximation Molecules Plum-Pudding Molecules (the "United Atom" Limit) Understanding Bonds (Pairwise LCAO) "Energy-Match & Overlap" Reality: Structure (and Dynamics) of XH 3 Molecules Reactivity HOMOs and LUMOs Recognizing Functional Groups Payoff for Organic Chemistry! How Organic Chemistry Really Developed (Intro)

2 -e Wave Function ? Y(r , q , f , r , q , f ) = 2 1 1 1 2 2 2 a(r 1, q 1, f 1) b(r 2, q 2, f 2) Multiply 1 -e Wave Functions No way can electrons be independent! They repel one another.

Tricks for Salvaging Orbitals
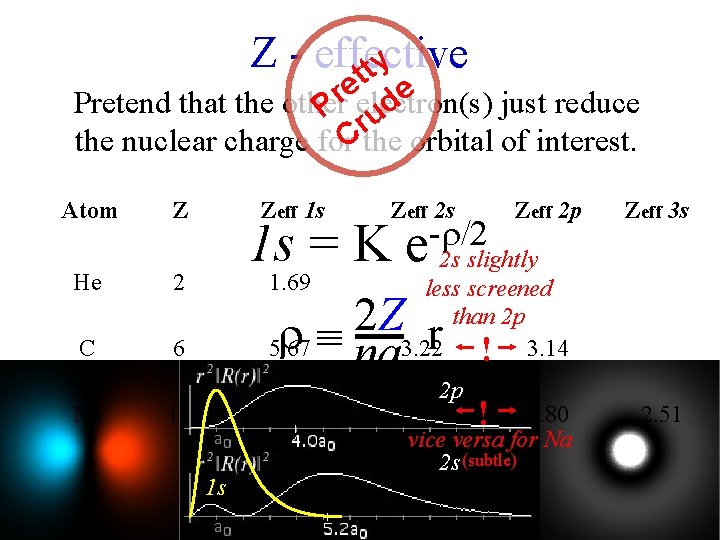
Z - effective tty e e r Pretend that the other electron(s) just reduce d P ru the nuclear charge for. Cthe orbital of interest. Atom Z He 2 C 6 Na Zeff 1 s Zeff 2 s 1 s = K 1. 69 - /2 e Zeff 2 p 2 s slightly less screened than 2 p 3. 22 ! 3. 14 2 Z na r 5. 67 o 11 1 s Zeff 3 s 2 p 10. 63 6. 57 ! 6. 80 2. 51 vice versa for Na 2 s (subtle) "Clementi-Raimondi" values for Zeff (best fit to better calculations)

Self-Consistent Field (SCF) 1. Find approximate orbitals for all electrons (e. g. using Zeff) 2. Calculate potential from nuclei and. . . fixed clouds for all electrons but one. 3. Use this new potential to calculate an. . improved orbital for that one electron. 4. Repeat steps 2 and 3 to improve the orbital for another electron. . Improve all orbitals one by one. Cycle back to improve 1 st orbital further, etc. Quit When orbital shapes stop changing

Still Wrong! because real electrons are not fixed clouds. They keep out of each other’s way by correlating their motions. True Energy < SCF Energy

Hide the residual error after full SCF calculation to the “Hartree-Fock” limit by giving it a fancy name: "Correlation Energy" Where to get correct energy (& total electron density)? by experiment or by a whopping calculation: e. g. “Configuration Interaction” (CI) or “Density Functional Theory” (DFT)

If we’re really lucky, "Correlation Energy" might be Negligible.

Nucleus (2 109) Energy Fortunately nuclear energy 2) Loses 0. 1 amu (E = mc 0. 001% change in nuclear energy is totally unchanged log (Energy Change kcal / mole 12 C 8 would during overwhelm all of Coulomb. chemistry! + • + + +6 - • +CC- • + - 6 Magnitudes Should Chemists care about the error in Orbital Theory? C Core (2 104) C Atom (3 103) correlation error ≈ bond Changes in 1/2 4 Single Bonds (2 102) "correlation energy" C "Correlation Energy" (102) can be ~10 -15% "Non-bonded" Contacts (1 -20) of Bond Energy. 4 2 0 -2 ~ He • He @ 52Å! (2 10 -6) Orbital Theory is fine for Qualitative Understanding of Bonding.

Orbitals can't be “true” for >1 electron, because of e-e repulsion but we'll use them to understand bonding, structure, energy, and reactivity
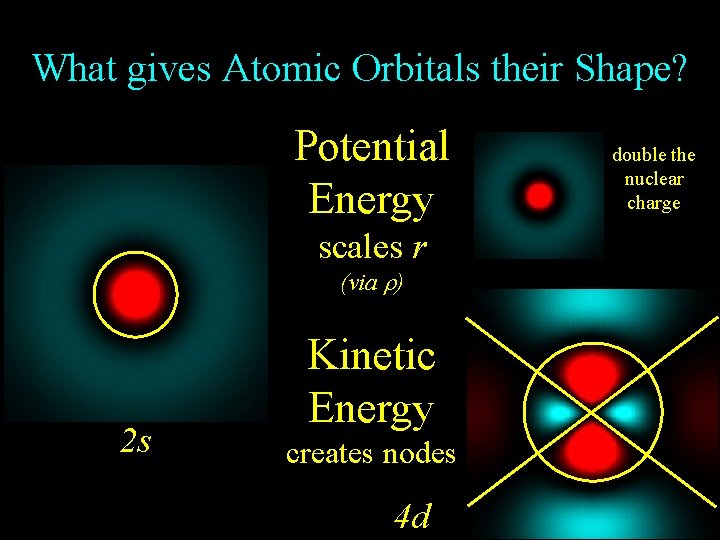
What gives Atomic Orbitals their Shape? Potential Energy scales r (via ) 2 s Kinetic Energy creates nodes 4 d double the nuclear charge

If we use orbitals, how should we reckon total electron density? Density of electron 1 = 1 2(x 1, y 1, z 1) Density of electron 2 = 2 2(x 2, y 2, z 2) Total density (x, y, z) = 1 2(x, y, z) + 2 2(x, y, z) (Sum, not Product. Not a question of joint probability)

How Lumpy is the N Atom? (2 px)2 = K x 2 e- (2 py)2 = K y 2 e- (2 pz)2 = K z 2 e- Total = K(x 2 + y 2 + z 2) e- Total = K(r 2) e- Spherical ! [from an Organic Text]
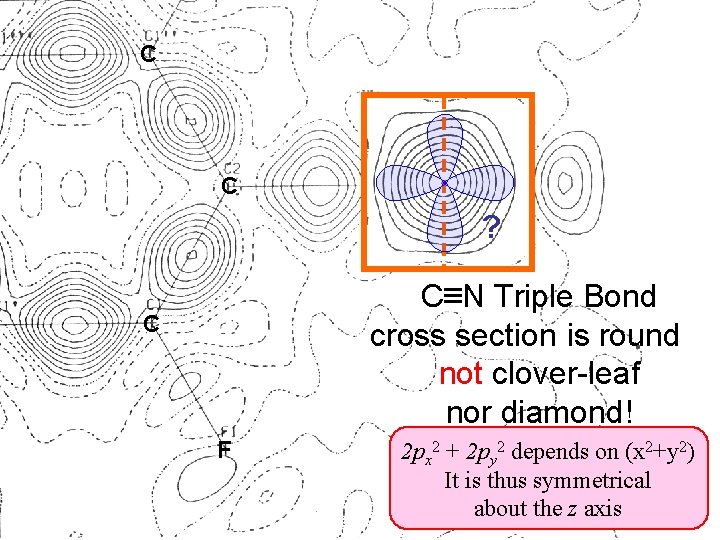
C TFDCB C C N ? C N Triple Bond cross section is round not clover-leaf nor diamond! C F 2 px 2 + 2 py 2 depends on (x 2+y 2) It is thus symmetrical about the z axis

Atoms 3 -Dimensional Reality (H-like Atoms) Hybridization Orbitals for Many-Electron Atoms (Wrong!) Recovering from the Orbital Approximation Molecules Plum-Pudding MOs (the "United Atom" Limit) Understanding Bonds (Pairwise LCAO-MOs) “Overlap & Energy-Match"

Ways of Looking at an Elephant
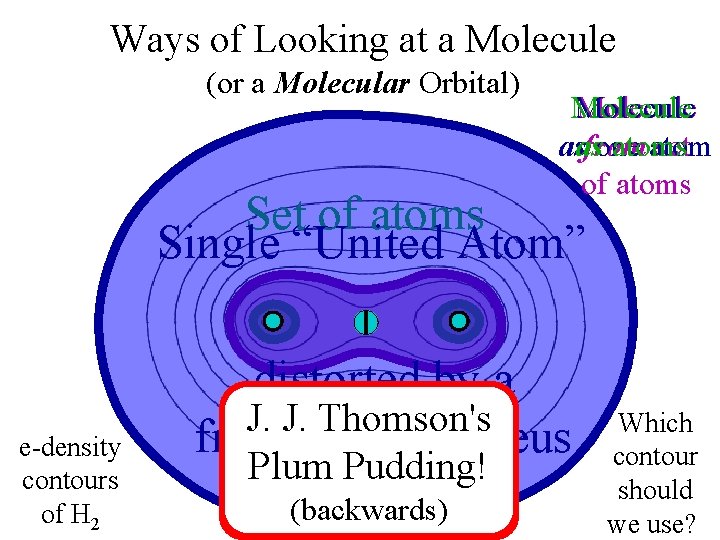
Ways of Looking at a Molecule (or a Molecular Orbital) Molecule asas from one atoms atom set of atoms Single “United Atom” e-density contours of H 2 distorted by a Atoms with J. Nuclei J. Thomson's embedded in fragmented nucleus a cloud of electrons small bonding Plum Pudding! dispersed and “noded” (backwards) by kinetic energy distortion Which contour should we use?

How the Plums Distort Electronic Puddings

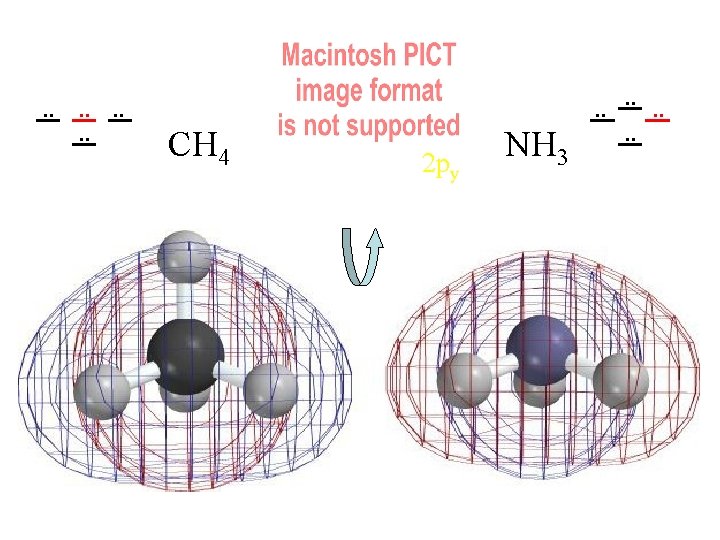
Methane & Ammonia Spartan 6 -31 G* calculates good SCF MOs (on my laptop!) We want to understand them visually.

Compare MOs to AOs of Ne (4 electron pairs with n=2) H H C H H N H H H 4 Pairs of Valence Electrons
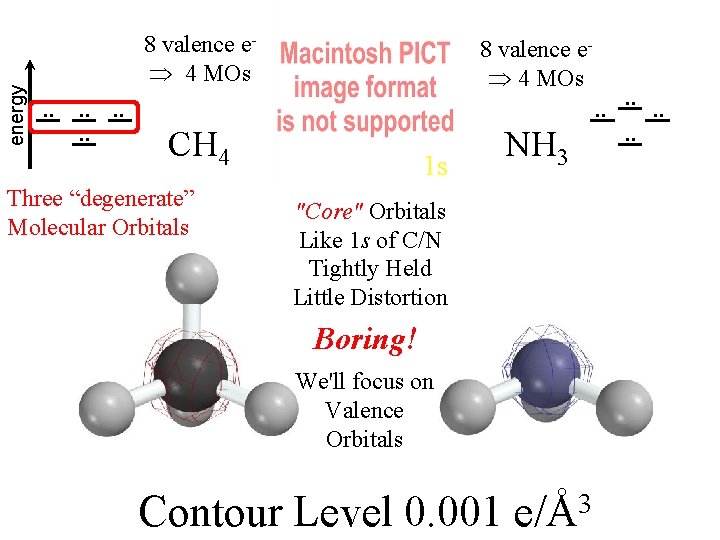
energy . . . . 8 valence e 4 MOs CH 4 NH 3 Three “degenerate” Molecular Orbitals 1 s "Core" Orbitals Like 1 s of C/N Tightly Held Little Distortion Boring! We'll focus on Valence Orbitals Contour Level 0. 001 e/Å3 . . . .
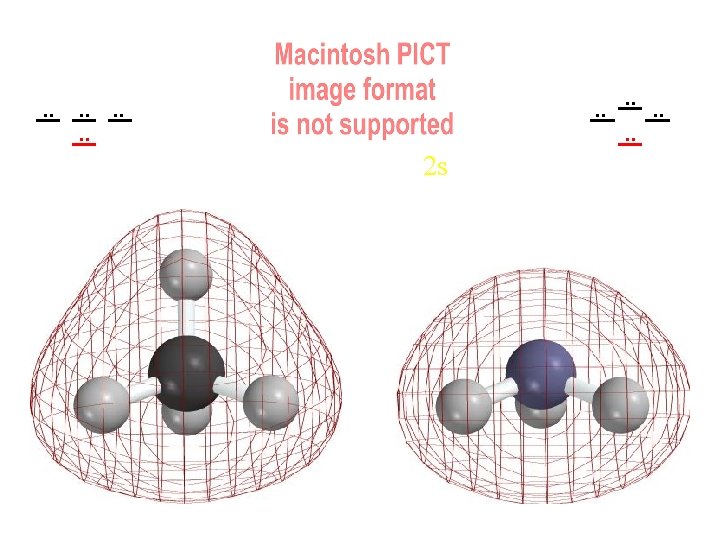
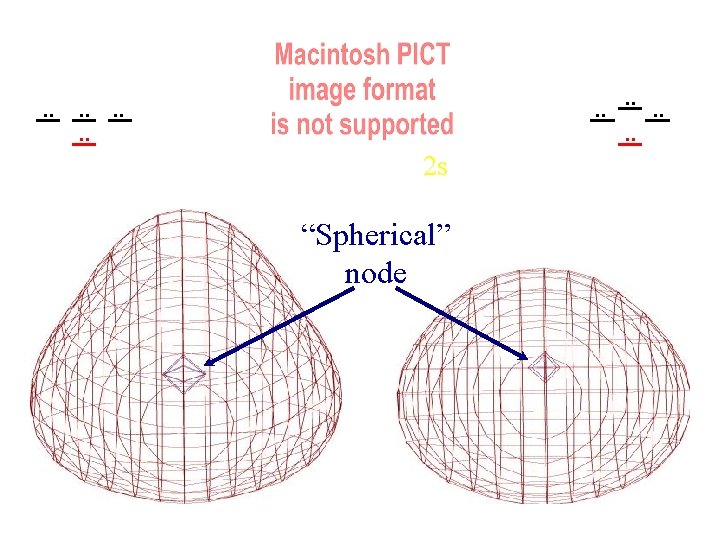
. . . . 2 s “Spherical” node
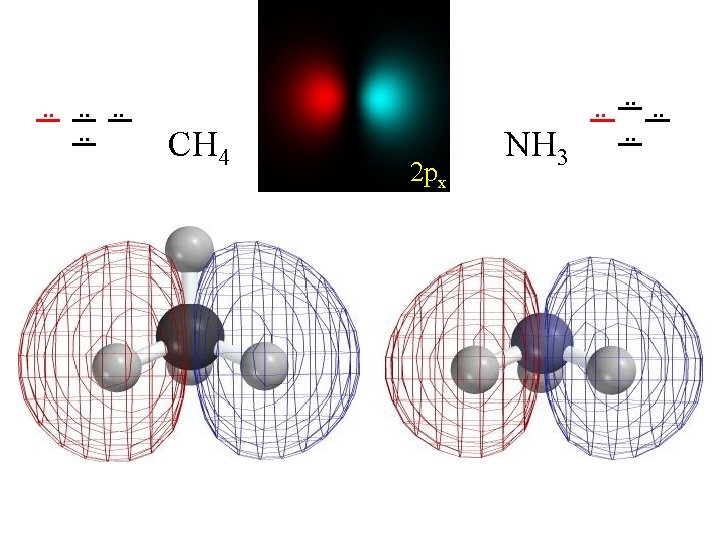

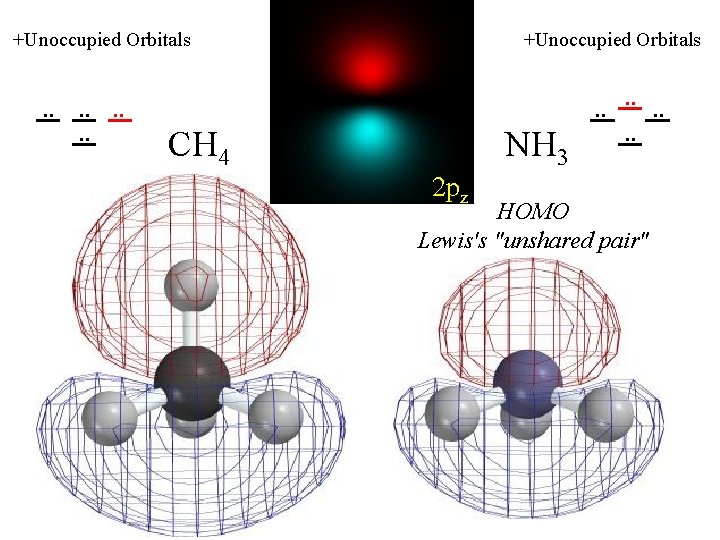
+Unoccupied Orbitals . . . . +Unoccupied Orbitals CH 4 NH 3 2 pz . . . . HOMO Lewis's "unshared pair"
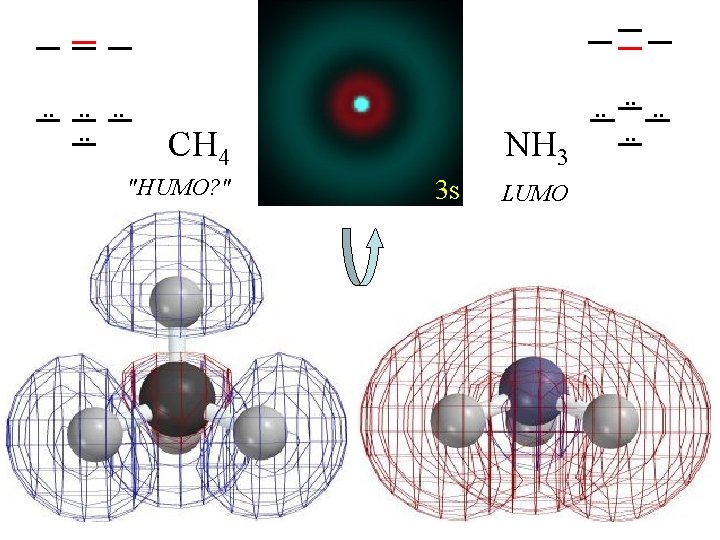
. . . . CH 4 "HUMO? " NH 3 3 s LUMO . . . .
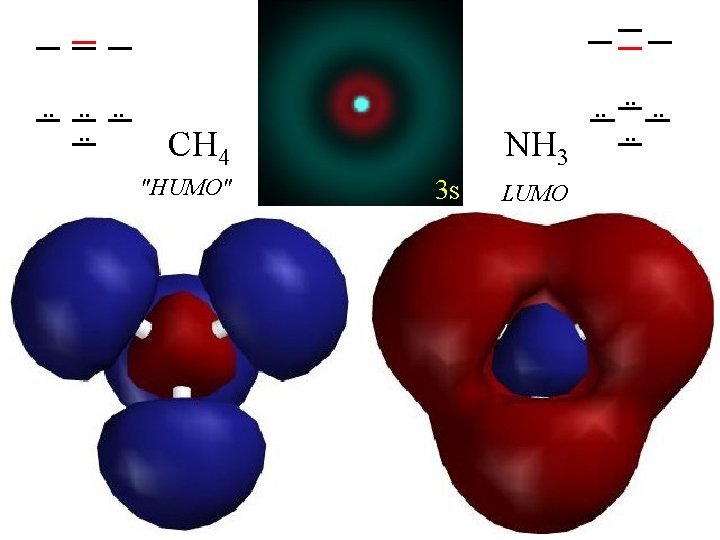
. . . . 2 s CH 4 "HUMO" NH 3 3 s LUMO . . . .
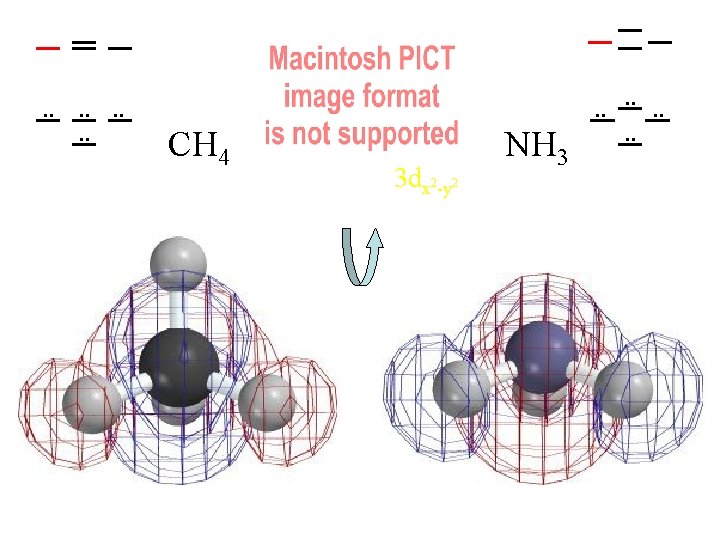
. . . . CH 4 3 dx 2 -y 2 NH 3 . . . .
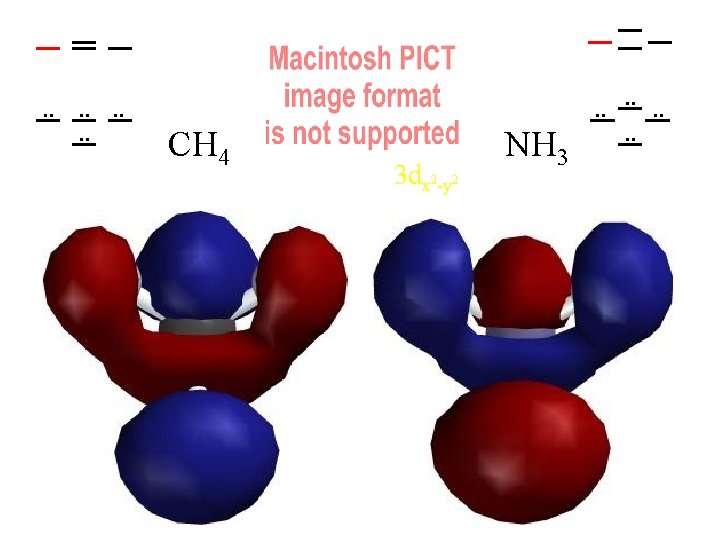
. . . . CH 4 3 dx 2 -y 2 NH 3 . . . .
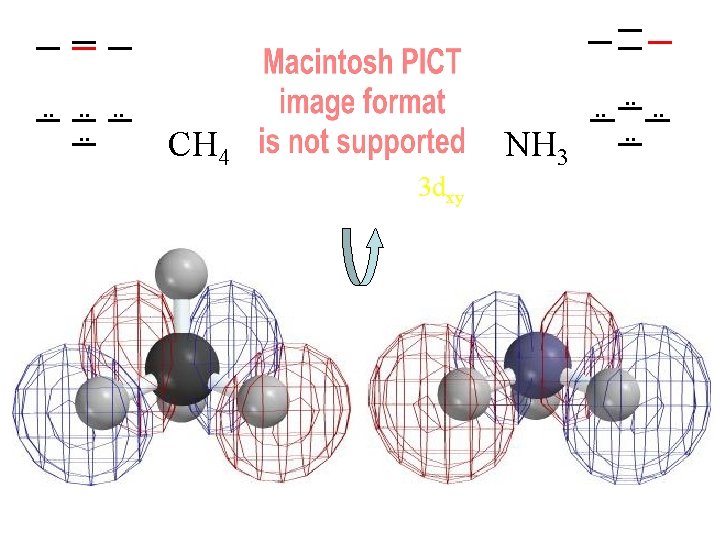
. . . . CH 4 NH 3 3 dxy . . . .
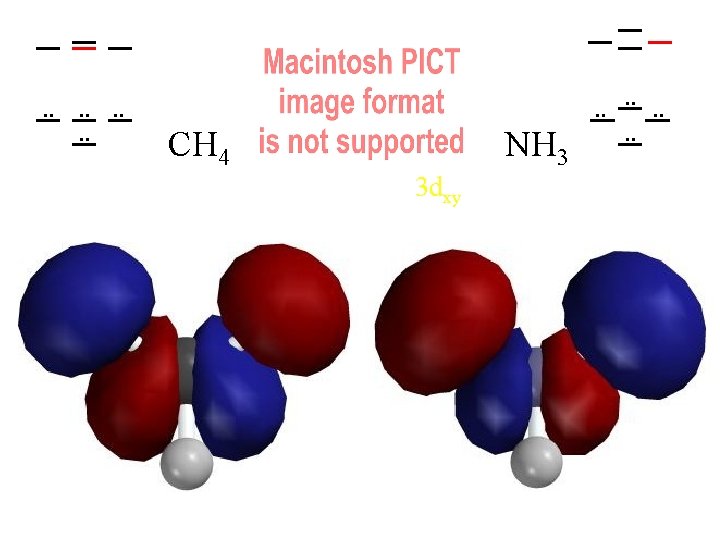
. . . . CH 4 NH 3 3 dxy . . . .
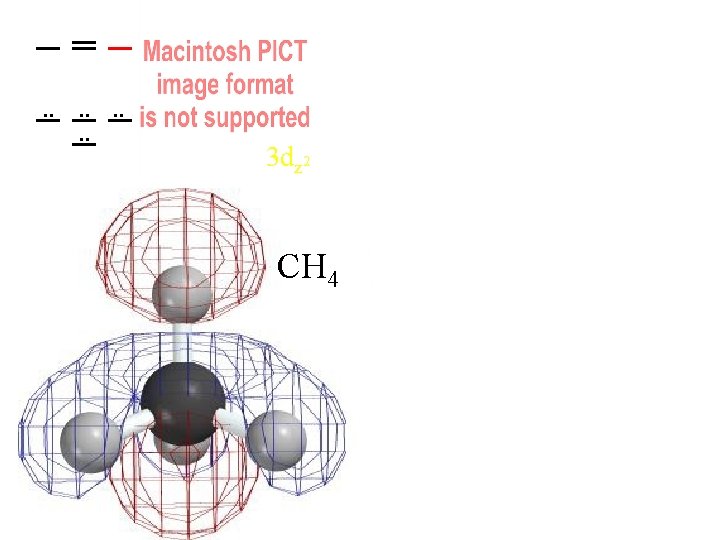
. . . . 3 dz 2 CH 4 3 dz 2

End of Lecture 11 Sept. 29, 2008 Copyright © J. M. Mc. Bride 2009. Some rights reserved. Except for cited third-party materials, and those used by visiting speakers, all content is licensed under a Creative Commons License (Attribution-Non. Commercial-Share. Alike 3. 0). Use of this content constitutes your acceptance of the noted license and the terms and conditions of use. Materials from Wikimedia Commons are denoted by the symbol . Third party materials may be subject to additional intellectual property notices, information, or restrictions. The following attribution may be used when reusing material that is not identified as third-party content: J. M. Mc. Bride, Chem 125. License: Creative Commons BY-NC-SA 3. 0
- Slides: 36