Chemical Thermodynamics Entropy Free Energy and Equilibrium Chapter

Chemical Thermodynamics: Entropy, Free Energy and Equilibrium Chapter 16

Chemical Thermodynamics n Science of interconversion of energy n n n Heat into other forms of energy Amount of heat gained/released from a system Spontaneity of a reaction Gibbs free energy function Relationship between Gibbs Free Energy and chemical equilibrium

Spontaneous Processes n n n Main objective Spontaneous Reaction- a reaction does occur under specific conditions Non-spontaneous Reaction- a reaction does not occur under specific conditions

Spontaneous Processes n n n A waterfall runs downhill A lump of sugar dissolves in a cup of coffee At 1 atm, water freezes below 0ºC and ice melts above 0ºC Heat flows from a hotter object to a colder object Iron exposed to oxygen and water forms rust

Spontaneous Processes

Spontaneous Processes Does a decrease in enthalpy mean a reaction proceeds spontaneously? Spontaneous reactions CH 4 (g) + 2 O 2 (g) H+ (aq) + OH- (aq) H 2 O (s) NH 4 NO 3 (s) CO 2 (g) + 2 H 2 O (l) DH 0 = -890. 4 k. J H 2 O (l) DH 0 = -56. 2 k. J H 2 O (l) DH 0 = 6. 01 k. J H 2 O NH 4+(aq) + NO 3 - (aq) DH 0 = 25 k. J

Entropy n To predict spontaneity we need: n n Change in enthalpy Entropy- a measure of the randomness or disorder of a system. ↑ Disorder = ↑ Entropy

Entropy n n n New Deck Order Shuffled Deck Order Probability Ordered state Disordered State

Entropy and Disorder If the change from initial to final results in an increase in randomness Sf > Si DS > 0 For any substance, the solid state is more ordered than the liquid state and the liquid state is more ordered than gas state Ssolid < Sliquid << Sgas
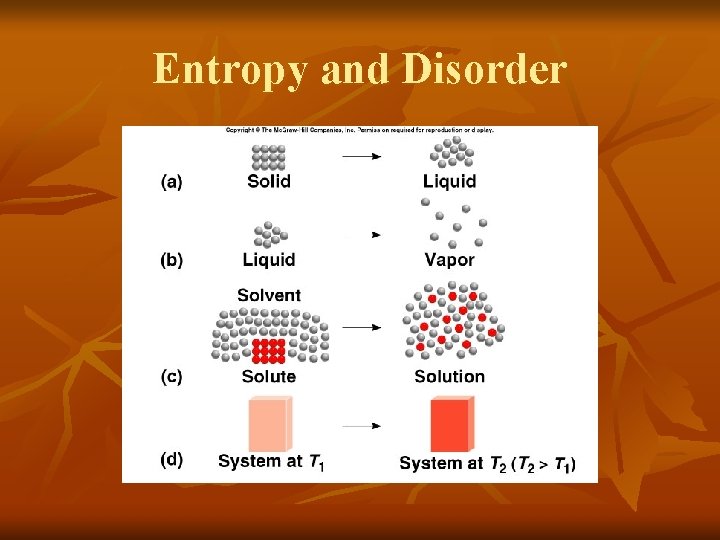
Entropy and Disorder

Entropy and Disorder How does the entropy of a system change for each of the following processes? (a) Forming sucrose crystals from a supersaturated solution Randomness decreases Entropy decreases (DS < 0) (b) Heating hydrogen gas from 600 C to 800 C Randomness increases Entropy increases (DS > 0)

Entropy and Disorder

Standard Entropy The standard entropy of reaction (DS 0 rxn) is the entropy change for a reaction carried out at 1 atm and 250 C.

The Second Law of Thermodynamics n The entropy of the universe increases in a spontaneous process and remains unchanged in an equilibrium process. n Importance? Spontaneous process: DSuniv = DSsys + DSsurr > 0 Equilibrium process: DSuniv = DSsys + DSsurr = 0

Entropy Changes in the System n n To calculate ΔSuniv, we need both ΔSsys ΔSsurr ΔSsys a. A + b. B c. C + d. D DS 0 rxn = S n. S 0(products) - S m. S 0(reactants)

Entropy Changes in the System

Entropy Changes in the System When gases are produced (or consumed) • If a reaction produces more gas molecules than it consumes, DS 0 > 0. • If the total number of gas molecules diminishes, DS 0 < 0. • If there is no net change in the total number of gas molecules, then DS 0 may be positive or negative BUT DS 0 will be a small number.

Entropy Changes in the System
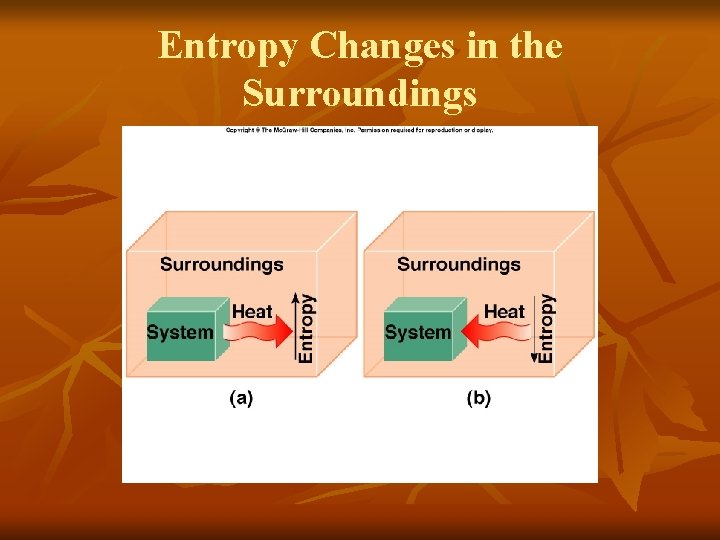
Entropy Changes in the Surroundings

Entropy Changes in the Surroundings ΔSsurr = -ΔHsys T Using the information from Example 18. 2, determine whether or not the reaction is spontaneous. N 2(g) + 3 H 2(g) → 2 NH 3(g) ΔHºrxn = -92. 6 k. J/mol ΔSsys = -199 J/K ∙ mol ΔSsurr = -(-92. 6 x 1000) J/mol 298 K ΔSsurr = 311 J/mol ΔSuniv = ΔSsys + ΔSsurr ΔSuniv = -199 J/K ∙ mol + 311 J/mol ΔSUNIV = 112 J/K ∙ mol
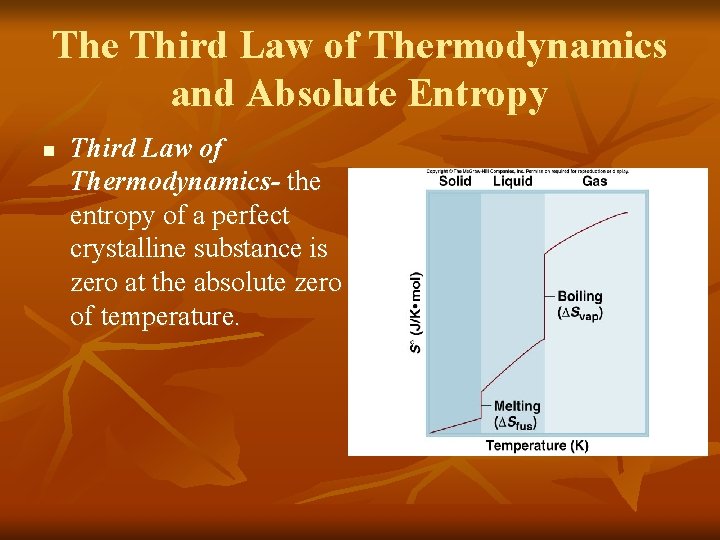
The Third Law of Thermodynamics and Absolute Entropy n Third Law of Thermodynamics- the entropy of a perfect crystalline substance is zero at the absolute zero of temperature.

Gibbs Free Energy n n n Predicts the direction of a spontaneous reaction. Uses properties of the system to calculate. For a constant pressure-temperature process: DG = DHsys -TDSsys DG < 0 DG > 0 DG = 0 The reaction is spontaneous in the forward direction. The reaction is nonspontaneous as written. The reaction is spontaneous in the reverse direction. The reaction is at equilibrium.

Standard Free-Energy Changes The standard free-energy of reaction (DG 0 rxn) is the freeenergy change for a reaction when it occurs under standardstate conditions. Standard free energy of formation (DG 0 f ) is the free-energy change that occurs when 1 mole of the compound is formed from its elements in their standard states. 0 DGrxn = S n. DG 0 f (products) - S m. DG 0 f (reactants)

Standard Free-Energy Changes

Factors Affecting ΔG

Free Energy and Chemical Equilibrium DG = DG 0 + RT ln. Q R is the gas constant (8. 314 J/K • mol) T is the absolute temperature (K) Q is the reaction quotient At Equilibrium DG = 0 Q=K 0 = DG 0 + RT ln. K DG 0 = - RT ln. K

Free Energy and Chemical Equilibrium

Free Energy and Chemical Equilibrium

Free Energy and Chemical Equilibrium
Thermodynamics of a Rubber Band
- Slides: 30