CHEMICAL REACTIONS TUTORIAL Lets get started Goals In












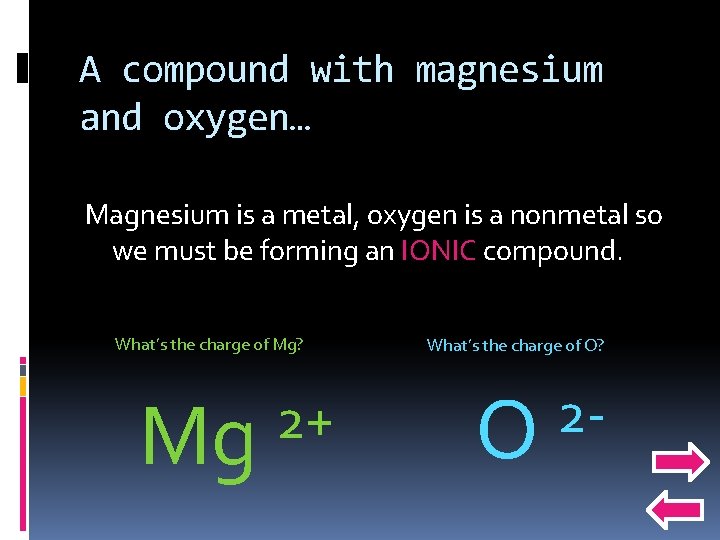








































- Slides: 56

CHEMICAL REACTIONS TUTORIAL Let’s get started!!

Goals In this tutorial, you will learn… How to recognize 3 of the 5 types of reactions Predict the products of reactions when only given the reactants

What are the 5 types of reactions? Synthesis Double replacement Single replacement Decomposition Combustion

Which 3 will be we learning about? ? Synthesis Decomposition Combustion

SYNTHESIS

Synthesis Two or more reactants combine to form… ONE PRODUCT!! A + B AB

Synthesis Continued 2 Mg + O 2 2 Mg. O Two reactants form one product BUT WAIT!! Don’t forget about everything you have already learned! Is your equation balanced? ?

Predicting products Before we keep going… lets learn a few things about predicting products

Predicting products Here a few important things to remember when predicting products: The compounds form must be neutral ionic compounds (which means you’ll be paying attention to their charges) You do NOT carry subscripts from the reactants to the products. You always balance your equation LAST

Let’s go back to that synthesis example Remember the reaction: Mg + O 2 Helpful hint for later…why is oxygen O 2 and not just O? Click to find out why

Why O 2? ? Oxygen is a diatomic molecule, which means when oxygen is by itself it must be O 2. Remember that this DOES NOT mean that oxygen is O 2 in compounds, only by itself! What are the other diatomic elements? (hint: remember the heavenly 7! H 2 I 2 Br 2 O 2 N 2 F 2 Cl 2 Go back

Now to predict the products… Mg + O 2 Remember A + B AB so Mg + O 2 will make a compound with magnesium and oxygen. What does a compound with magnesium and oxygen look like? ?
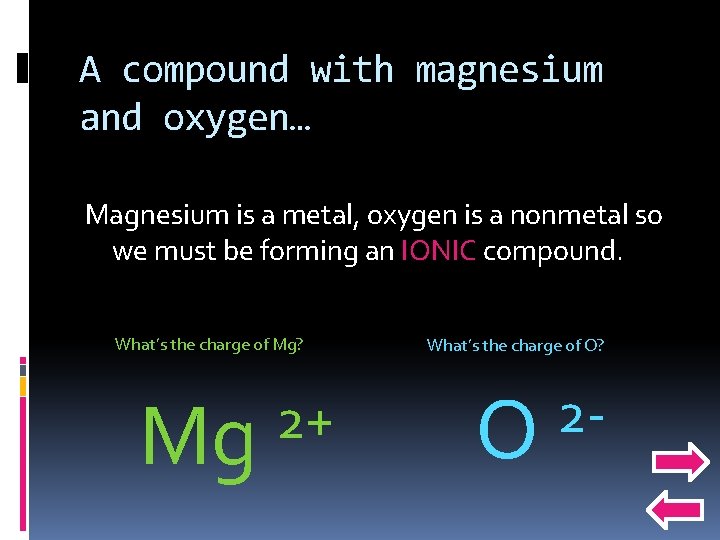
A compound with magnesium and oxygen… Magnesium is a metal, oxygen is a nonmetal so we must be forming an IONIC compound. What’s the charge of Mg? Mg 2+ What’s the charge of O? O 2 -

So what’s the compound? Pick… 2+ Mg Mg. O 2 2 O Mg 2 O 2

Nope! Mg. O 2 +2 + 2(-2) DOES NOT EQUAL ZERO Try again!

Nope! Mg 2 O 2 2(+2) + 2(-2) =0 But remember ionic compounds need to be in the simplest form Try again!

Yes! Very good! Mg. O is a neutral, correct ionic compound so our formula is Mg + O 2 Mg. O Finally, balance the equation to satisfy the law of conservation of mass… 2 Mg + O 2 2 Mg. O

So remember… When predicting products: Make correct ionic compounds THEN balance Let’s move on to the rest of the reaction types…

Decomposition

Decomposition ONE REACTANT… forms two or more products AB A + B

Decomposition Continued The electrolysis of water is when water decomposes into hydrogen and oxygen gas as shown below: 2 H 2 O 2 H 2 + O 2

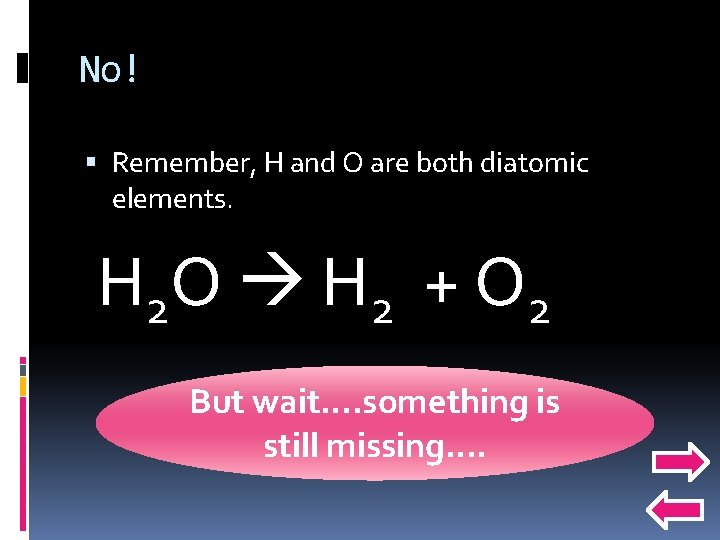
Predicting products and decomposition Lets use that same reaction. What if we were not already given the products, how would we predict them? H 2 O ? ? Water is made of hydrogen and oxygen, so will my products just be H and O?

No! Remember, H and O are both diatomic elements. H 2 O H 2 + O 2 But wait…. something is still missing….

Balance! Don’t forget to balance your equation after you make your products! Your final answer should look like this: 2 H 2 O 2 H 2 + O 2

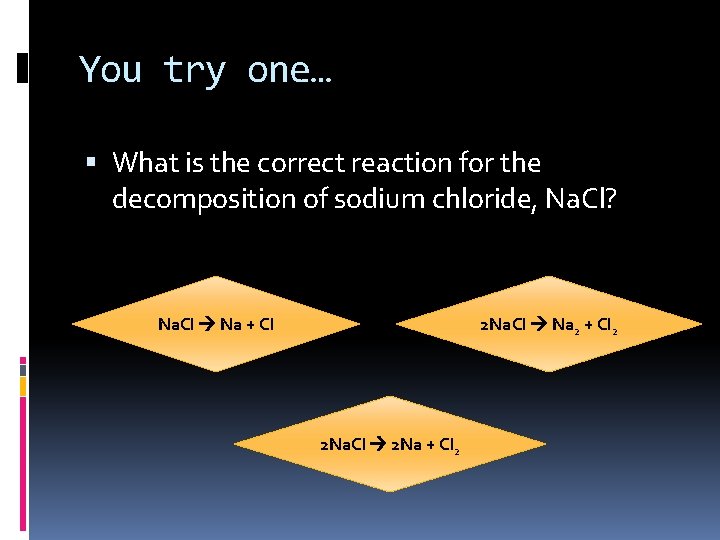
You try one… What is the correct reaction for the decomposition of sodium chloride, Na. Cl? Na. Cl Na + Cl 2 Na. Cl Na 2 + Cl 2 2 Na. Cl 2 Na + Cl 2

Nope! Na. Cl Na + Cl Don’t forget that chlorine is a diatomic element, so it must be Cl 2 Try again!

Nope! 2 Na. Cl Na 2 + Cl 2 Although this reaction is correctly balanced, sodium is NOT a diatomic element Try again!

Yes! Very good! 2 Na. Cl 2 Na + Cl 2 This is a correct reaction for the decomposition of sodium chloride. Ready for the last type of reaction?

Combustion

Combustion is the burning of hydrocarbons. Hydrocarbon? ? What’s that? ? Click to find out! When a hydrocarbon reacts with oxygen (O 2), the products are ALWAYS…. . Yes, ALWAYS… Carbon dioxide and water CO 2 + H 2 O

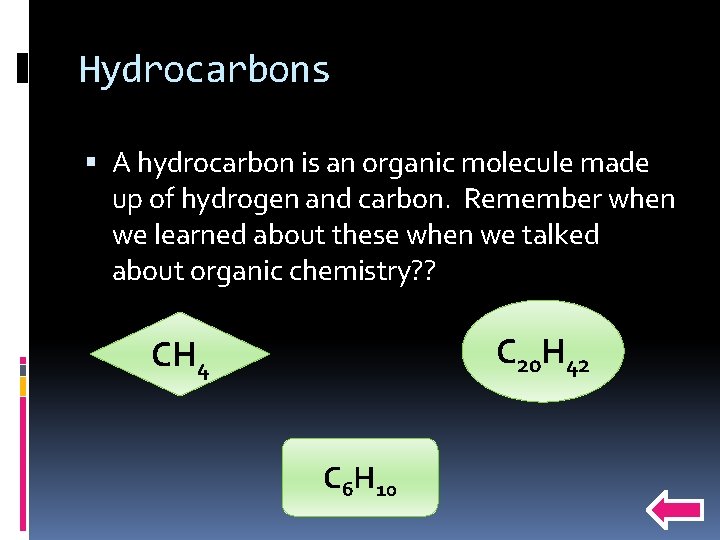
Hydrocarbons A hydrocarbon is an organic molecule made up of hydrogen and carbon. Remember when we learned about these when we talked about organic chemistry? ? C 20 H 42 CH 4 C 6 H 10

Combustion Cont… Since the products of a combustion reaction are always CO 2 and H 2 O, the hardest part about combustion reactions is balancing them CH 4 + O 2 CO 2 + H 2 O Balance this equation…

Combustion Cont. CH 4 + O 2 CO 2 + 2 H 2 O Be aware… this reaction was pretty easy to balance, some of the combustion reactions get pretty complicated! Like this one… 4 C 3 H 9 + 21 O 2 12 CO 2 + 18 H 2 O

Ready for your quiz? ? Yes! Not quite, review the types of reactions with me…

Review Synthesis reactions: two or more reactants combine to form ONE product A + B AB Decomposition reactions: ONE reactant breaks down into two or more products AB A + B Combustion reactions: a hydrocarbon reacts with oxygen to produce carbon dioxide and water. Hydrocarbon + O 2 CO 2 + H 2 O Ok, now I’m ready for the quiz!

Quiz Question 1/5 Which of the following is a decomposition reaction? 2 Fe + O 2 2 Fe. O Na + Ag. Cl Ag + Na. Cl Na 2 S 2 Na + S

Nope! 2 Fe + O 2 2 Fe. O This is two reactants forming ONE product which is an example of a synthesis reaction Try again!

Nope! Na + Ag. Cl Ag + Na. Cl This reaction does not just form one product. This reaction is an example of a single replacement reaction which we will learn about later. Try again!

Yes! Very good! Na 2 S 2 Na + S This reaction is ONE reactant breaking down into two or more products, which is an example of a decomposition reaction. Back to the question! Next question!

Quiz Question 2/5 How can you recognize a combustion reaction? A combustion reaction is the only reaction always has at least two reactants and two products. One of the reactants is always oxygen gas. A combustion reaction always has a hydrocarbon reacting with oxygen to produce carbon dioxide and water. A combustion reaction always has a hydrocarbon reacting with nitrogen, which is a very explosive reaction.

Nope! A combustion reaction is the only reaction always has at least two reactants and two products. One of the reactants is always oxygen gas. We will learn about other reactions that can have more than one product and more than one reactant. This is NOT the way to identify a combustion reaction. Try again!

Nope! A combustion reaction always has a hydrocarbon reacting with nitrogen, which is a very explosive reaction. Although combustion reactions are typically explosive, it is not because they react with nitrogen. Try again!

Yes! Very good! A combustion reaction always has a hydrocarbon reacting with oxygen to produce carbon dioxide and water. This is how we identify a combustion reaction Back to the question! Next question!

Quiz Question 3/5 Pick correct bubble that finishes the reaction below. K + Cl 2 ? ? K + Cl 2 KCl 2 K + Cl 2 2 KCl K + Cl 2 KCl 2

Nope! K + Cl 2 KCl 2 Remember to not carry subscripts over to your products. K has a +1 charge and Cl has a -1 charge so the product of KCl 2 is not neutral and cannot be correct. Try again!

Almost! But not quite… K + Cl 2 KCl Although this product is correct, the equation is not balanced! Try again!

Yes! Very good! 2 K + Cl 2 2 KCl The products are correct and the reaction is balanced! Great job! Back to the question! Next question!

Quiz Question 4/5 Pick correctangle that finishes the reaction below. C 2 H 8 + O 2 ? ? C 2 H 8 + O 2 CO 2 + H 2 O C 2 H 8 + O 2 C 2 H 8 + 4 O 2 2 CO 2 + 4 H 2 O

Almost, but not quite… C 2 H 8 + O 2 CO 2 + H 2 O Great job predicting the products, but…. don’t forget to balance!! Try again!

Nope! C 2 H 8 + O 2 C 2 H 8 O 2 Remember that combustion reactions always have carbon dioxide and water as products Try again!

Yes! Very good! C 2 H 8 + 4 O 2 2 CO 2 + 4 H 2 O Products are correct and the equation is correctly balanced. Great job! Back to the question! Next question!

Quiz Question 5/5 Pick correctangle that finishes the reaction below. Al + N 2 ? ? Al + N 2 Al. N 2 3 Al + N 2 Al 3 N 2 2 Al + N 2 2 Al. N

Nope! Al + N 2 Al. N 2 Remember we do not carry over subscripts. Aluminum has a +3 charge and nitrogen has a -3 charge so a formula of Al. N 2 is not a neutral compound Try again!

Nope! 3 Al + N 2 Al 3 N 2 Although this equation is balanced, since aluminum has a +3 charge and nitrogen has a -3 charge, this is not a neutral compound Try again!

Yes! Very good! 2 Al + N 2 2 Al. N A correct neutral compound for the product and a balanced equation. Great! Back to the question! Finish!

Congratulations! You have finished the chemical reactions tutorial Go back to the start