Chemistry 11 Chemical Bonding Electronegativity the tendency of
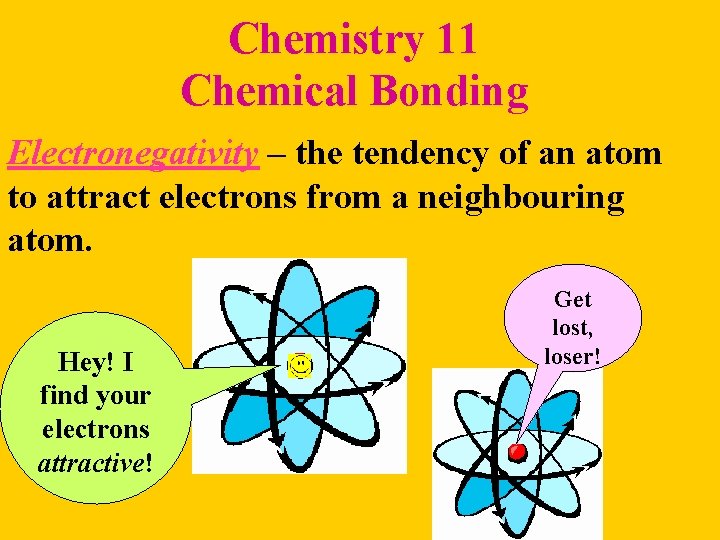
Chemistry 11 Chemical Bonding Electronegativity – the tendency of an atom to attract electrons from a neighbouring atom. Hey! I find your electrons attractive! Get lost, loser!
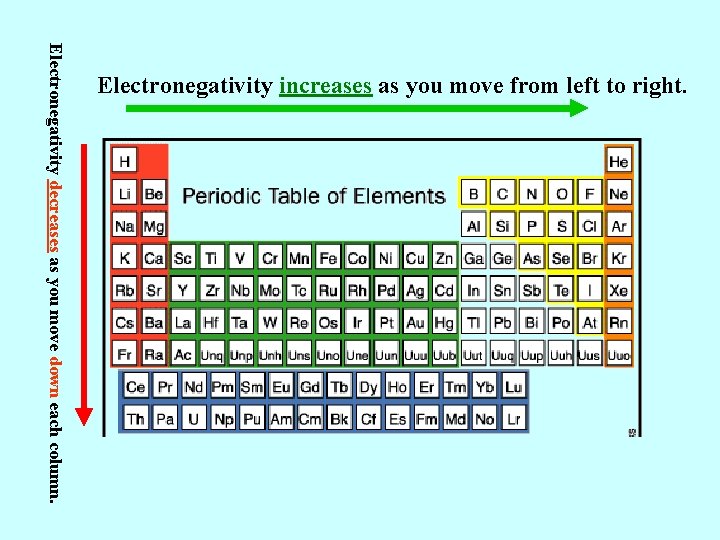
Electronegativity decreases as you move down each column. Electronegativity increases as you move from left to right.

Valence Electrons – electrons in the outermost occupied energy level. (s and p electrons outside the core) Valence electrons can be represented by “dots” drawn around the atom.

When the electronegativities of two atoms are quite different from each other: One atom loses an electron (or electrons) The other atom gains an electron (or electrons) This results in an Ionic Bond.
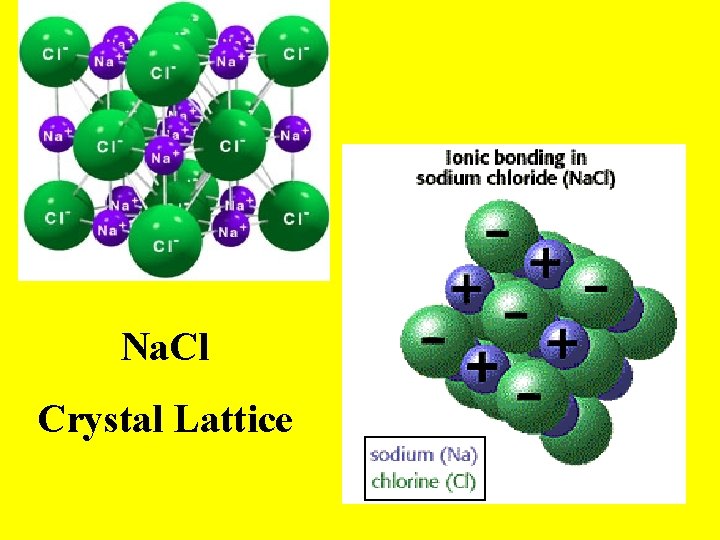
Na. Cl Crystal Lattice
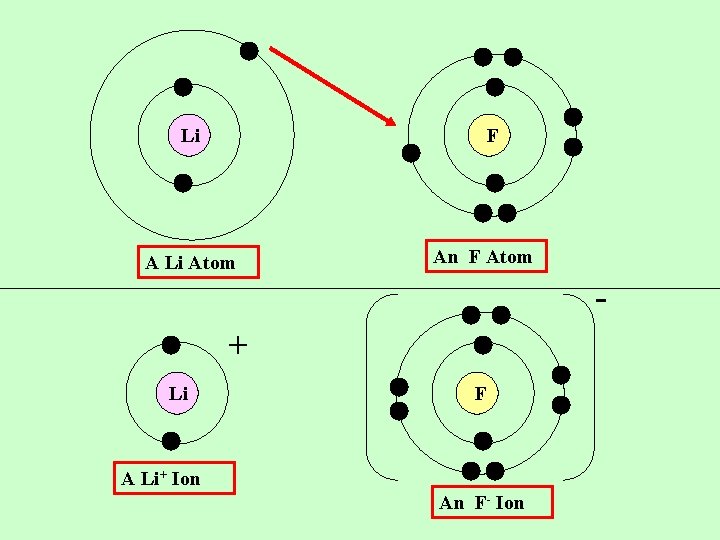
Li F A Li Atom An F Atom + Li F A Li+ Ion An F- Ion
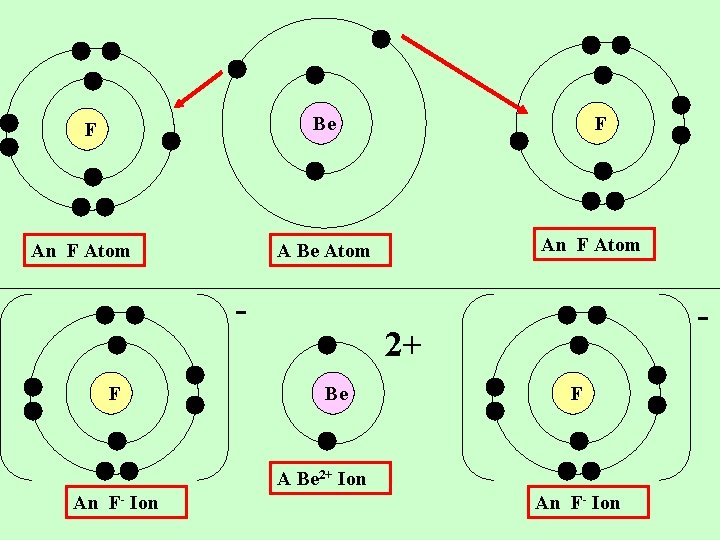
Be F An F Atom A Be Atom F F - 2+ Be F A Be 2+ Ion An F- Ion

The melting points of some Ionic Compounds are as follows: Na. F KCl Li. Cl 993 o. C 770 o. C 605 o. C These high melting points are experimental evidence that Ionic Bonds are VERY STRONG. (Hard to break just by heating).

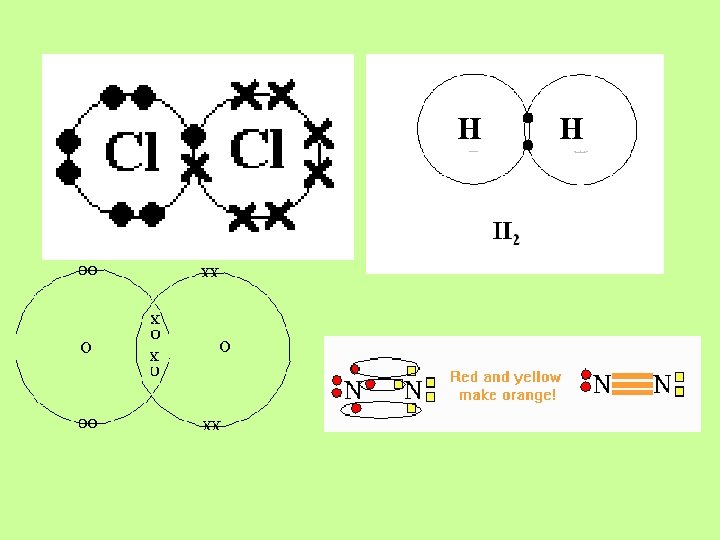
When Electronegativities of bonding atoms are the same (as they are in diatomic molecules) or close to the same, they SHARE electrons. Bonds formed when atoms share electrons are called Covalent Bonds. In diatomic molecules (like H 2 or Cl 2), the electronegativities of both atoms are exactly the same so electrons are shared equally!
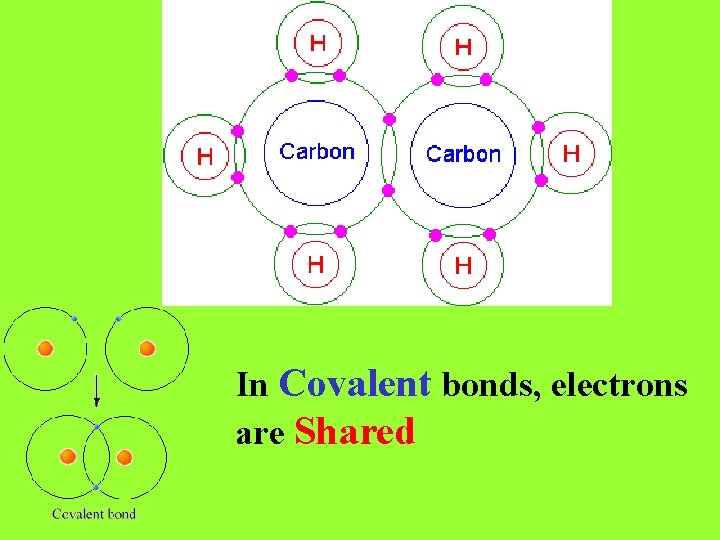
In Covalent bonds, electrons are Shared
H H
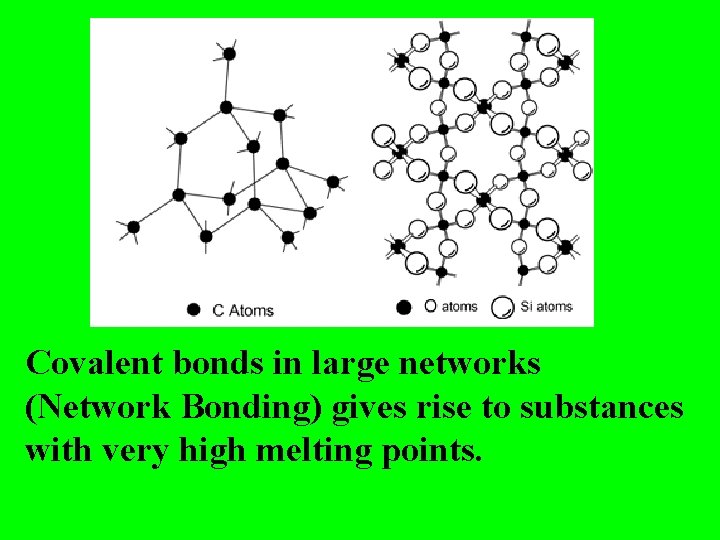
Covalent bonds in large networks (Network Bonding) gives rise to substances with very high melting points.

diamond structure Diamonds are “forever”!

Some melting points of Network Solids: Diamond (Carbon) 3550 o. C Silicon Carbide (Si. C) 2700 o. C Boron Nitride (BN) 3000 o. C Covalent bonds are very strong!

When there is a difference between the electronegativities of two atoms, electrons are shared unequally between them and the bond is called Polar Covalent.

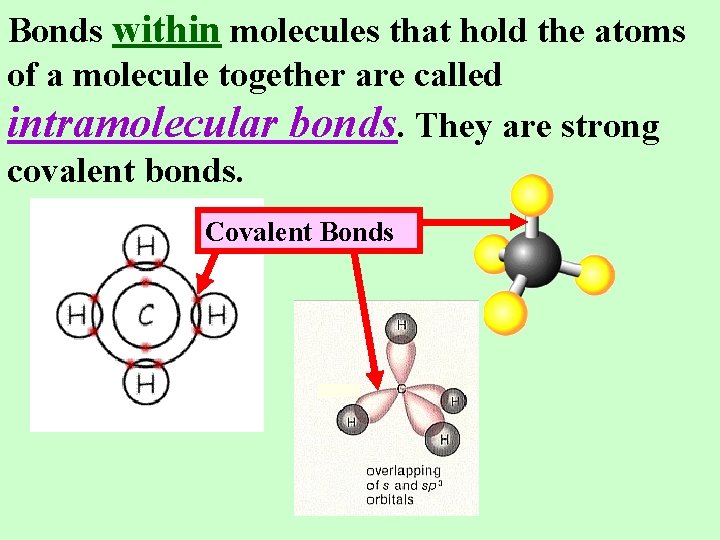
Bonds within molecules that hold the atoms of a molecule together are called intramolecular bonds. They are strong covalent bonds. Covalent Bonds
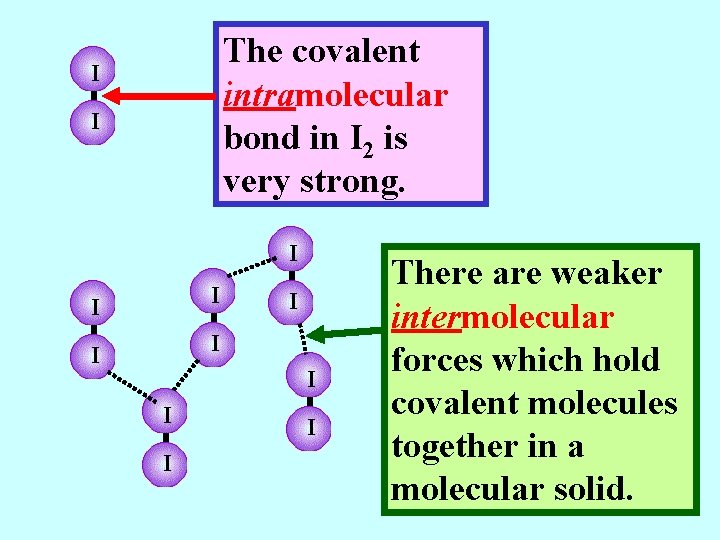
The covalent intramolecular bond in I 2 is very strong. I I I There are weaker intermolecular forces which hold covalent molecules together in a molecular solid.
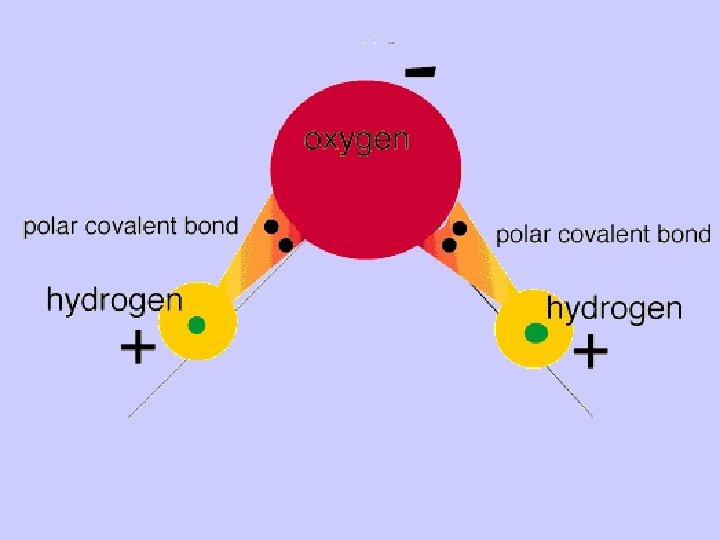

A dipole is a partial separation of charge which exists when one end of a molecule has a slight positive charge and the other end has a slight negative charge. Eg. A water molecule has two dipoles. The Greek letter d “delta” means “partial”
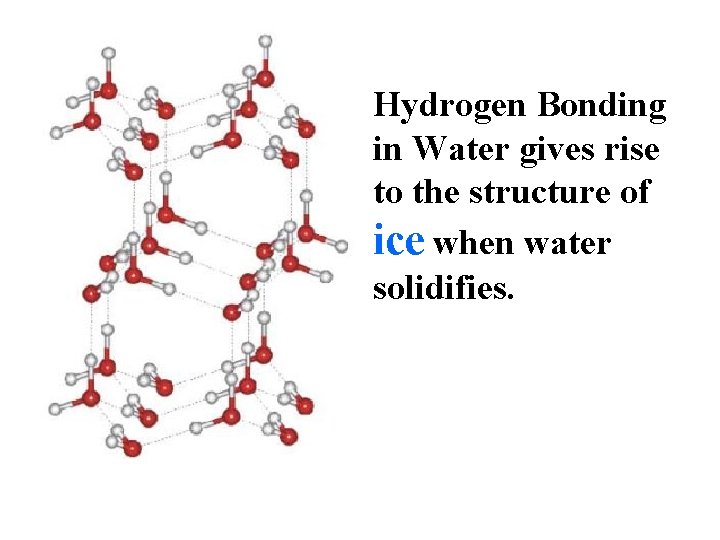
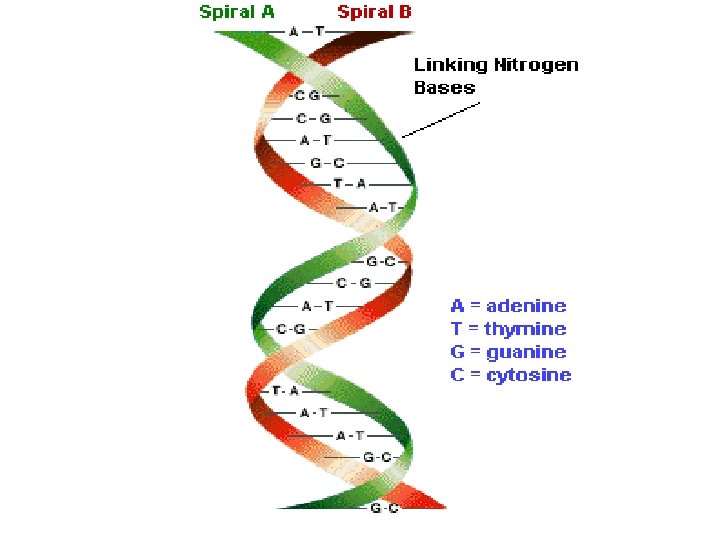
Hydrogen Bonding in Water gives rise to the structure of ice when water solidifies.
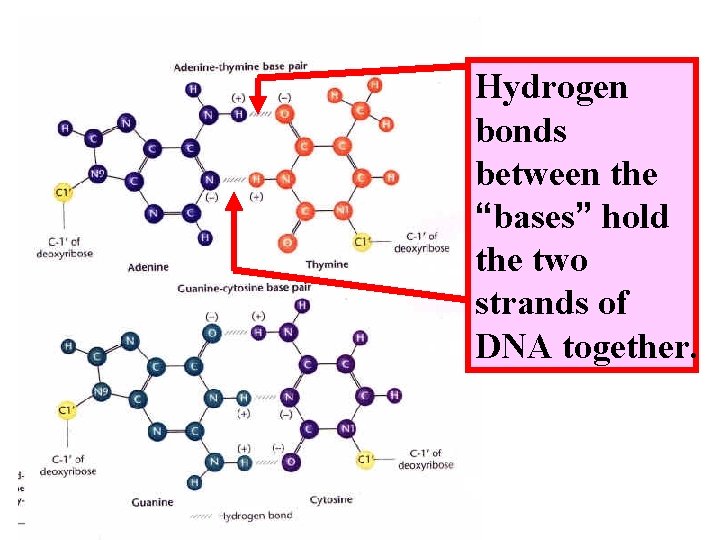
Hydrogen bonds between the “bases” hold the two strands of DNA together.
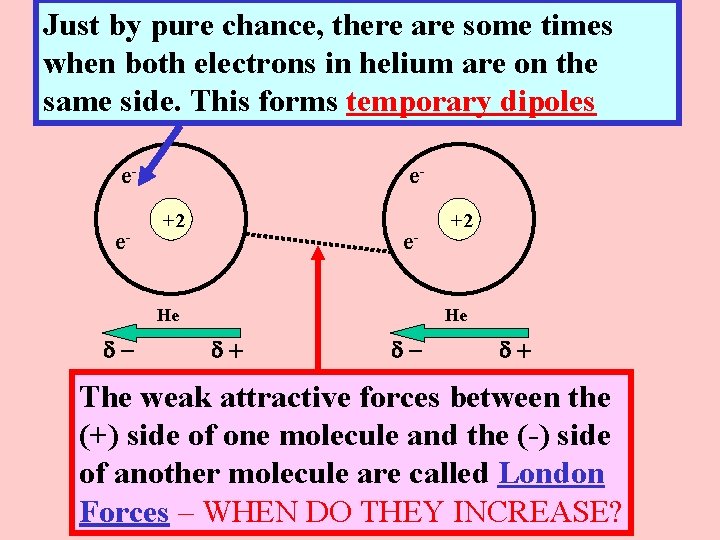
Just by pure chance, there are some times when both electrons in helium are on the same side. This forms temporary dipoles ee- e+2 e- He d- +2 He d+ d- d+ The weak attractive forces between the (+) side of one molecule and the (-) side of another molecule are called London Forces – WHEN DO THEY INCREASE?
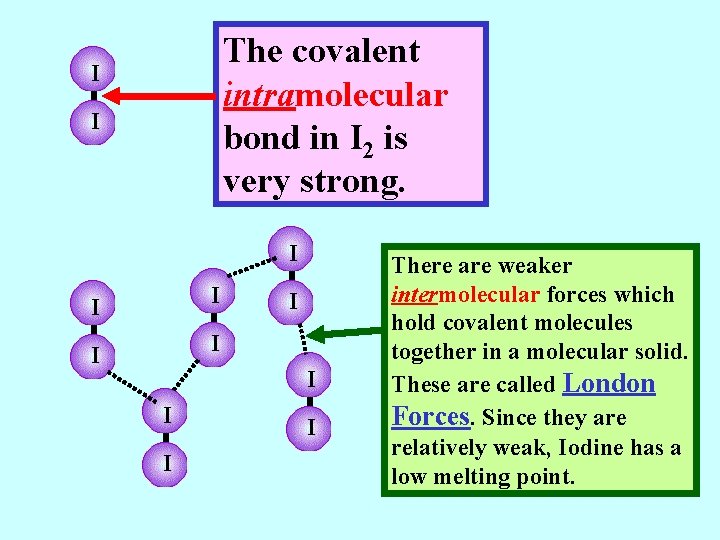
The covalent intramolecular bond in I 2 is very strong. I I I There are weaker intermolecular forces which hold covalent molecules together in a molecular solid. These are called London Forces. Since they are relatively weak, Iodine has a low melting point.

MELTING TEMPERATURE Increases with increasing bond strength *WHY? Think trends… Strength: INTRA > INTER WHY DOES THE MELTING TEMPERATURE INCREASE GOING DOWN THE HALOGEN FAMILY?

METALLIC BONDING A metal is a lattice of positive metal 'ions' in a 'sea' of delocalised electrons. Metallic bonding refers to the interaction between the delocalised electrons and the metal nuclei. WHY DOES THE MELTING TEMPERATURE DECREASE GOING DOWN THE ALKALI METALS?

Gilbert Newton Lewis Invented “Electron-dot” formulas or “Lewis Structures” I’m so tired of writing all those useless inner electrons, in the Bohring models!

Lewis Structures (Electron-dot formulas) for Ionic Compounds. Remember, in an ionic compound, the metal loses e-’s and the non-metal gains. There is no sharing. Here is the e-dot formula for sodium chloride (Na. Cl) Na+ Cl

Here is the e-dot formula (Lewis Structure) for the ionic compound Mg. F 2 : F Mg 2+ F Notice, there is no sharing. The F atoms took both valence e-’s from Mg, forming ions which do not share electrons. The + and – charges on the ions cause them to attract each other.

Electron-dot Formulas (Lewis Structures) for Covalent Compounds. When atoms form covalent bonds, they are trying to achieve stable noble gas electron arrangements: Hydrogen will share e-’s until it feels 2 e-’s like Helium. Other elements share e-’s to achieve what is called a “Stable Octet” (8 valence e-’s)
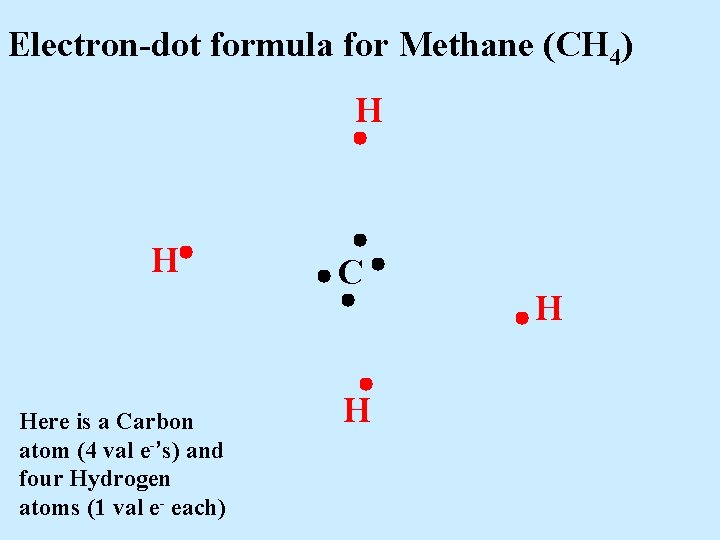
Electron-dot formula for Methane (CH 4) H H Here is a Carbon atom (4 val e-’s) and four Hydrogen atoms (1 val e- each) C H H
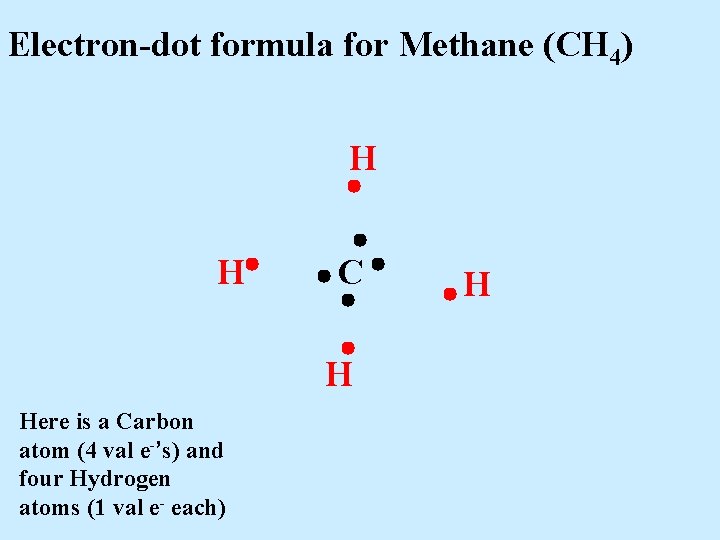
Electron-dot formula for Methane (CH 4) H H C H Here is a Carbon atom (4 val e-’s) and four Hydrogen atoms (1 val e- each) H
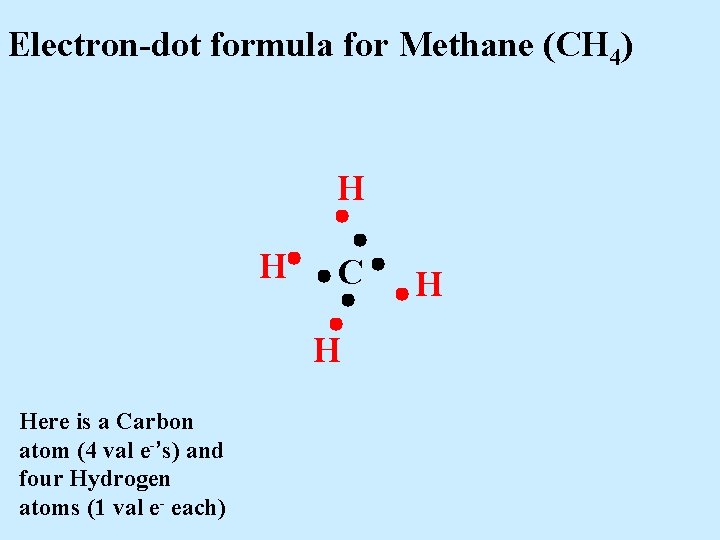
Electron-dot formula for Methane (CH 4) H H C H Here is a Carbon atom (4 val e-’s) and four Hydrogen atoms (1 val e- each) H
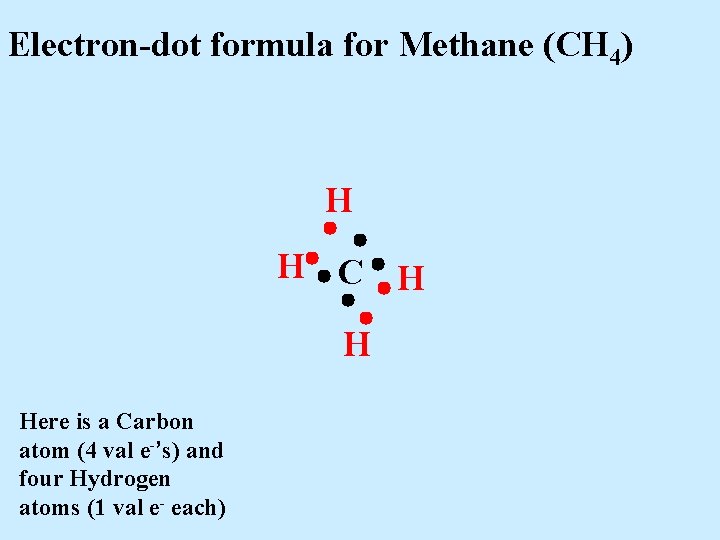
Electron-dot formula for Methane (CH 4) H H C H H Here is a Carbon atom (4 val e-’s) and four Hydrogen atoms (1 val e- each)
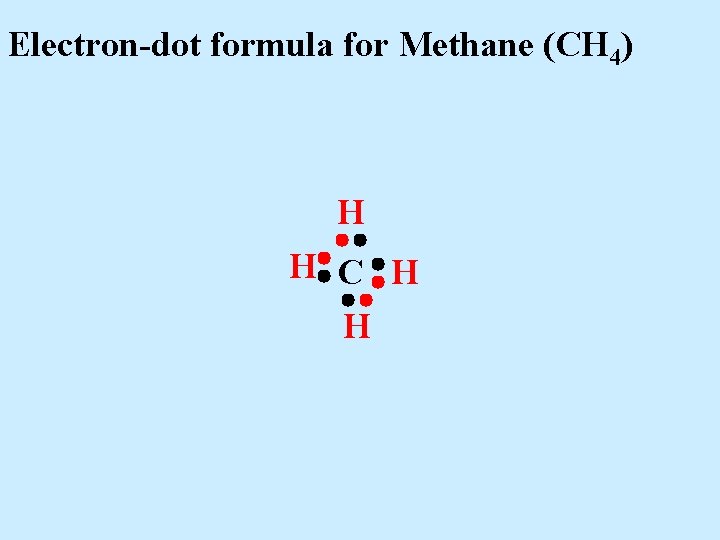
Electron-dot formula for Methane (CH 4) H H C H H
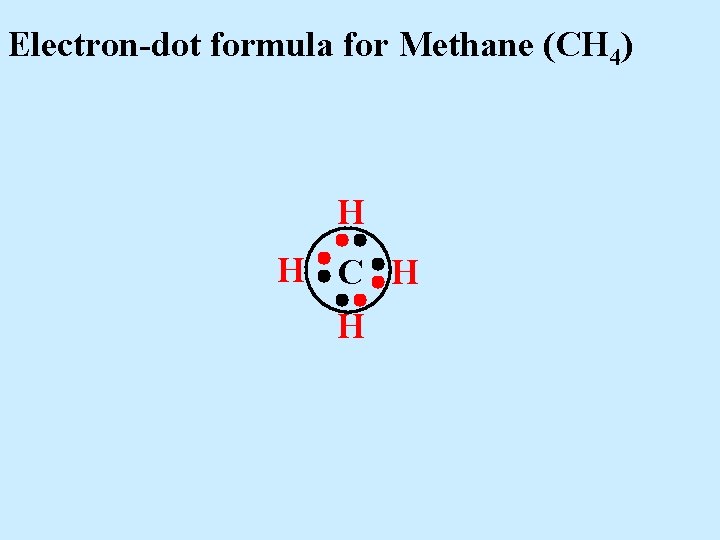
Electron-dot formula for Methane (CH 4) H H C H H
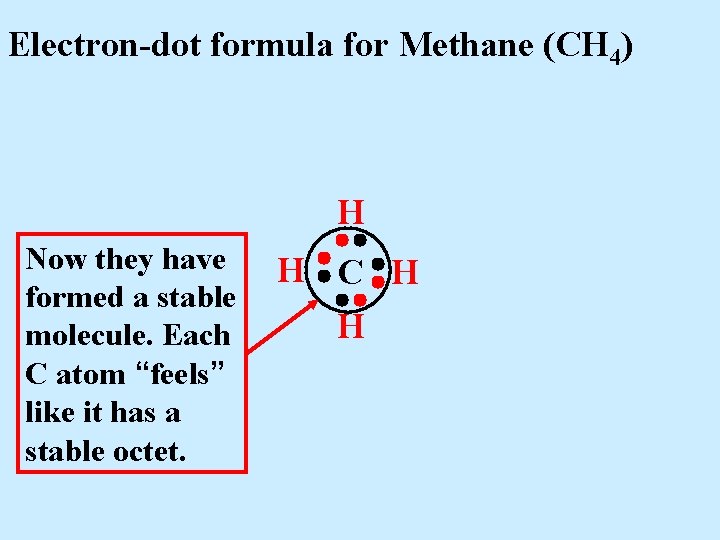
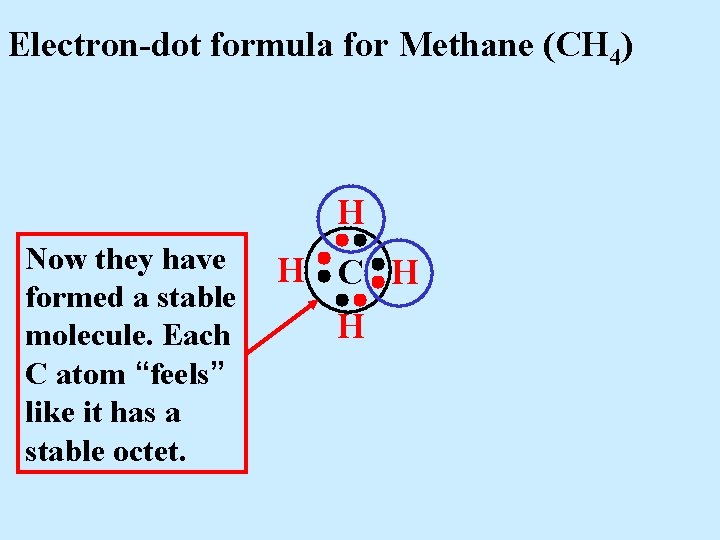
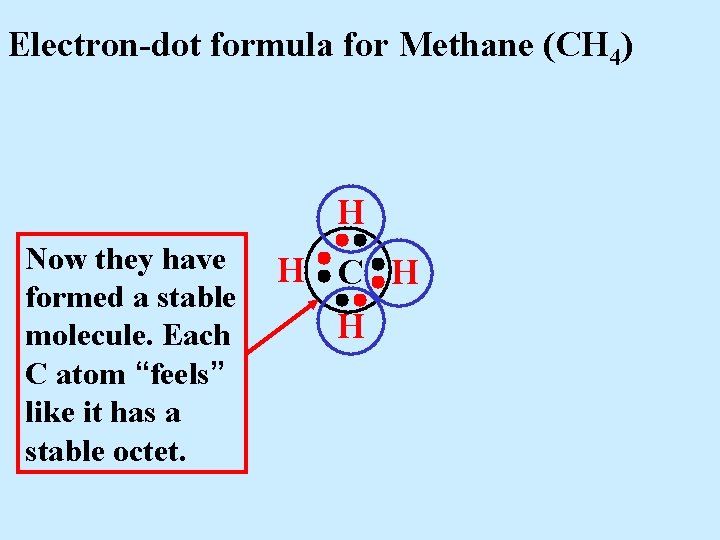
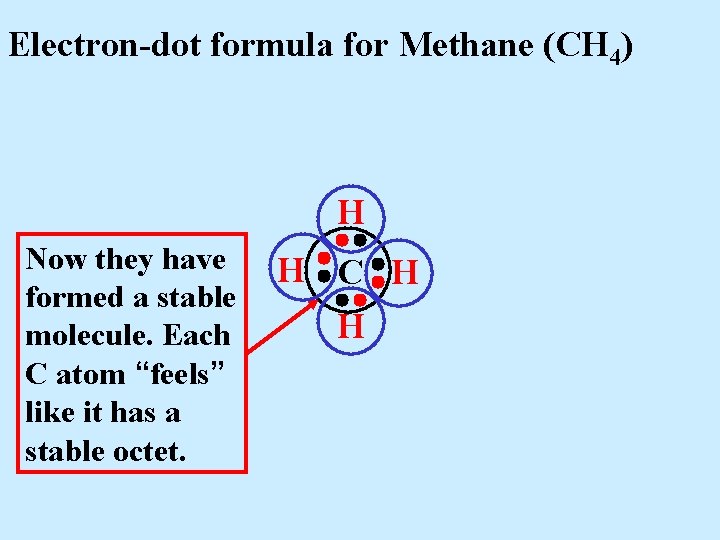
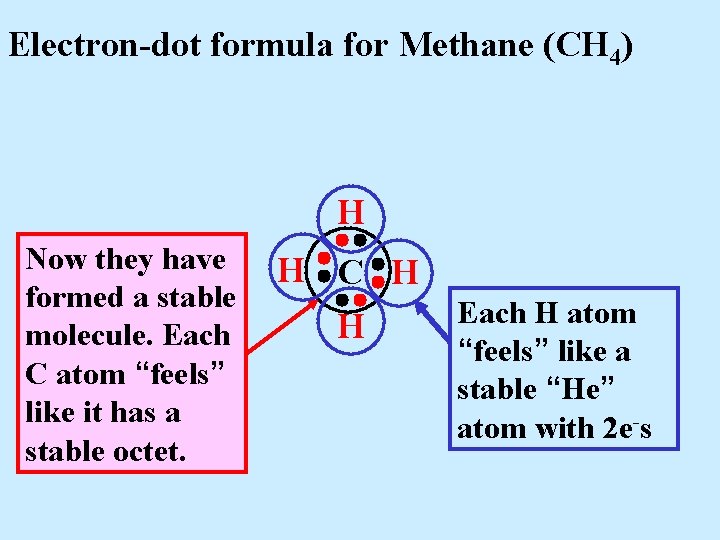
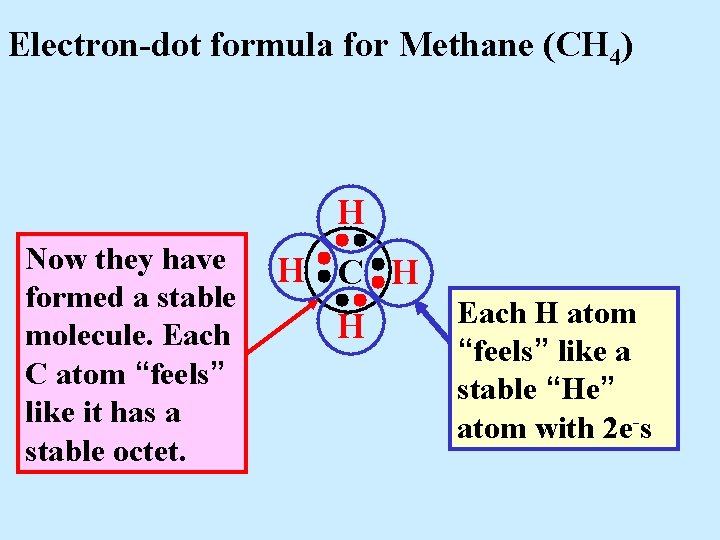
Electron-dot formula for Methane (CH 4) H Now they have formed a stable molecule. Each C atom “feels” like it has a stable octet. H C H H
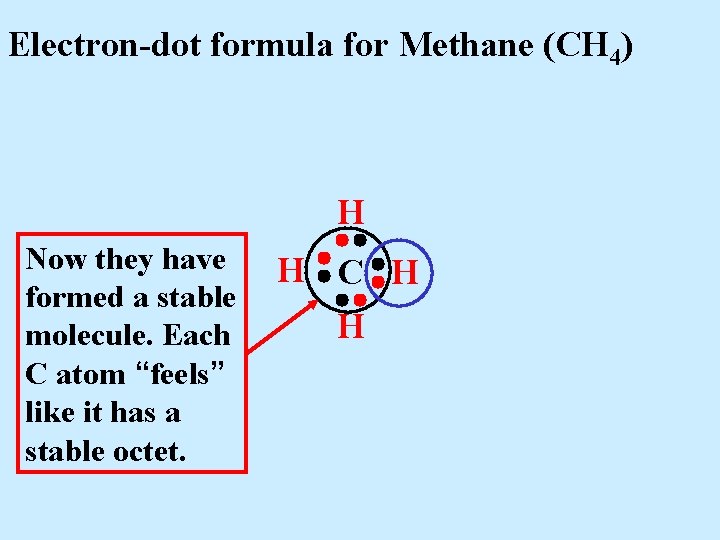
Electron-dot formula for Methane (CH 4) H Now they have formed a stable molecule. Each C atom “feels” like it has a stable octet. H C H H
Electron-dot formula for Methane (CH 4) H Now they have formed a stable molecule. Each C atom “feels” like it has a stable octet. H C H H
Electron-dot formula for Methane (CH 4) H Now they have formed a stable molecule. Each C atom “feels” like it has a stable octet. H C H H
Electron-dot formula for Methane (CH 4) H Now they have formed a stable molecule. Each C atom “feels” like it has a stable octet. H C H H
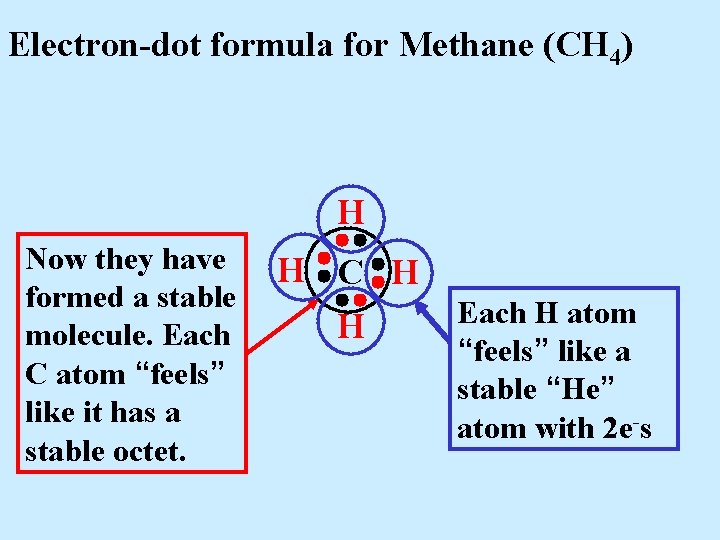
Electron-dot formula for Methane (CH 4) H Now they have formed a stable molecule. Each C atom “feels” like it has a stable octet. H C H H Each H atom “feels” like a stable “He” atom with 2 e-s
Electron-dot formula for Methane (CH 4) H Now they have formed a stable molecule. Each C atom “feels” like it has a stable octet. H C H H Each H atom “feels” like a stable “He” atom with 2 e-s
Electron-dot formula for Methane (CH 4) H Now they have formed a stable molecule. Each C atom “feels” like it has a stable octet. H C H H Each H atom “feels” like a stable “He” atom with 2 e-s
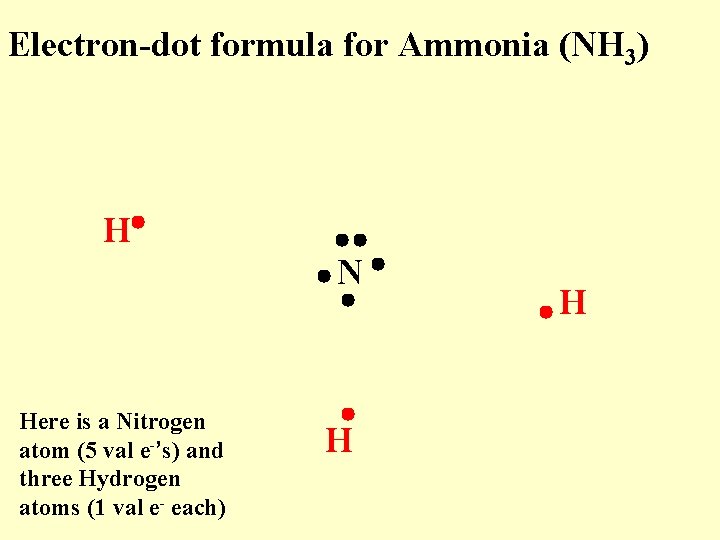
Electron-dot formula for Ammonia (NH 3) H N Here is a Nitrogen atom (5 val e-’s) and three Hydrogen atoms (1 val e- each) H H
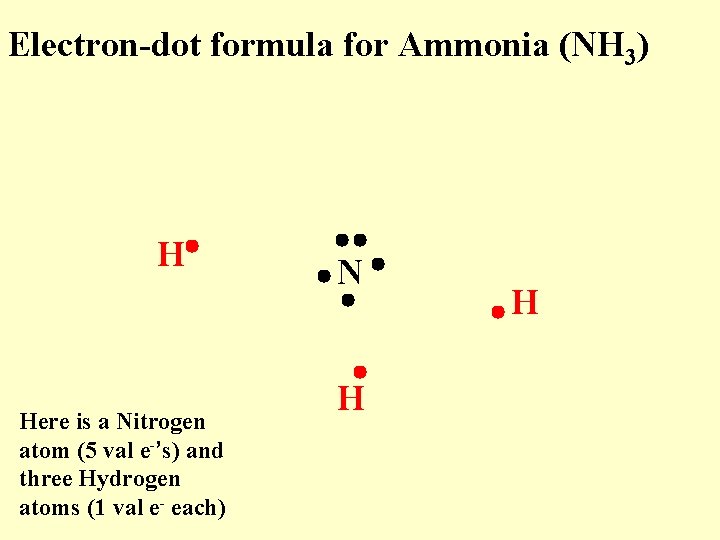
Electron-dot formula for Ammonia (NH 3) H Here is a Nitrogen atom (5 val e-’s) and three Hydrogen atoms (1 val e- each) N H H
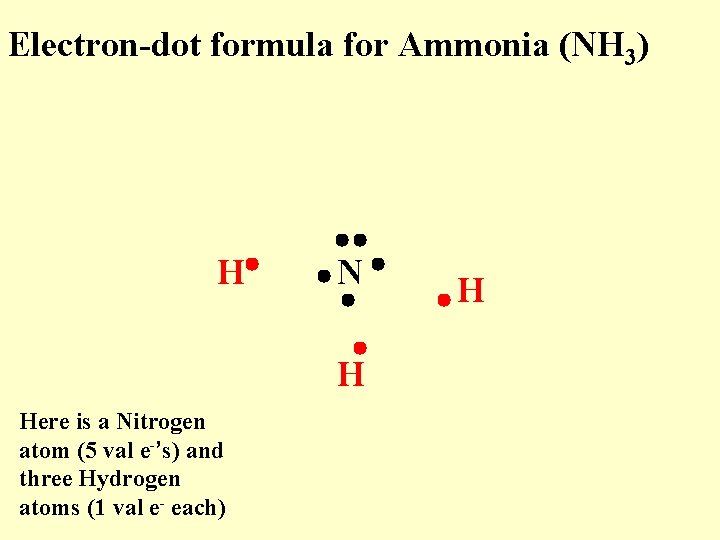
Electron-dot formula for Ammonia (NH 3) H N H Here is a Nitrogen atom (5 val e-’s) and three Hydrogen atoms (1 val e- each) H
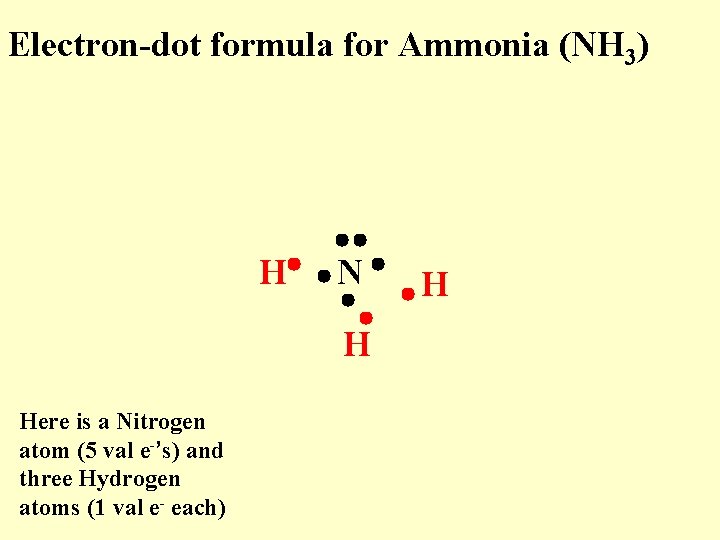
Electron-dot formula for Ammonia (NH 3) H N H Here is a Nitrogen atom (5 val e-’s) and three Hydrogen atoms (1 val e- each) H
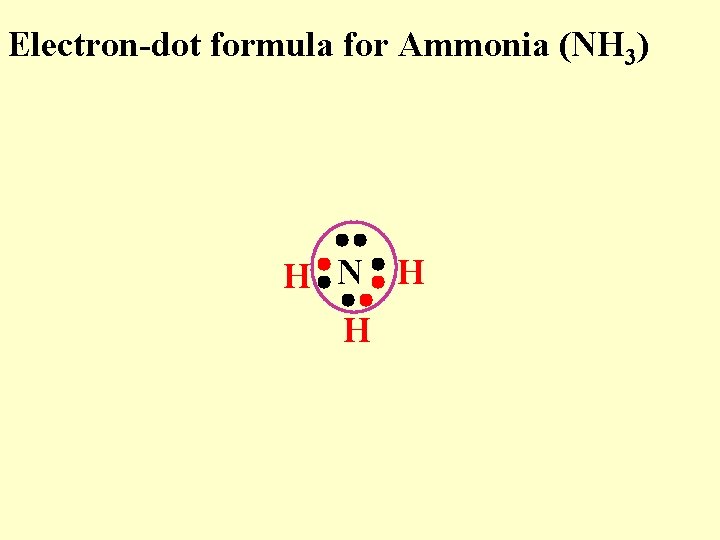
Electron-dot formula for Ammonia (NH 3) H N H H
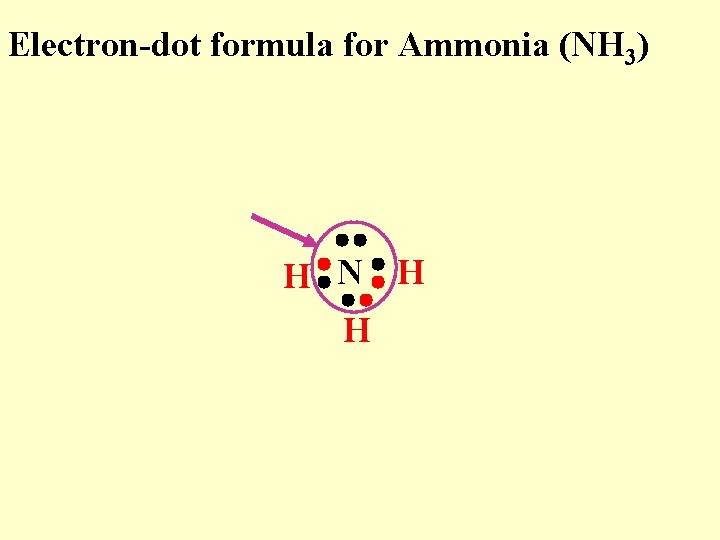
Electron-dot formula for Ammonia (NH 3) H N H H
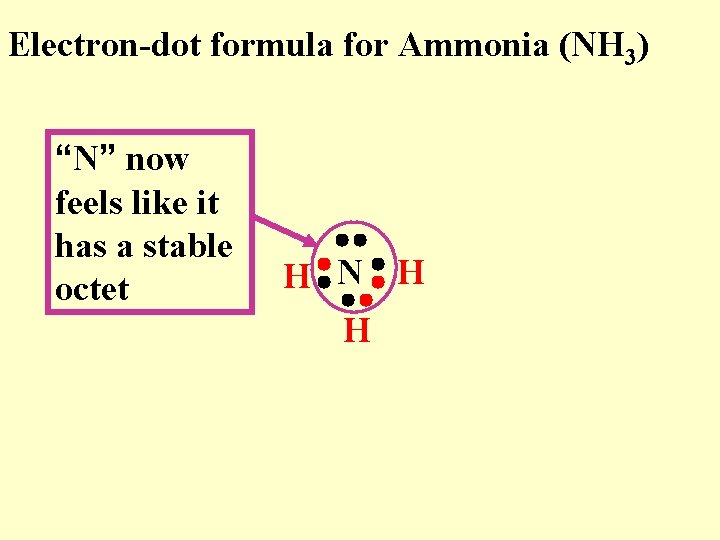
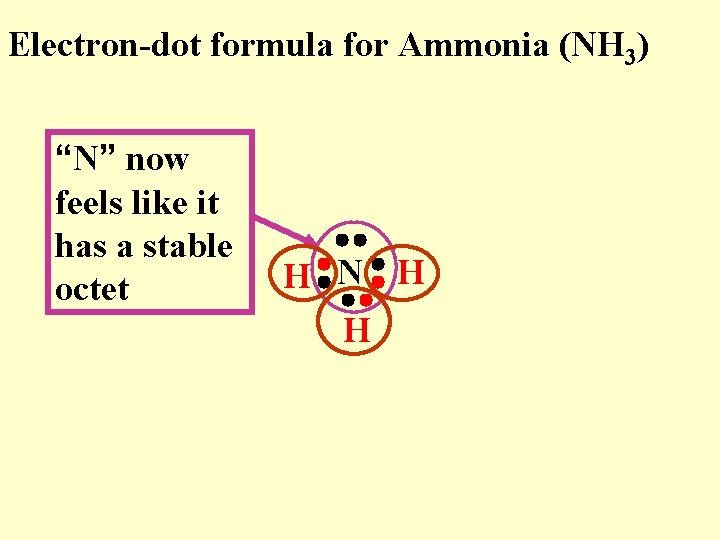
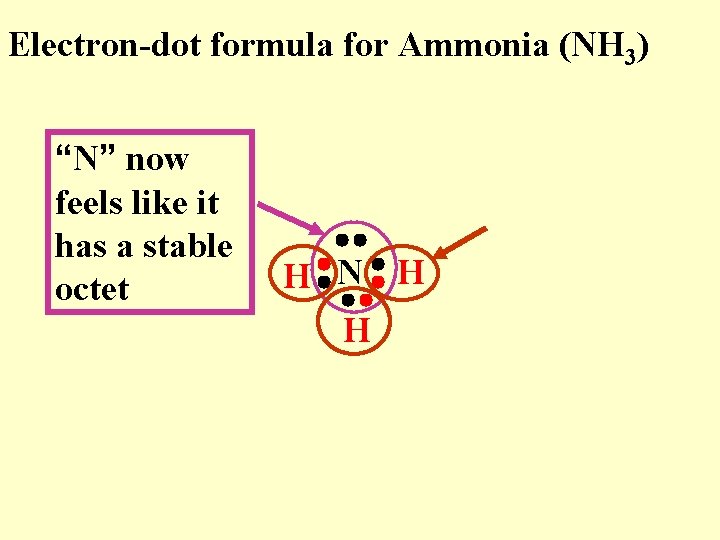
Electron-dot formula for Ammonia (NH 3) “N” now feels like it has a stable octet H N H H
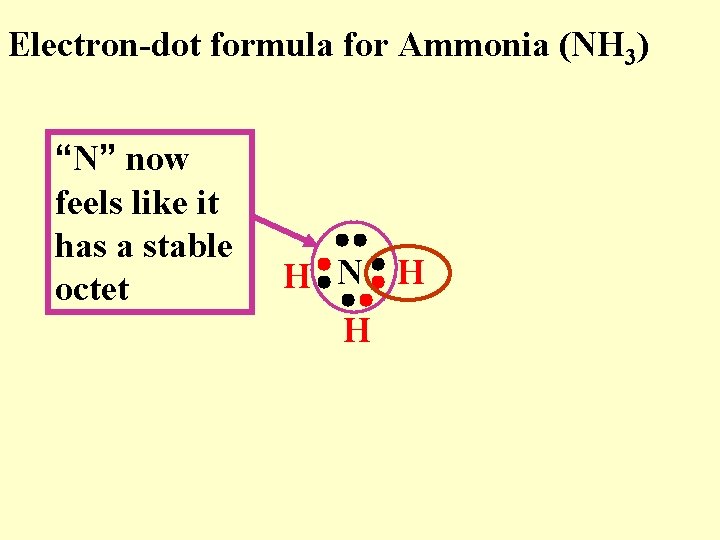
Electron-dot formula for Ammonia (NH 3) “N” now feels like it has a stable octet H N H H
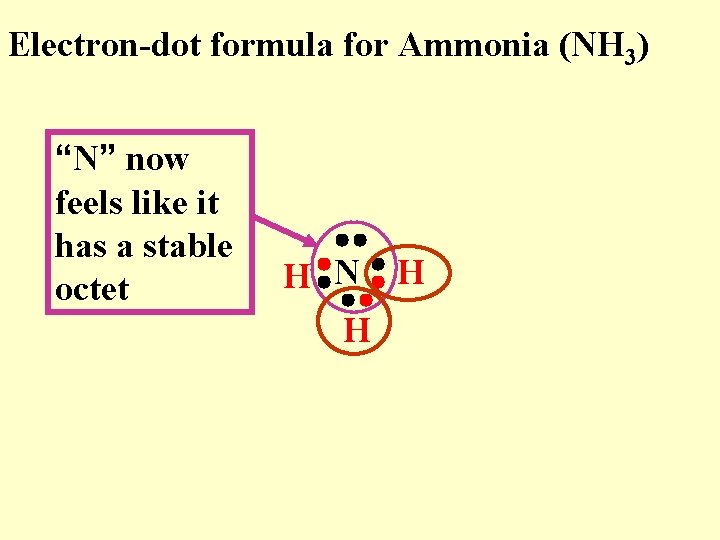
Electron-dot formula for Ammonia (NH 3) “N” now feels like it has a stable octet H N H H
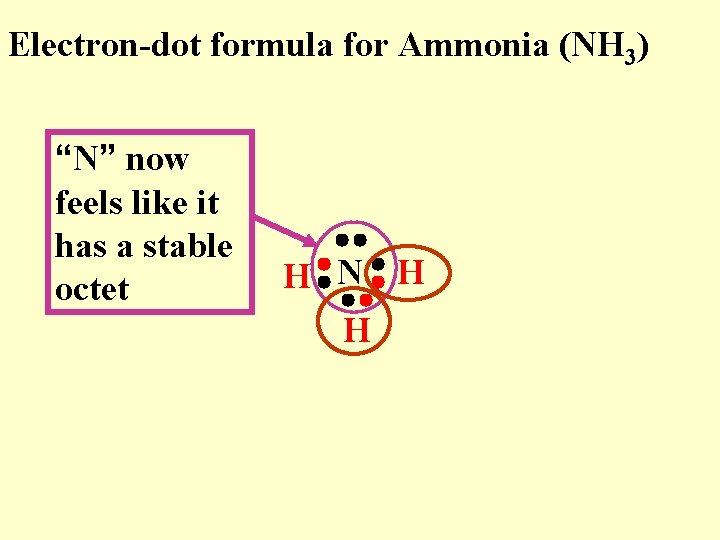
Electron-dot formula for Ammonia (NH 3) “N” now feels like it has a stable octet H N H H
Electron-dot formula for Ammonia (NH 3) “N” now feels like it has a stable octet H N H H
Electron-dot formula for Ammonia (NH 3) “N” now feels like it has a stable octet H N H H
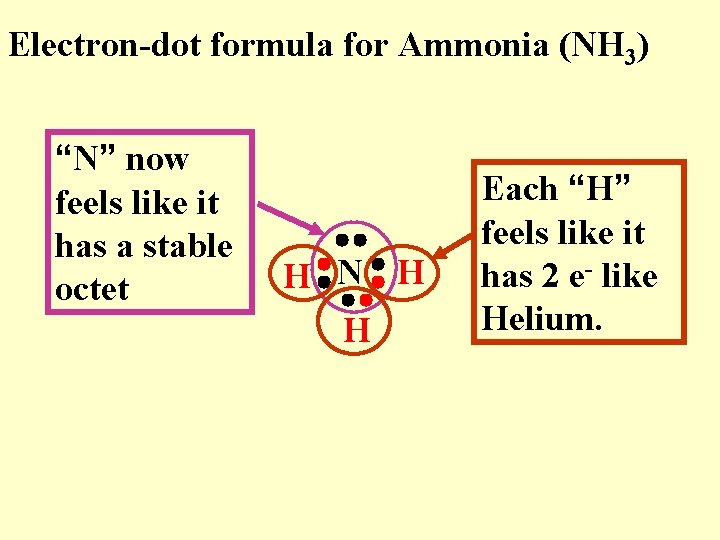
Electron-dot formula for Ammonia (NH 3) “N” now feels like it has a stable octet H N H H Each “H” feels like it has 2 e- like Helium.
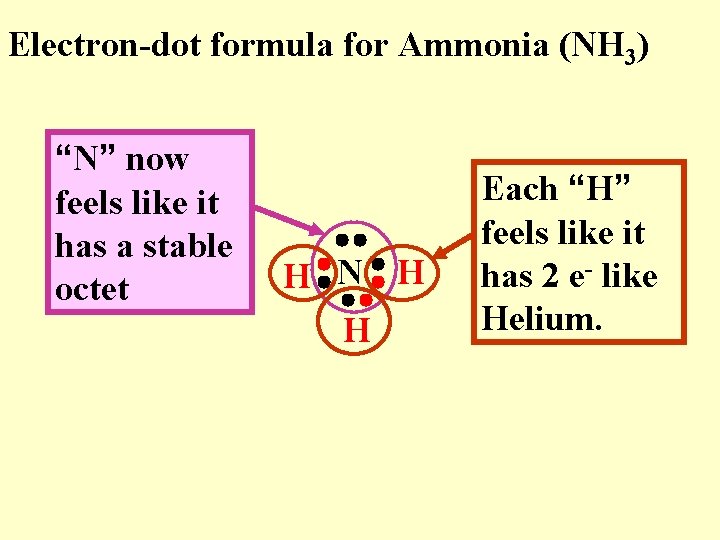
Electron-dot formula for Ammonia (NH 3) “N” now feels like it has a stable octet H N H H Each “H” feels like it has 2 e- like Helium.
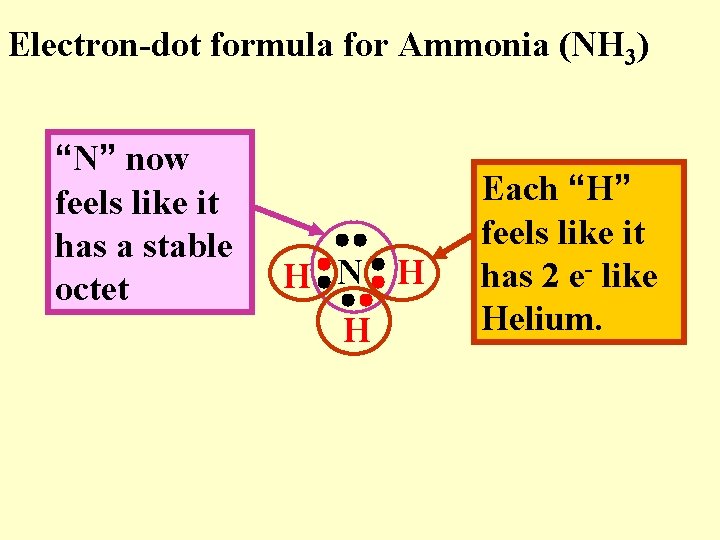
Electron-dot formula for Ammonia (NH 3) “N” now feels like it has a stable octet H N H H Each “H” feels like it has 2 e- like Helium.
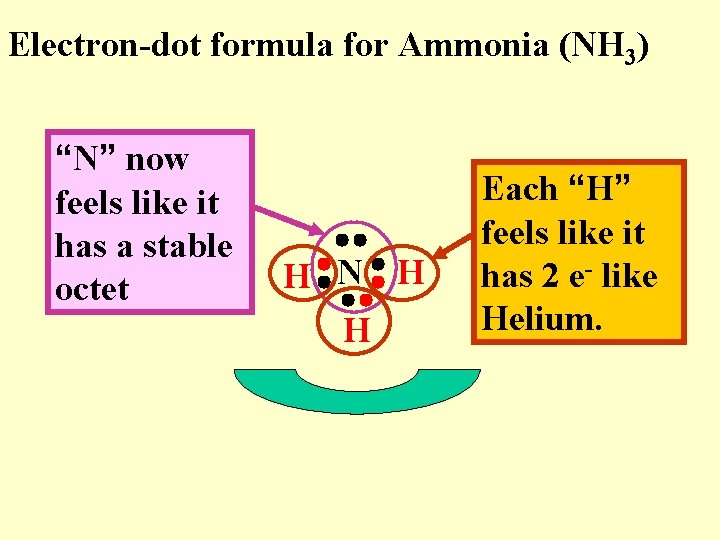
Electron-dot formula for Ammonia (NH 3) “N” now feels like it has a stable octet H N H H Each “H” feels like it has 2 e- like Helium.
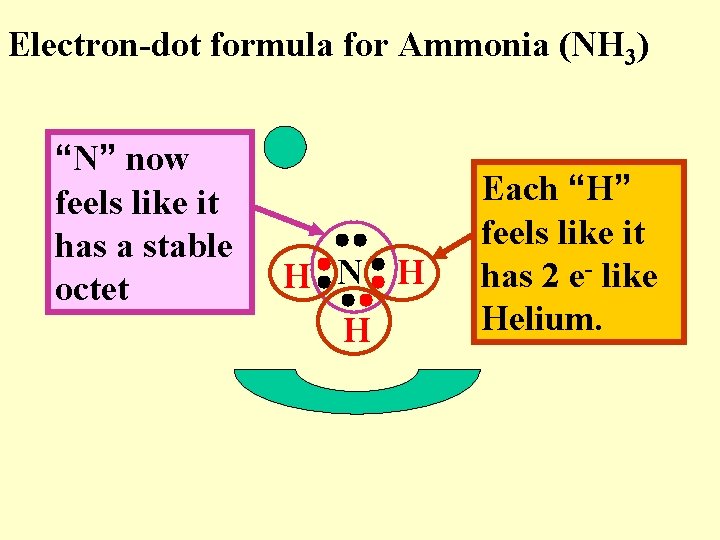
Electron-dot formula for Ammonia (NH 3) “N” now feels like it has a stable octet H N H H Each “H” feels like it has 2 e- like Helium.
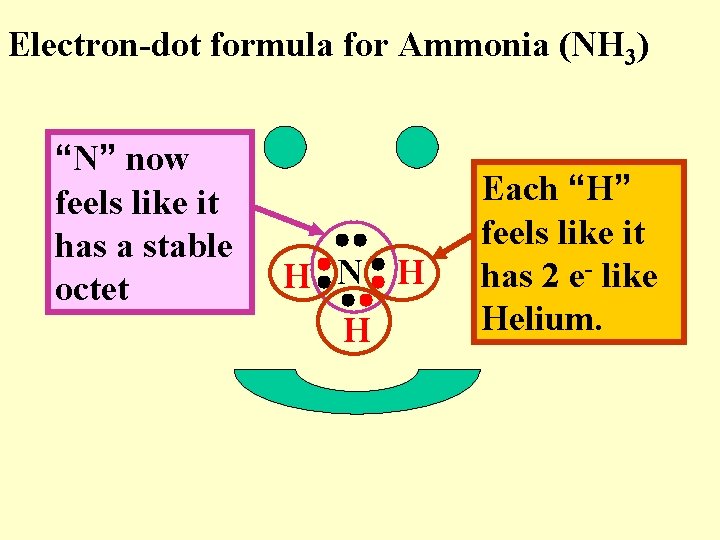
Electron-dot formula for Ammonia (NH 3) “N” now feels like it has a stable octet H N H H Each “H” feels like it has 2 e- like Helium.

Electron-dot formula for Ammonia (NH 3) H N H H Each “H” feels like it has 2 e- like Helium.
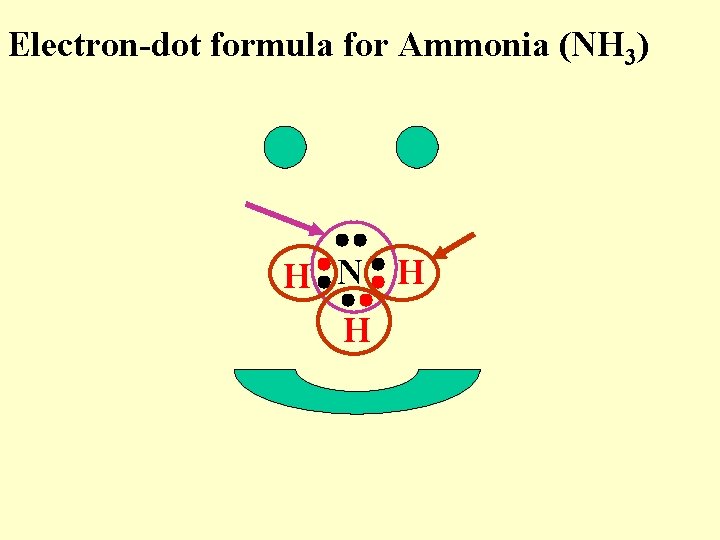
Electron-dot formula for Ammonia (NH 3) H N H H
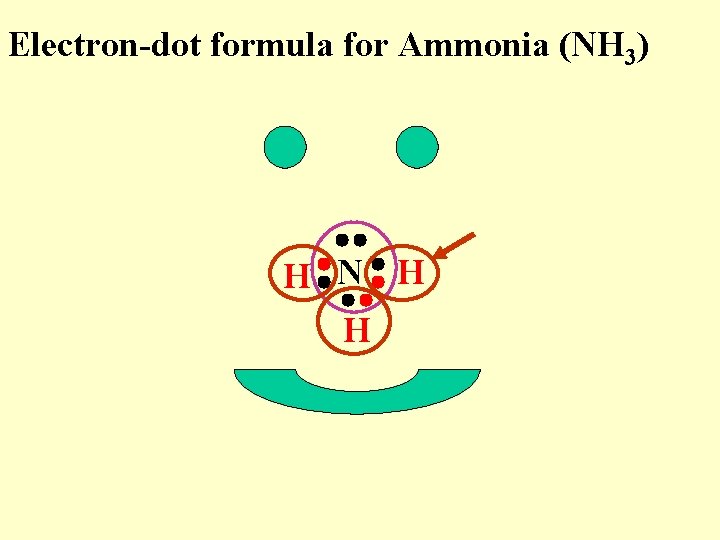
Electron-dot formula for Ammonia (NH 3) H N H H
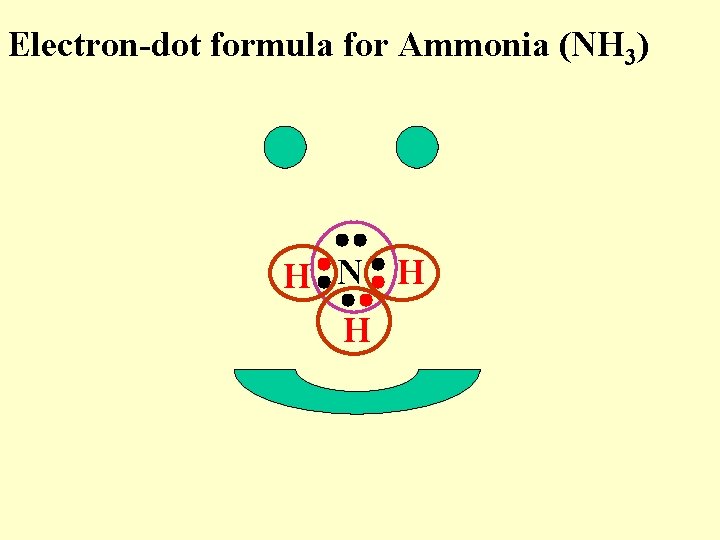
Electron-dot formula for Ammonia (NH 3) H N H H
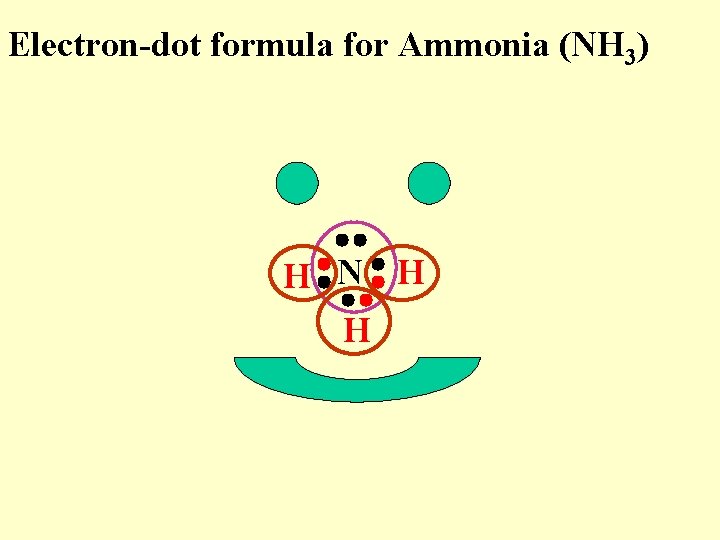
Electron-dot formula for Ammonia (NH 3) H N H H
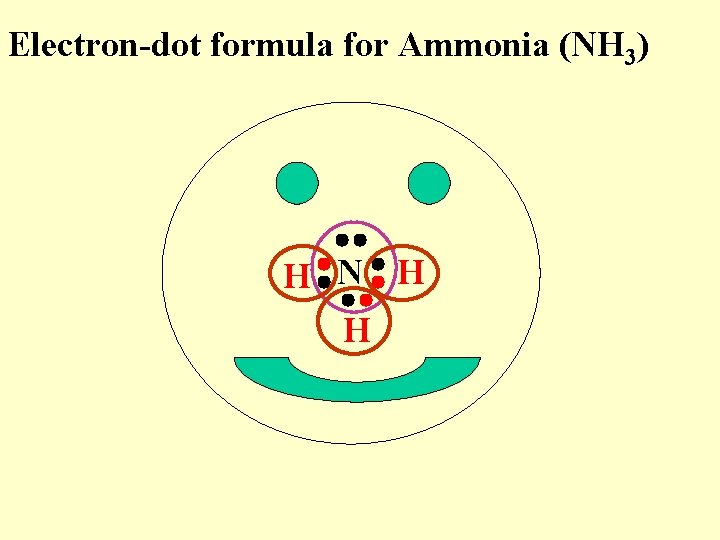
Electron-dot formula for Ammonia (NH 3) H N H H
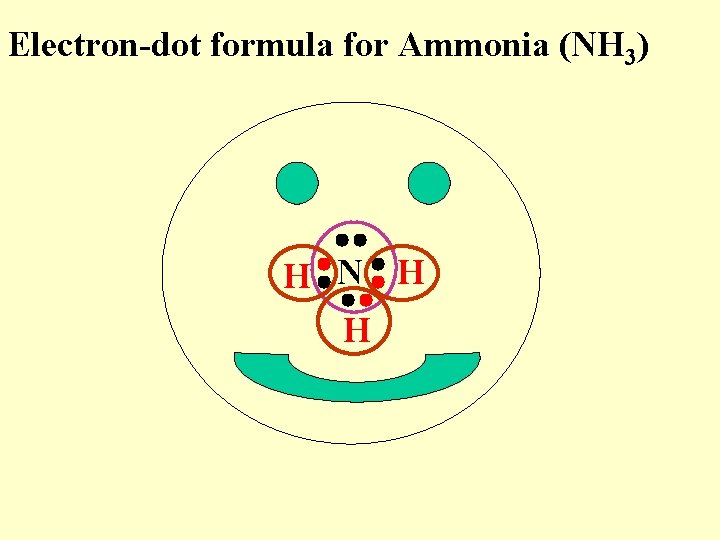
Electron-dot formula for Ammonia (NH 3) H N H H
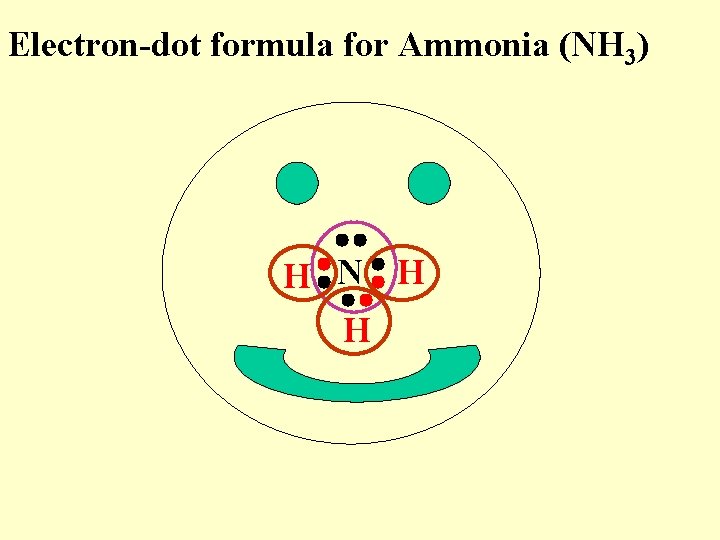
Electron-dot formula for Ammonia (NH 3) H N H H
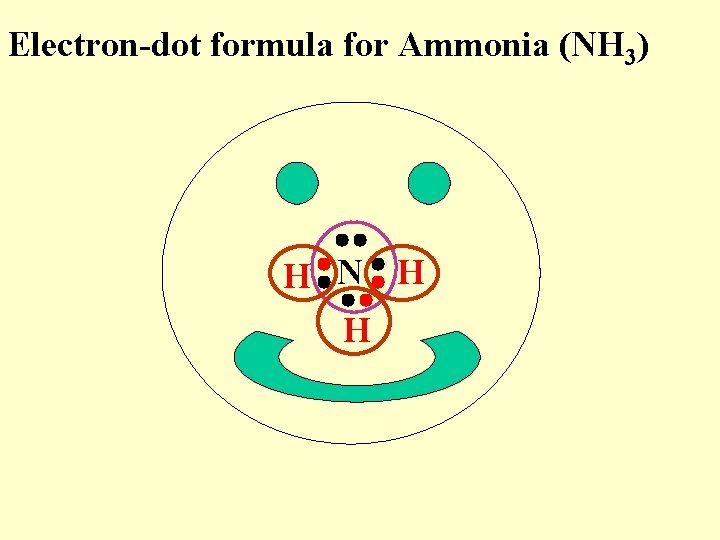
Electron-dot formula for Ammonia (NH 3) H N H H
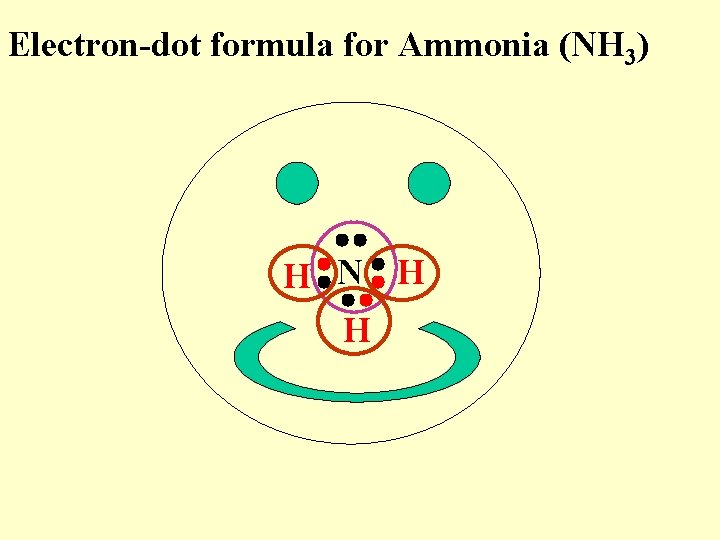
Electron-dot formula for Ammonia (NH 3) H N H H
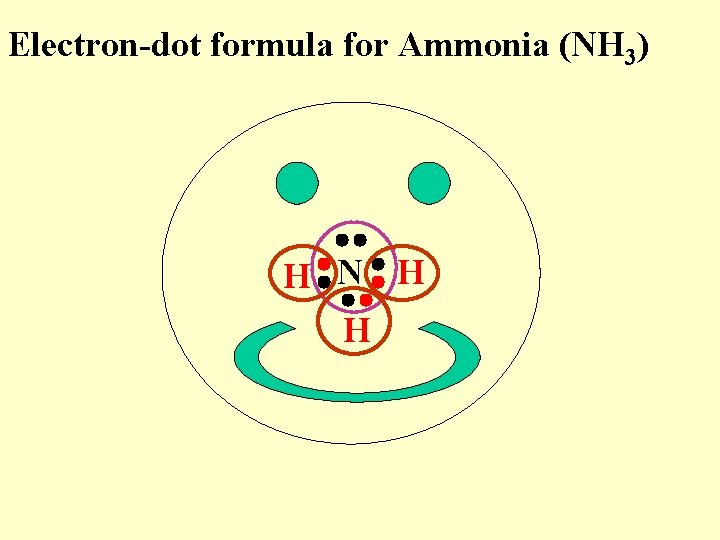
Electron-dot formula for Ammonia (NH 3) H N H H
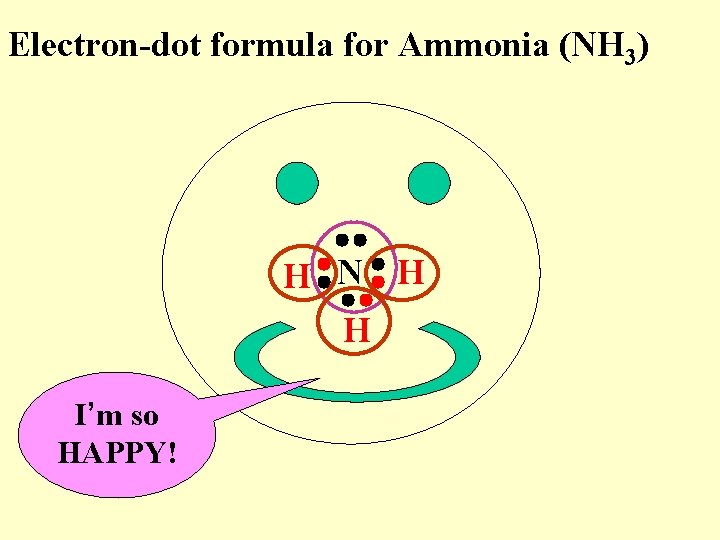
Electron-dot formula for Ammonia (NH 3) H N H H I’m so HAPPY!

Write the electron-dot formula for CF 4
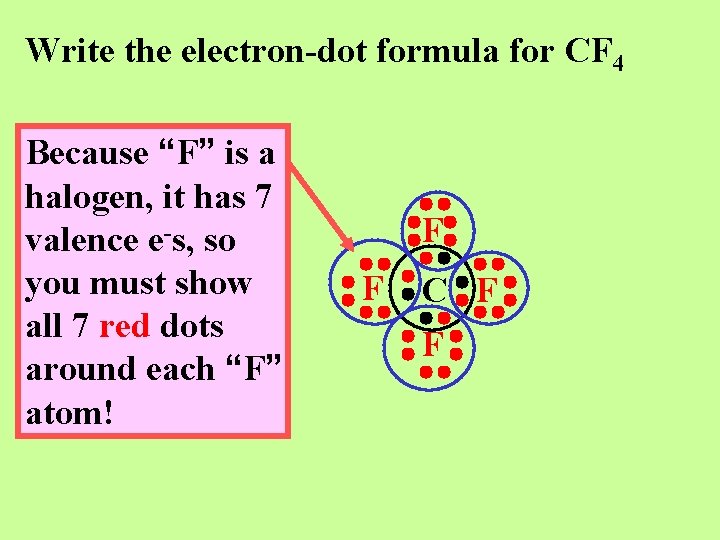
Write the electron-dot formula for CF 4 Because “F” is a halogen, it has 7 valence e-s, so you must show all 7 red dots around each “F” atom! F F C F F

Write the electron-dot formula for H 2 S
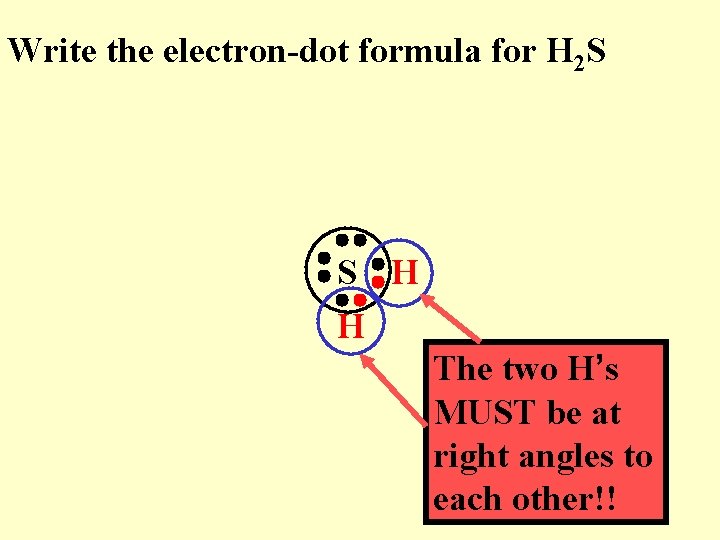
Write the electron-dot formula for H 2 S S H H The two H’s MUST be at right angles to each other!!
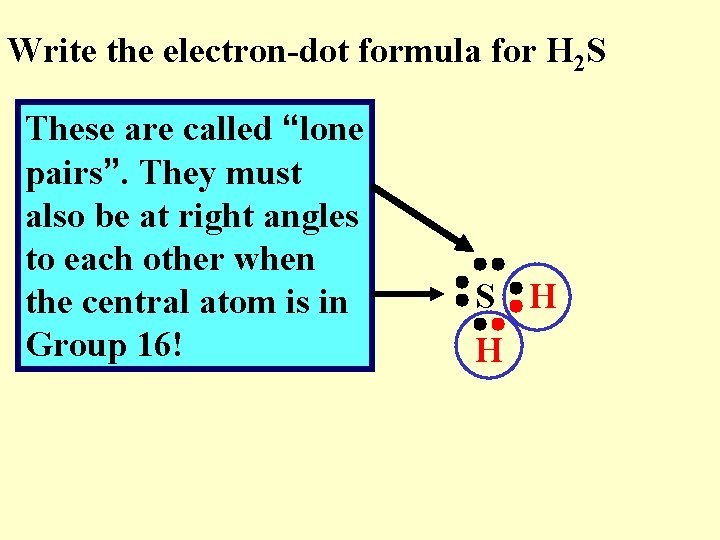
Write the electron-dot formula for H 2 S These are called “lone pairs”. They must also be at right angles to each other when the central atom is in Group 16! S H H

Write the Electron-Dot Formula for Se. F 2

Write the Electron-Dot Formula for Se. F 2 Se F F
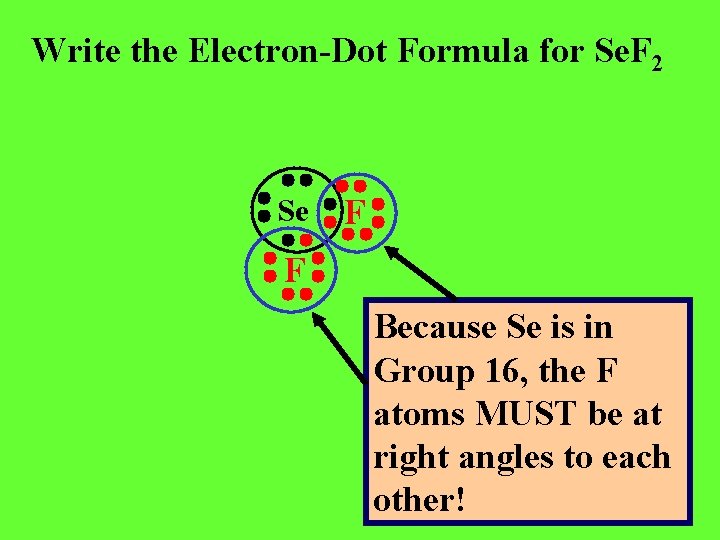
Write the Electron-Dot Formula for Se. F 2 Se F F Because Se is in Group 16, the F atoms MUST be at right angles to each other!

Write the Electron-Dot Formula for Se. F 2 Se F F Because “F” is in Group 17, they have 7 valence e-s, so they must have 7 red dots around them.
- Slides: 84