Chemical Symbols Sun Moon Mars Venus Saturn Jupiter
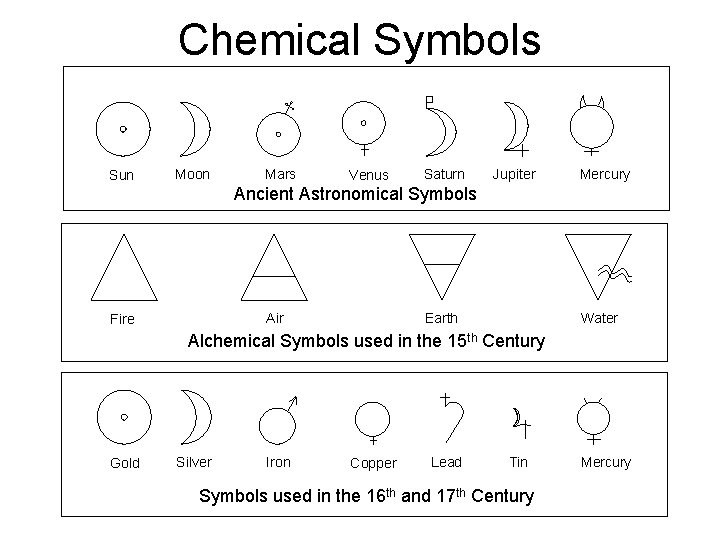
Chemical Symbols Sun Moon Mars Venus Saturn Jupiter Mercury Ancient Astronomical Symbols Air Fire Earth Water Alchemical Symbols used in the 15 th Century Gold Silver Iron Copper Lead Tin Symbols used in the 16 th and 17 th Century Mercury
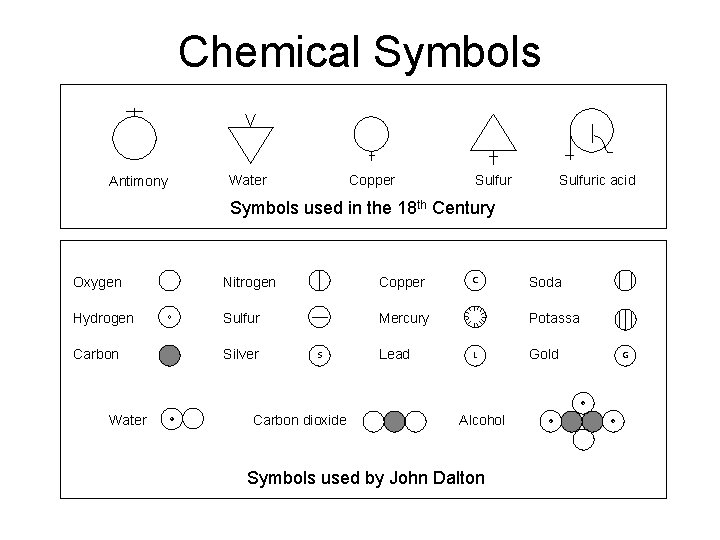
Chemical Symbols Antimony Water Copper Sulfuric acid Symbols used in the 18 th Century Oxygen Nitrogen Copper Hydrogen Sulfur Mercury Carbon Silver Water S Carbon dioxide Lead C Soda Potassa L Alcohol Symbols used by John Dalton Gold G

Origin of the Names of Elements Title Pre-chemical Names from celestial bodies Names from mythology / superstition Names from minerals / ores, other than geographical names Names from colors Names from properties other than color Geographical names from the domicile or workplace of the discoverer(s) Geographical names from minerals / ores Constructed names Names from persons Number of Elements 10 8 10 13 9 8 13 10 16 10
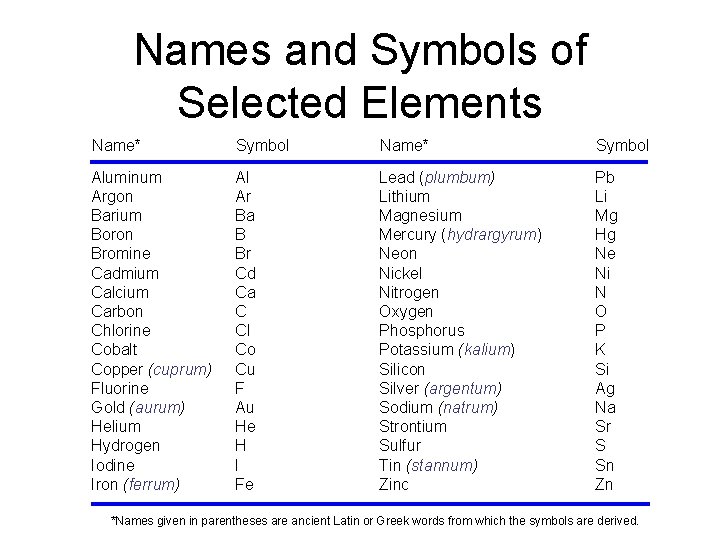
Names and Symbols of Selected Elements Name* Symbol Aluminum Argon Barium Boron Bromine Cadmium Calcium Carbon Chlorine Cobalt Copper (cuprum) Fluorine Gold (aurum) Helium Hydrogen Iodine Iron (ferrum) Al Ar Ba B Br Cd Ca C Cl Co Cu F Au He H I Fe Lead (plumbum) Lithium Magnesium Mercury (hydrargyrum) Neon Nickel Nitrogen Oxygen Phosphorus Potassium (kalium) Silicon Silver (argentum) Sodium (natrum) Strontium Sulfur Tin (stannum) Zinc Pb Li Mg Hg Ne Ni N O P K Si Ag Na Sr S Sn Zn *Names given in parentheses are ancient Latin or Greek words from which the symbols are derived.
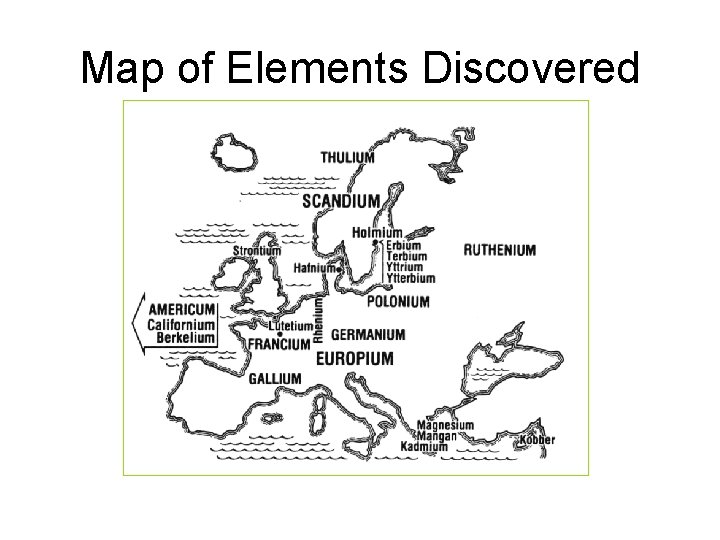
Map of Elements Discovered
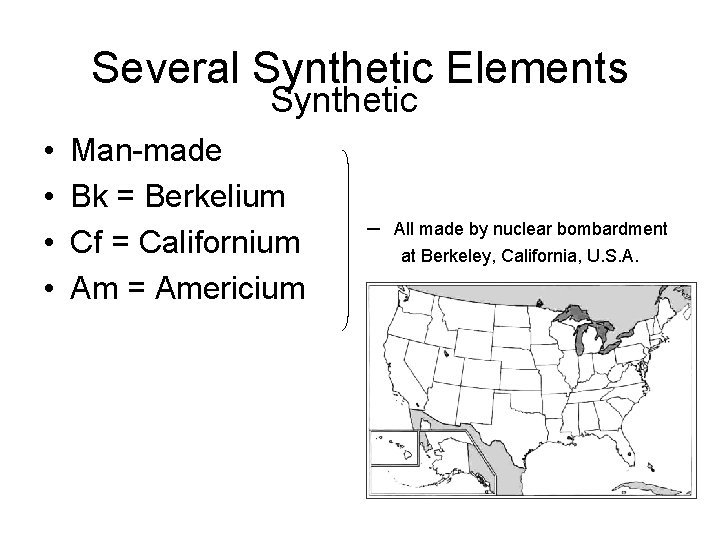
Several Synthetic Elements Synthetic • • Man-made Bk = Berkelium Cf = Californium Am = Americium – All made by nuclear bombardment at Berkeley, California, U. S. A.

How to Organize Elements… Periodic Table Designs

Dutch Periodic Table 115 114 113 112 111 110 109 108 107 106 117 118
“Mayan” Periodic Table
Stowe’s Periodic Table

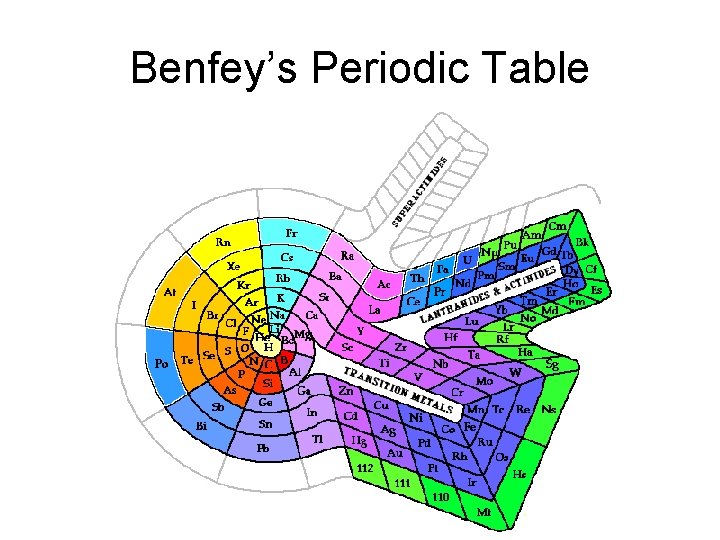
Benfey’s Periodic Table
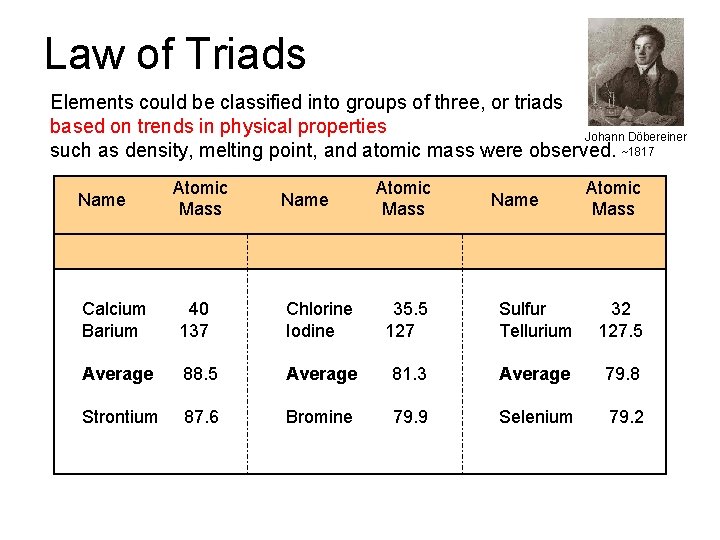
Law of Triads Elements could be classified into groups of three, or triads based on trends in physical properties Johann Döbereiner such as density, melting point, and atomic mass were observed. ~1817 Name Atomic Mass Calcium Barium 40 137 Chlorine Iodine 35. 5 127 Sulfur Tellurium 32 127. 5 Average 88. 5 Average 81. 3 Average 79. 8 Strontium 87. 6 Bromine 79. 9 Selenium 79. 2

Newlands Law of Octaves Arranged the 62 known elements into groups of seven according to increasing atomic mass and proposed that an eighth column would then repeat the properties of the first element in the previous group. Newlands Law of Octaves 1 2 3 4 5 6 7 Li Na K Be Mg B Al C Si N P O S F Cl John Newlands ~1863

Dmitri Mendeleev • Russian • Invented periodic table • Organized elements by properties • Arranged elements by atomic mass • Predicted existence of several unknown elements • Noted some discrepancies with atomic mass Dmitri Mendeleev
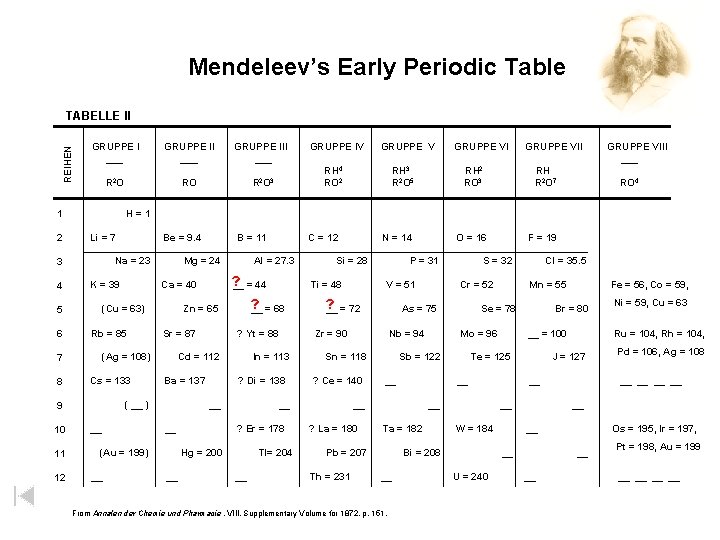
Mendeleev’s Early Periodic Table REIHEN TABELLE II GRUPPE I ___ Li = 7 K = 39 11 12 RH 3 R 2 O 5 Cs = 133 Sr = 87 GRUPPE VI RH 2 RO 3 In = 113 ? Di = 138 __ __ (Au = 199) __ ? Yt = 88 Ba = 137 __ Si = 28 GRUPPE VII RH R 2 O 7 ? Er = 178 Tl= 204 __ V = 51 GRUPPE VIII ___ RO 4 Cr = 52 Nb = 94 Sn = 118 ? Ce = 140 From Annalen der Chemie und Pharmacie , VIII, Supplementary Volume for 1872, p. 151. __ W = 184 Pd = 106, Ag = 108 __ __ U = 240 Ni = 59, Cu = 63 Ru = 104, Rh = 104, J = 127 __ Bi = 208 __ __ = 100 __ Ta = 182 Fe = 56, Co = 59, Br = 80 Te = 125 __ Pb = 207 Mn = 55 Mo = 96 __ Cl = 35. 5 Se = 78 Sb = 122 __ ? La = 180 F = 19 S = 32 As = 75 Zr = 90 Th = 231 O = 16 P = 31 ? __ = 72 __ Hg = 200 N = 14 Ti = 48 ? __ = 68 Cd = 112 ( __ ) __ Al = 27. 3 Zn = 65 (Ag = 108) C = 12 ? __ = 44 Ca = 40 Rb = 85 9 10 GRUPPE V RH 4 RO 2 B = 11 Mg = 24 (Cu = 63) 7 8 Be = 9. 4 Na = 23 5 6 RO R 2 O 3 GRUPPE IV H=1 3 4 GRUPPE III ___ R 2 O 1 2 GRUPPE II ___ Os = 195, Ir = 197, __ __ Pt = 198, Au = 199 __ __
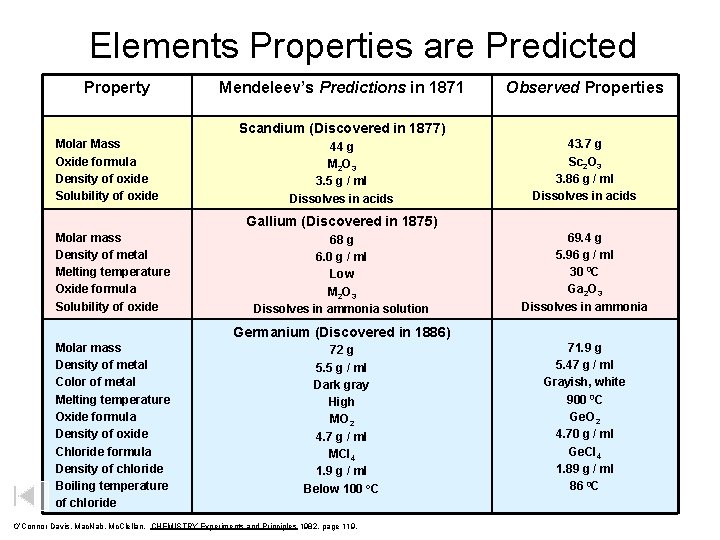
Elements Properties are Predicted Property Mendeleev’s Predictions in 1871 Observed Properties Scandium (Discovered in 1877) Molar Mass Oxide formula Density of oxide Solubility of oxide 44 g M 2 O 3 3. 5 g / ml Dissolves in acids 43. 7 g Sc 2 O 3 3. 86 g / ml Dissolves in acids Gallium (Discovered in 1875) Molar mass Density of metal Melting temperature Oxide formula Solubility of oxide 68 g 6. 0 g / ml Low M 2 O 3 Dissolves in ammonia solution 69. 4 g 5. 96 g / ml 30 0 C Ga 2 O 3 Dissolves in ammonia Germanium (Discovered in 1886) Molar mass Density of metal Color of metal Melting temperature Oxide formula Density of oxide Chloride formula Density of chloride Boiling temperature of chloride 72 g 5. 5 g / ml Dark gray High MO 2 4. 7 g / ml MCl 4 1. 9 g / ml Below 100 o. C O’Connor Davis, Mac. Nab, Mc. Clellan, CHEMISTRY Experiments and Principles 1982, page 119, 71. 9 g 5. 47 g / ml Grayish, white 900 0 C Ge. O 2 4. 70 g / ml Ge. Cl 4 1. 89 g / ml 86 0 C

Modern Periodic Table • Determined the atomic numbers of elements from their Xray spectra (1914) • Resolved discrepancies in Mendeleev’s arrangement. • Arranged elements by increasing atomic number Henry G. J. Moseley 1887 - 1915
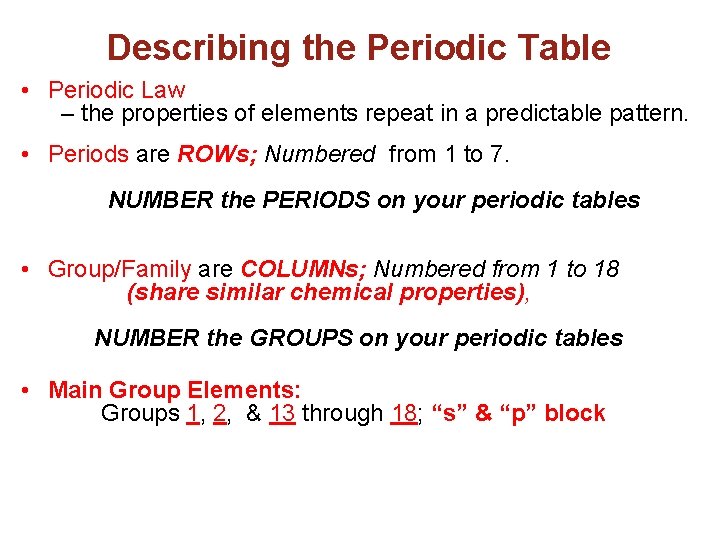
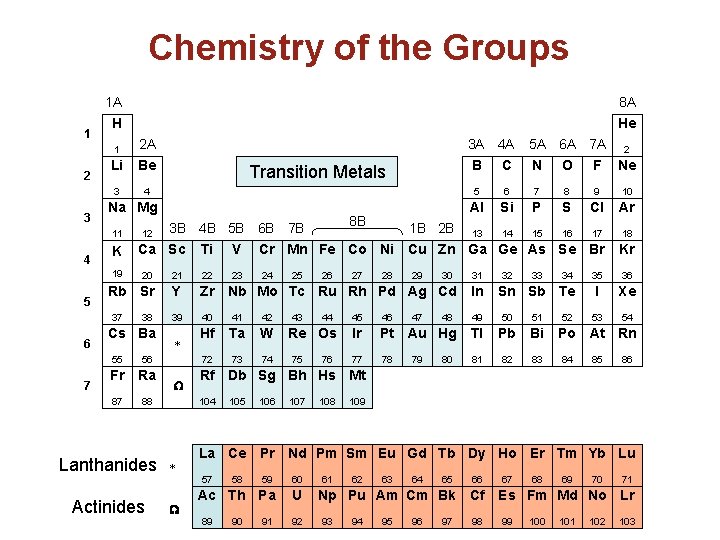
Describing the Periodic Table • Periodic Law – the properties of elements repeat in a predictable pattern. • Periods are ROWs; Numbered from 1 to 7. NUMBER the PERIODS on your periodic tables • Group/Family are COLUMNs; Numbered from 1 to 18 (share similar chemical properties), NUMBER the GROUPS on your periodic tables • Main Group Elements: Groups 1, 2, & 13 through 18; “s” & “p” block
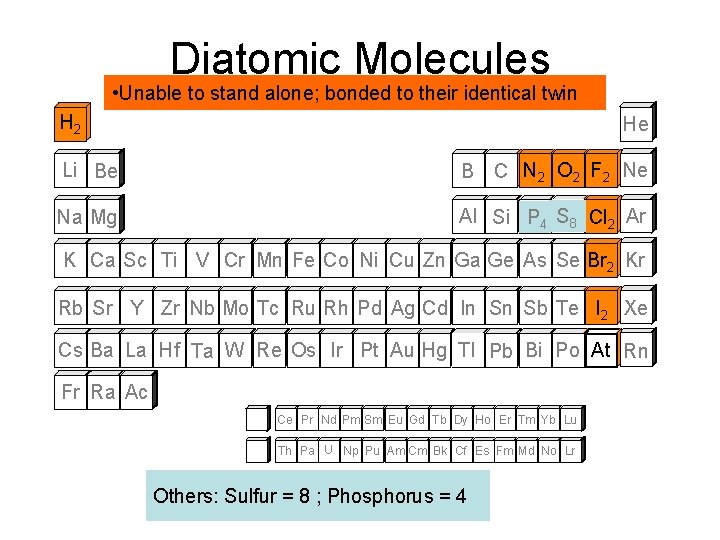
Diatomic Molecules Diatomic Elements • Unable to stand alone; bonded to their identical twin H 2 He Li Be B C N 2 O 2 F 2 Ne Na Mg ` SS Cl Ar Al Si P 8 2 4 K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br 2 Kr Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I 2 Xe Cs Ba La Hf Ta W Re Os Ir Pt Au Hg Tl Pb Bi Po At Rn Fr Ra Ac Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No Lr Others: Sulfur = 8 ; Phosphorus = 4

Stop & Stations
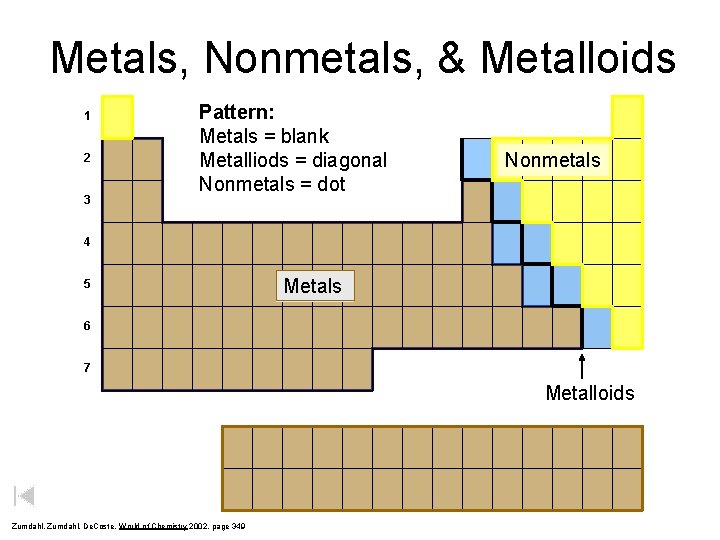
Metals, Nonmetals, & Metalloids 1 2 3 Pattern: Metals = blank Metalliods = diagonal Nonmetals = dot Nonmetals 4 5 Metals 6 7 Metalloids Zumdahl, De. Coste, World of Chemistry 2002, page 349

Properties of Metals • Good conductors of heat and electricity. • Lustrous • Ductile (stretched into thin wires) • Malleable (pounded into thin sheets) • Chemical property: Reaction with water which results in corrosion • Chemical Reactions: lose electrons to form positively charged ions • All are solids except for mercury

Properties of Non-Metals Sulfur • Poor Conductors of heat and electricity. • Dull • Not ductile • Not malleable • Solid non-metals are brittle • Can be gases, liquids or solids. • Tend to gain electrons to form negatively charged ions • Share electrons in reaction with other non-metals.

Properties of Metalloids • Metalloids (metal-like) • Exhibit properties of both metals and non-metals • Solids that can be shiny or dull. • Conduct heat and electricity better than non-metals but not as well as metals. • Ductile and malleable. Silicon
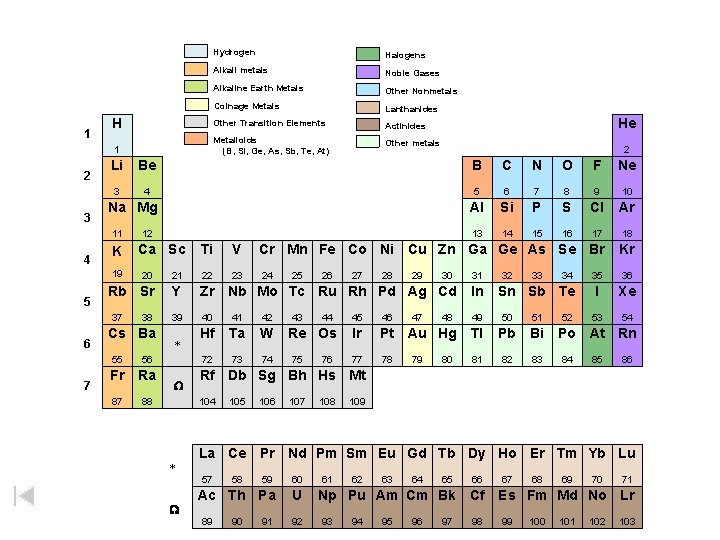
1 2 3 Hydrogen Halogens Alkali metals Noble Gases Alkaline Earth Metals Other Nonmetals Coinage Metals Lanthanides H Other Transition Elements Actinides 1 Metalloids (B, Si, Ge, As, Sb, Te, At) Other metals Be B C N O F Ne 3 4 5 6 7 8 9 10 Al Si P S Cl Ar 13 14 15 16 17 18 Na Mg K 19 5 7 12 Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr 23 24 35 36 I Xe 53 54 20 21 22 Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In 39 40 41 42 49 50 51 Hf Ta W 72 73 74 37 6 2 Li 11 4 He 38 Cs Ba 55 56 Fr Ra 87 88 * W 44 Re Os 75 76 27 28 29 47 30 32 33 46 Ir Pt Au Hg Tl Pb Bi 77 78 81 82 83 80 34 Sn Sb Te 45 79 48 31 52 Po At Rn 84 85 86 105 106 107 108 109 La Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu 59 60 Ac Th Pa U 57 W 43 26 Rf Db Sg Bh Hs Mt 104 * 25 89 58 90 91 92 61 62 63 64 65 66 Np Pu Am Cm Bk Cf 93 94 95 96 97 98 67 68 69 70 71 Es Fm Md No Lr 99 100 101 102 103
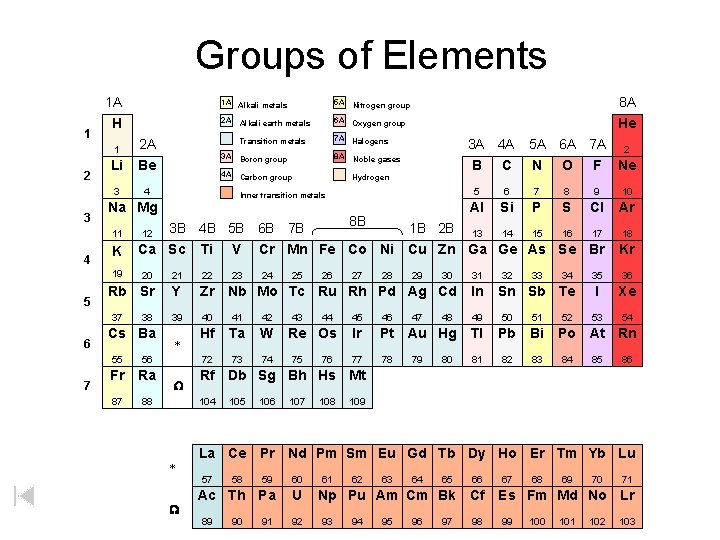
Groups of Elements 1 A 1 2 3 H 1 2 A Li Be 3 4 K 19 5 7 Nitrogen group 2 A Alkali earth metals 6 A Oxygen group Transition metals 7 A Halogens 3 A Boron group 8 A Noble gases 4 A Carbon group 8 A He 3 A 4 A B C Hydrogen Inner transition metals 8 B 5 A 6 A 7 A 2 N O F Ne 5 6 7 8 9 10 Al Si P S Cl Ar 3 B 4 B 5 B 6 B 7 B 1 B 2 B 13 14 15 16 17 Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br 12 20 21 22 Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In 39 40 41 42 49 50 51 Hf Ta W 72 73 74 37 6 5 A Na Mg 11 4 1 A Alkali metals 38 Cs Ba 55 56 Fr Ra 87 88 * W 25 43 26 44 Re Os 75 76 27 28 29 47 30 32 33 46 Ir Pt Au Hg Tl Pb Bi 77 78 81 82 83 80 34 Sn Sb Te 45 79 48 31 52 35 36 I Xe 53 54 Po At Rn 84 85 86 105 106 107 108 109 La Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu 57 W 24 Kr Rf Db Sg Bh Hs Mt 104 * 23 18 59 60 Ac Th Pa U 89 58 90 91 92 61 62 63 64 65 66 Np Pu Am Cm Bk Cf 93 94 95 96 97 98 67 68 69 70 71 Es Fm Md No Lr 99 100 101 102 103
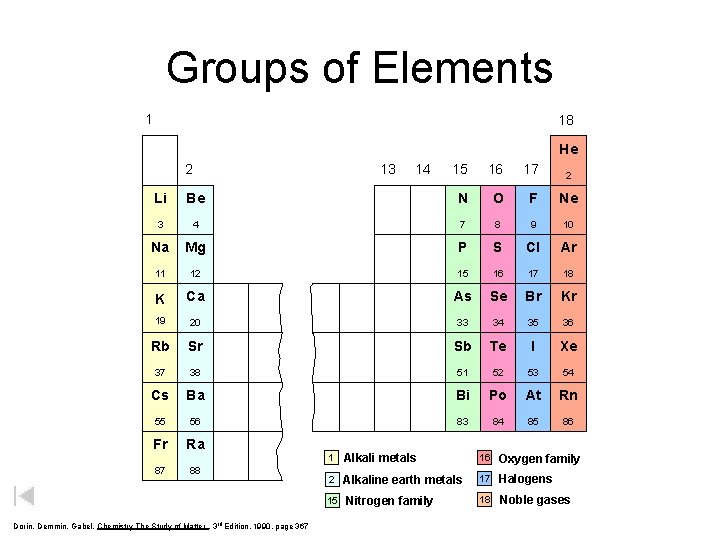
Groups of Elements 1 18 He 2 13 14 15 16 17 2 Li Be N O F Ne 3 4 7 8 9 10 Na Mg P S Cl Ar 11 12 15 16 17 18 K Ca As Se Br Kr 19 20 33 34 35 36 Rb Sr Sb Te I Xe 37 38 51 52 53 54 Cs Ba Bi Po At Rn 55 56 83 84 85 86 Fr Ra 87 88 1 Alkali metals 16 Oxygen family 2 Alkaline earth metals 17 Halogens 18 Noble gases 15 Dorin, Demmin, Gabel, Chemistry The Study of Matter , 3 rd Edition, 1990, page 367 Nitrogen family

Hydrogen • is in a class of its own. • only needs 2 electrons to complete its valence shell. • Characteristics Gas at room temperature. Flammable
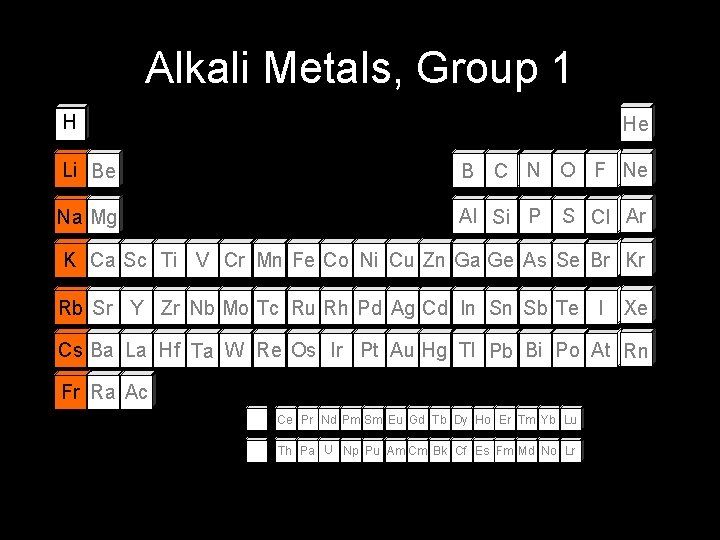
Alkali Metals, Group 1 H He Li Be B C N O F Ne Na Mg Al Si P S Cl Ar K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I Xe Cs Ba La Hf Ta W Re Os Ir Pt Au Hg Tl Pb Bi Po At Rn Fr Ra Ac Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No Lr

Alkali Metals • Most REACTIVE metals. • Characteristics Soft (Clay consistency) Dull Silver color Low Density Low Melting Point React violently with water React w/ halogens to form salt • ns 1 valence electron configuration
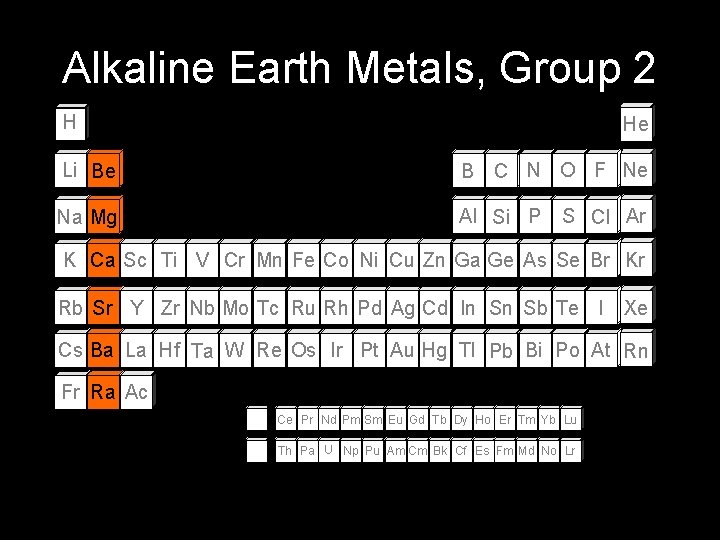
Alkaline Earth Metals, Group 2 H He Li Be B C N O F Ne Na Mg Al Si P S Cl Ar K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I Xe Cs Ba La Hf Ta W Re Os Ir Pt Au Hg Tl Pb Bi Po At Rn Fr Ra Ac Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No Lr

Alkaline Earth Metals • Characteristics Soft Shiny silver color Low Density Found naturally as a compound Glow different colors • “ns 2” valence electron configuration
Transition Metals • Column 11 is known as “coinage metals” contains copper, silver & gold.

Transition Metals • usually brightly colored • often used to color paints. • Characteristics High Density High Melting/Boiling Pt Good Catalysts Hard, Tough & Strong • Multiple oxidation (charge) states • Form compounds called oxides

Group 13: Boron Family • 3 valence electrons forms +3 charged ions. • Family includes a metalloid (boron), and the rest are metals. • Family includes the most abundant metal in the earth’s crust; aluminum. • Highly reactive & form stable compounds with oxygen • “ns 2 np 1” valence electron configuration

Group 14: Carbon Family • 4 valence electrons forms 3 oxidation states -4, +4 & +2 (loss of two np 2 gives a filled ns 2 subshell) • Family includes a non-metal, metalloids, and metals. • Carbon is called the “basis of life. ” • “ns 2 np 2” valence-electron configuration.
Group 15: Pnicogens H He Li Be B C N O F Ne Na Mg Al Si P S Cl Ar K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I Xe Cs Ba La Hf Ta W Re Os Ir Pt Au Hg Tl Pb Bi Po At Rn Fr Ra Ac Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No Lr

Group 15: Nitrogen Family • 5 valence electrons forms 3 oxidation states -3, +5 & +3 (three np electrons lost to give a filled ns 2 subshell) • Family includes non-metals, metalloids and metals • Nitrogen makes up 79% of our atmosphere. • “ns 2 np 3” valence electron configuration
Group 16: Chalcogens H He Li Be B C N O F Ne Na Mg Al Si P S Cl Ar K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I Xe Cs Ba La Hf Ta W Re Os Ir Pt Au Hg Tl Pb Bi Po At Rn Fr Ra Ac Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No Lr

Group 16: Oxygen Family O 8 S 16 Se 34 Te 52 Po • 6 valence electrons forms 3 oxidation states -2, +6 & +4 (four np electrons lost give filled ns 2 subshell) 84 • Oxygen is the most abundant element in the earth’s crust. It is extremely active and combines with almost all elements. • “ns 2 np 4” valence-electron configuration
Halogens, Group 17 H He Li Be B C N O F Ne Na Mg Al Si P S Cl Ar K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I Xe Cs Ba La Hf Ta W Re Os Ir Pt Au Hg Tl Pb Bi Po At Rn Fr Ra Ac Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No Lr

Group 17: Halogen (Salt Makers) Family • Most active non-metals. - never found free in nature • Only group in all 3 states of matter • React with alkali metals to form SALTS • High electronegativity • Non-metal qualities – dull, brittle, nonconductor • “ns 2 np 5” valence electron configuration
Noble Gases, Group 18 H He Li Be B C N O F Ne Na Mg Al Si P S Cl Ar K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I Xe Cs Ba La Hf Ta W Re Os Ir Pt Au Hg Tl Pb Bi Po At Rn Fr Ra Ac Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No Lr

Group 18: Noble Gases • Characteristics Inert (Non-reactive) due to their filled octet Colorless gas Odorless Tasteless Nonflammable Non-metal qualities – dull, brittle, non-conductor • Found in small amounts in the earth's atmosphere. • “ns 2 np 6” valence electron configuration
Lanthanide Series H He Li Be B C N O F Ne Na Mg Al Si P S Cl Ar K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I Xe Cs Ba La Hf Ta W Re Os Ir Pt Au Hg Tl Pb Bi Po At Rn Fr Ra Ac La Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No Lr
Actinide Series H He Li Be B C N O F Ne Na Mg Al Si P S Cl Ar K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I Xe Cs Ba La Hf Ta W Re Os Ir Pt Au Hg Tl Pb Bi Po At Rn Fr Ra Ac La Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu La Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No Lr
Rare Earth (Inner Transition) Elements • Rare earth elements not particularly rare but long & tedious processes were required to purify the metals from their oxides prior to 1945 • One element of the lanthanide series & most of the elements in the actinide series are called transuranium, which means synthetic or man-made • Characteristics High melting points Reactivity varies Usually bonded to non-metals Tarnish readily in air High electrical conductivity .
Chemistry of the Groups 1 A 1 2 3 H 1 2 A Li Be 3 4 K 19 5 7 8 B 5 A 6 A 7 A 2 N O F Ne 5 6 7 8 9 10 Al Si P S Cl Ar 3 B 4 B 5 B 6 B 7 B 1 B 2 B 13 14 15 16 17 Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br 12 20 21 22 Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In 39 40 41 42 49 50 51 Hf Ta W 72 73 74 37 6 3 A 4 A B C Transition Metals Na Mg 11 4 8 A He 38 Cs Ba 55 56 Fr Ra 87 88 * W * Actinides W 24 25 43 26 44 Re Os 75 76 27 28 29 47 30 32 33 46 Ir Pt Au Hg Tl Pb Bi 77 78 81 82 83 80 34 Sn Sb Te 45 79 48 31 52 Kr 35 36 I Xe 53 54 Po At Rn 84 85 86 Rf Db Sg Bh Hs Mt 104 Lanthanides 23 18 105 106 107 108 109 La Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu 57 59 60 Ac Th Pa U 89 58 90 91 92 61 62 63 64 65 66 Np Pu Am Cm Bk Cf 93 94 95 96 97 98 67 68 69 70 71 Es Fm Md No Lr 99 100 101 102 103
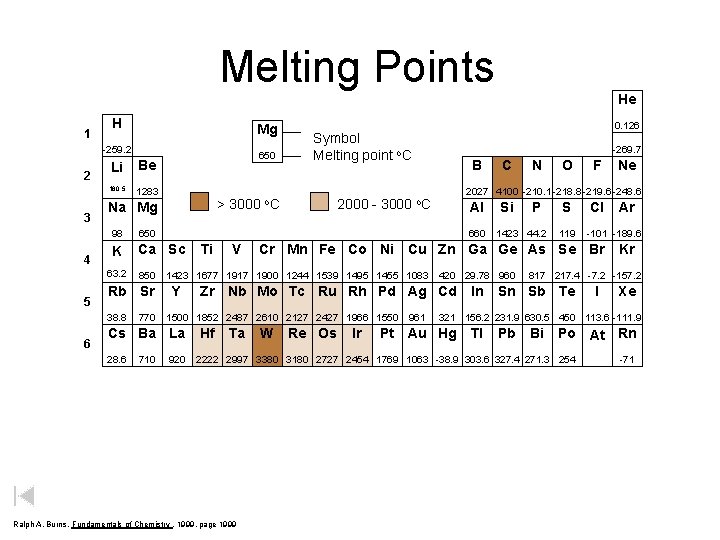
Melting Points 1 H Mg -259. 2 2 3 4 Li Be 180. 5 1283 98 650 K Ca Sc 850 Rb Sr 38. 8 6 > 3000 Na Mg 63. 2 5 650 770 710 2000 - 3000 -269. 7 B Ti V Al 1500 1852 2487 2610 2127 2427 1966 1550 920 Ta Si P 1423 44. 2 420 29. 78 960 Zr Nb Mo Tc Ru Rh Pd Ag Cd Hf N O S 119 W Re Os Ir 961 Ne Cl In Ar -101 -189. 6 Kr 817 217. 4 -7. 2 -157. 2 Sn Sb Te I Xe 321 156. 2 231. 9 630. 5 450 113. 6 -111. 9 Pt Au Hg Tl Pb Bi Po At Rn 2222 2997 3380 3180 2727 2454 1769 1063 -38. 9 303. 6 327. 4 271. 3 254 Ralph A. Burns, Fundamentals of Chemistry , 1999, page 1999 F Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br 1423 1677 1917 1900 1244 1539 1495 1455 1083 Y C 2027 4100 -210. 1 -218. 8 -219. 6 -248. 6 o. C 660 Cs Ba La 28. 6 He 0. 126 Symbol Melting point o. C He -71
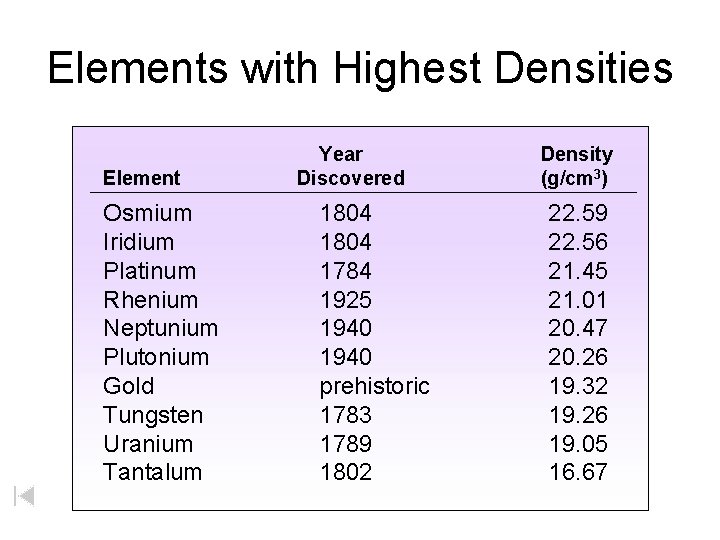
Elements with Highest Densities Element Osmium Iridium Platinum Rhenium Neptunium Plutonium Gold Tungsten Uranium Tantalum Year Discovered 1804 1784 1925 1940 prehistoric 1783 1789 1802 Density (g/cm 3) 22. 59 22. 56 21. 45 21. 01 20. 47 20. 26 19. 32 19. 26 19. 05 16. 67
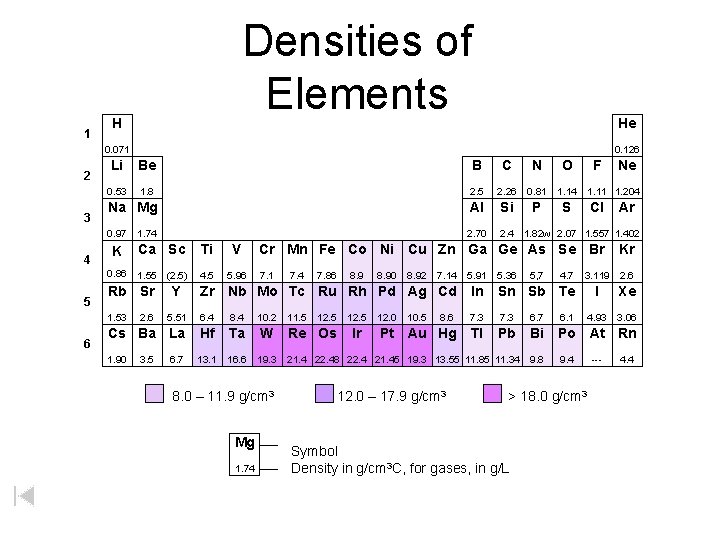
1 Densities of Elements H He 0. 071 2 3 4 5 Li Be B C N O 0. 53 1. 8 2. 5 2. 26 0. 81 1. 14 Na Mg Al Si P S 0. 97 2. 70 2. 4 1. 82 w 2. 07 1. 557 1. 402 1. 74 K Ca Sc Ti V 0. 86 1. 55 (2. 5) 4. 5 5. 96 Rb Sr Ne 1. 11 1. 204 Cl Ar Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr 7. 1 3. 119 2. 6 I Xe 4. 93 3. 06 7. 4 7. 86 8. 90 8. 92 7. 14 5. 91 5. 36 5, 7 4. 7 In Sn Sb Te 5. 51 6. 4 8. 4 10. 2 8. 6 7. 3 6. 7 6. 1 Cs Ba La Hf Ta W Pt Au Hg Tl Pb Bi Po At Rn 1. 90 13. 1 16. 6 19. 3 9. 8 9. 4 2. 6 3. 5 Y F Zr Nb Mo Tc Ru Rh Pd Ag Cd 1. 53 6 0. 126 6. 7 8. 0 – 11. 9 g/cm 3 Mg 1. 74 W 11. 5 12. 5 Re Os 12. 5 Ir 12. 0 10. 5 21. 4 22. 48 22. 4 21. 45 19. 3 13. 55 11. 85 11. 34 12. 0 – 17. 9 g/cm 3 > 18. 0 g/cm 3 Symbol Density in g/cm 3 C, for gases, in g/L --- 4. 4
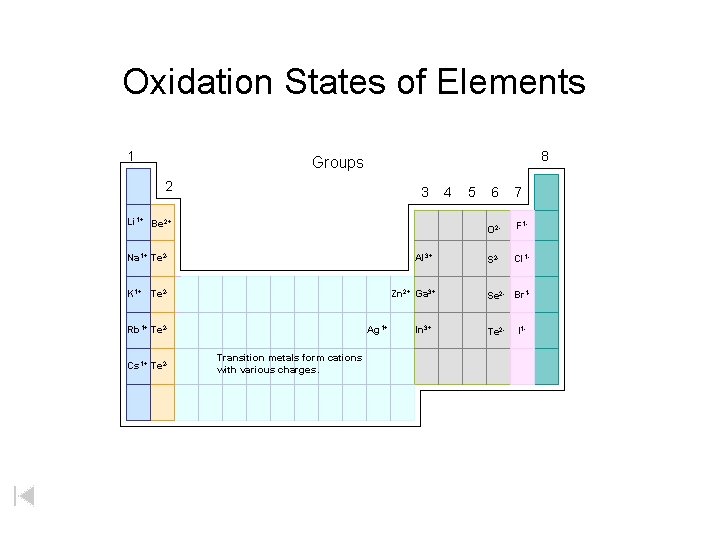
Oxidation States of Elements 1 8 Groups 2 3 Li 1+ Be 2+ Na 1+ Te 2 - Al 3+ K 1+ Te 2 - Zn 2+ Ga 3+ Rb 1+ Te 2 Cs 1+ Te 2 - Ag 1+ Transition metals form cations with various charges. In 3+ 4 5 6 7 O 2 - F 1 - S 2 - Cl 1 - Se 2 - Br 1 Te 2 - I 1 -
- Slides: 54