Chemistry 01 Quantitative Chemistry BY HEI MAN KWOK

Chemistry 01: Quantitative Chemistry BY HEI MAN KWOK 12 N 03 S

1. 1 MOLE CONCEPT + AVOGADRO’S CONSTANT

Terminology Clear up! • Element: contains only one type of atom • Compound: elements fixed chemically in specific proportions • Mixture: contains different types of particles without any fixed proportion • Molecule: small group of atoms joined together by covalent bonds • Particles:

What is a Mole? • 6. 02 x 10²³ units of any substance – Avogadro’s Number • How many particles in 2 moles of Carbon? • 2 x 6. 02 x 10²³ = 1. 204 x 10²⁴ particles

1. 2 FORMULAS

Definitions • Relative Atomic mass (Ar): average mass of an atom, taking into account the relative abundances of all the naturally occurring isotopes of the element, relative to one atom of C-12 – no units cuz its relative Eg. H = 1, O = 16 • Relative Molecular mass (Mr): average mass of a molecule, calculated by adding the relative atomic masses of its constituent atoms – no units cuz its relative Eg. H₂O = 18, Cl₂ = 71

Molar Mass • Molar Mass (m): mass of one mole of any substance expressed in grams per mole • No. of Moles (mols) = Mass (g) / Molar Mass (g/mol) [mass/ mern lolz]

Empirical and Molecular Formula • Empirical Formula: of a compound is the simplest whole number ratio of the atoms it contains – eg. Ethane: CH₂ • Molecular Formula: of a compound is the actual number of atoms of each element present; multiple of its empirical formula – eg. Ethane: C₂H₄

Finding Empirical Formula • When hydrated Magnesium Sulphate, Mg. SO₄. n. H₂O was heated until there was no further mass decrease. 4. 58 g of anhydrous magnesium was left behind and 2. 86 g of water was released Compound Mg. SO₄ H₂O Mass/percentage 4. 58 2. 86 Mr 120 18 Number of moles (m/Mr) 0. 039 0. 27 Simplest Ratio of moles 0. 039/0. 039 0. 27/0. 039= (divide by smallest) =1 6. 93 Empirical formula Mg. SO₄. 7 H₂O

Finding Molecular Formula • Find the Molecular Formula of a compound which has CH₂ as its empirical formula and has a molecular mass of 84 g 1. Mass of empirical formula: CH₂ = 12 + 1 = 14 2. Molecular Mass/ Empirical Mass = constant (k) [84 g/ 14 g = 6] 3. Constant (k) x empirical formula = 6 x CH₂

1. 3 CHEMICAL EQUATIONS
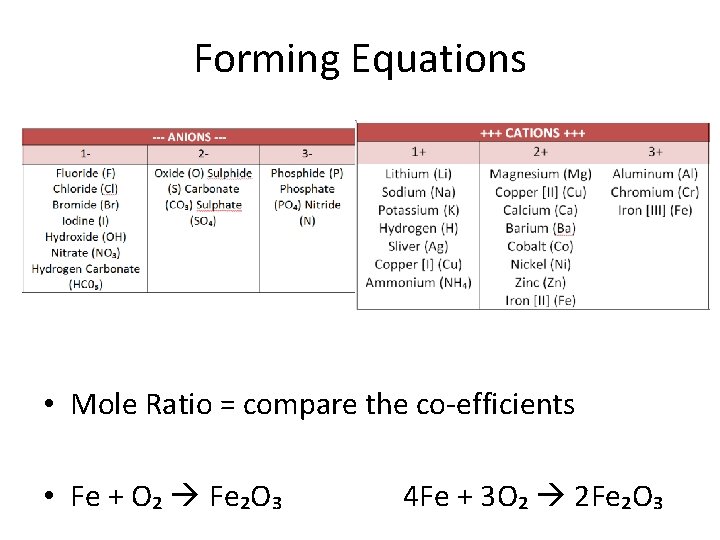
Forming Equations • Mole Ratio = compare the co-efficients • Fe + O₂ Fe₂O₃ 4 Fe + 3 O₂ 2 Fe₂O₃

1. 4 MASS AND GASEOUS VOLUME RELATIONSHIPS IN CHEM. REACTIONS

Percentage Yield limiting and excess reactant • Percentage of theoretical yield can be deduced from the chemical equation • Take into consideration the limiting reactant and excess reactant • Percentage Yield = Obtained Yield/ Theoretical Yield x 100%

Avogadro’s Law (Hypothesis) • Equal volumes of different gases contain equal numbers of particles at the same temp. and pressure • ^ implications – no need to convert volume/ mass to moles = can directly calculate

Molar Volume •

Combined Gas Law •

Ideal Gas Equation •

Ideal Gas Graph

1. 5 SOLUTIONS

Solution Terminology • Solute: substance dissolved in a solvent in forming a solution • Solvent: liquid that dissolves another solvent or substances to form a solution • Solution: homogeneous mixture of a liquid (a solvent) with another substance (the solute) – there is usually some interaction between the solvent and solute molecules • Concentration: amount of solute in a known volume of solution – in mol dm⁻ᶟ or g m⁻ᶟ

Concentration in Liquids/ Molarity • No. of Moles (mols) = Concentration (mols/dm³) x Volume (dm³) • eg. Find the Molarity of a 50 dm³ solution with 5 mols of Na. Cl in it • Concentration = 5 / 50 = 0. 1 mols/dm³ • REMARK: 1 dm³ = 1 L = 1000 cm³ = 1000 ml
- Slides: 22