Chemical Reactions Table of Contents Observing Chemical Change

Chemical Reactions Table of Contents Observing Chemical Change Describing Chemical Reactions Controlling Chemical Reactions Fire and Fire Safety

Chemical Reactions - Observing Chemical Change Properties and Changes in Matter can undergo both physical change and chemical change.
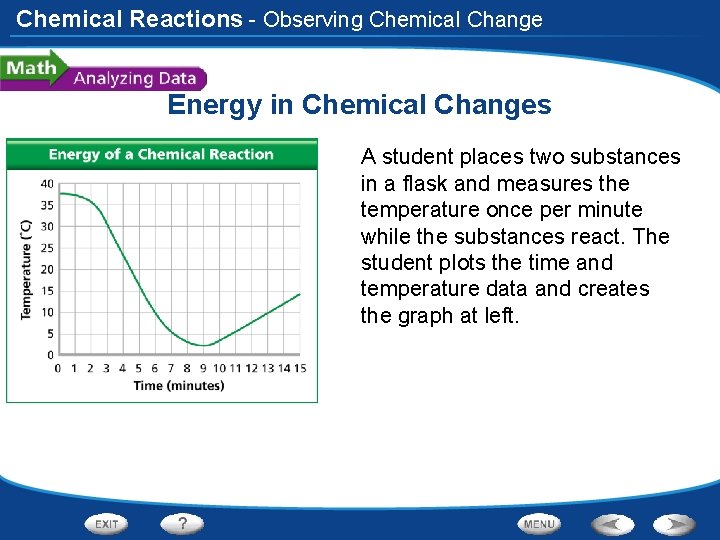
Chemical Reactions - Observing Chemical Change Energy in Chemical Changes A student places two substances in a flask and measures the temperature once per minute while the substances react. The student plots the time and temperature data and creates the graph at left.
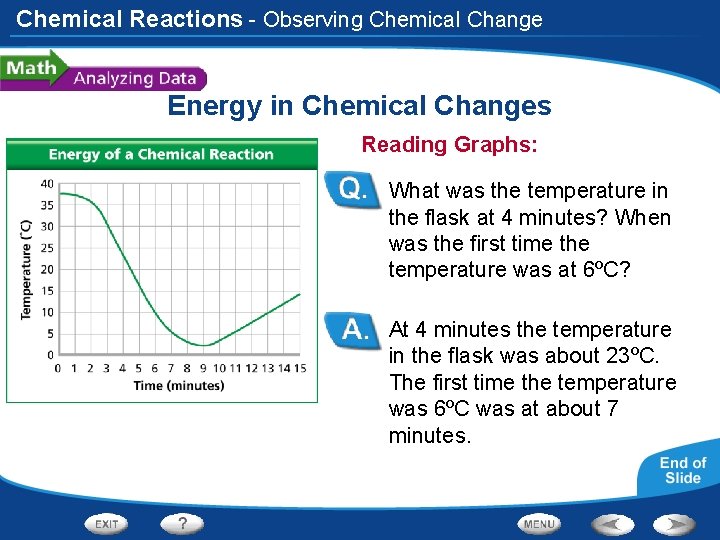
Chemical Reactions - Observing Chemical Change Energy in Chemical Changes Reading Graphs: What was the temperature in the flask at 4 minutes? When was the first time the temperature was at 6ºC? At 4 minutes the temperature in the flask was about 23ºC. The first time the temperature was 6ºC was at about 7 minutes.
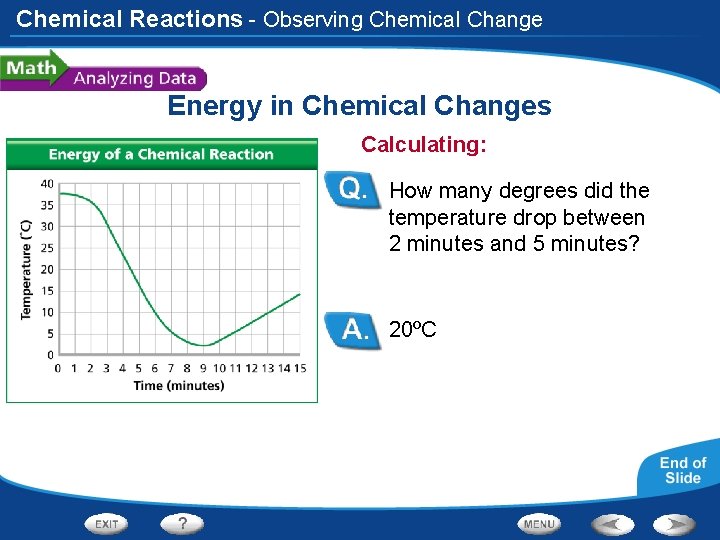
Chemical Reactions - Observing Chemical Change Energy in Chemical Changes Calculating: How many degrees did the temperature drop between 2 minutes and 5 minutes? 20ºC
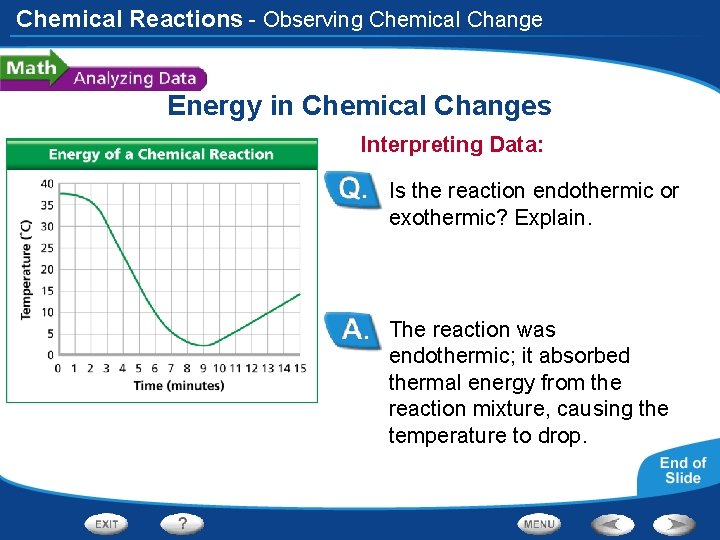
Chemical Reactions - Observing Chemical Change Energy in Chemical Changes Interpreting Data: Is the reaction endothermic or exothermic? Explain. The reaction was endothermic; it absorbed thermal energy from the reaction mixture, causing the temperature to drop.
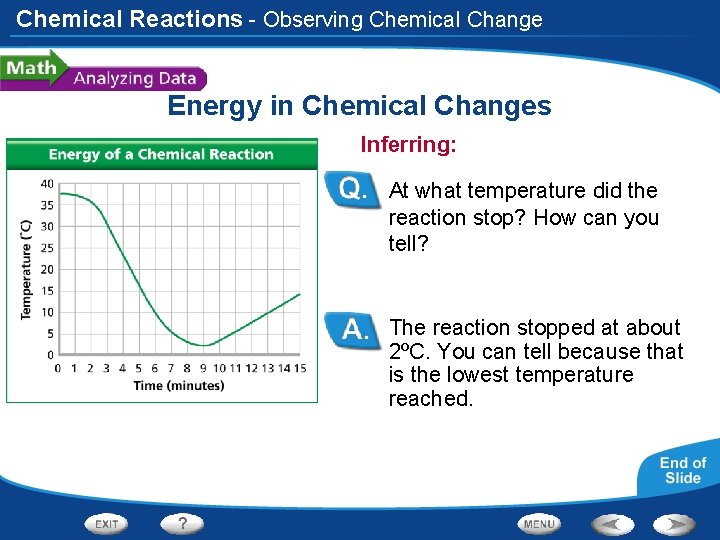
Chemical Reactions - Observing Chemical Change Energy in Chemical Changes Inferring: At what temperature did the reaction stop? How can you tell? The reaction stopped at about 2ºC. You can tell because that is the lowest temperature reached.
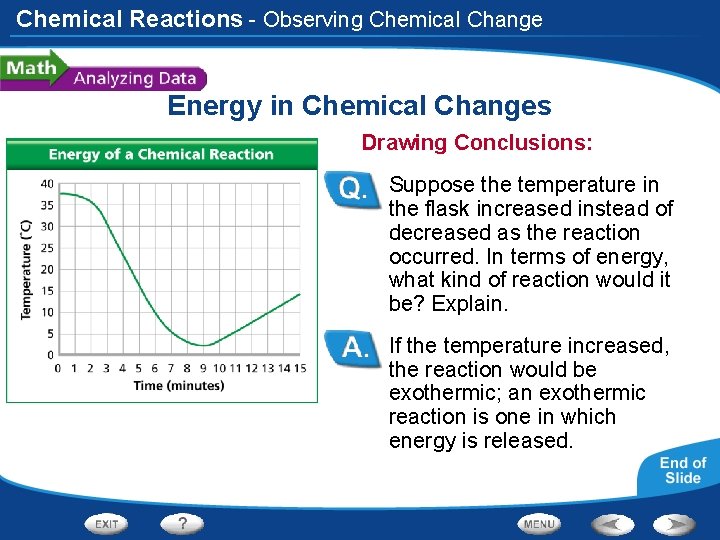
Chemical Reactions - Observing Chemical Change Energy in Chemical Changes Drawing Conclusions: Suppose the temperature in the flask increased instead of decreased as the reaction occurred. In terms of energy, what kind of reaction would it be? Explain. If the temperature increased, the reaction would be exothermic; an exothermic reaction is one in which energy is released.

Chemical Reactions - Observing Chemical Change Asking Questions Before you read, preview the red headings. In a graphic organizer like the one below, ask a what or how question for each heading. As you read, write answers to your questions. Properties and Changes of Matter Question Answer What are physical Physical properties are properties of matter? characteristics that can be observed without changing one substance into another. What is the evidence for The evidence for chemical reactions? reactions is the formation of new substances and changes in energy.

Chemical Reactions - Observing Chemical Change Links on Chemical Changes Click the Sci. Links button for links on chemical changes.

Chemical Reactions End of Section: Observing Chemical Change

Chemical Reactions Learning Objectives 1. Apply the principle of the conservation of mass to chemical reactions (in other words, balance chemical equations correctly). 2. Identify what a chemical equation contains (with an example). 3. Explain what a balanced chemical equation must show. 4. Identify the 4 main types of chemical reactions. (Synthesis tomorrow)

Chemical Reactions Conservation of Mass Demo (NO Lab Write-up) Goal- Determine if mass was conserved (remained constant) for the reaction of copper (II) sulfate with sodium hydroxide to form copper (II) hydroxide: Cu(OH)2. Data: Mass before = ? Mass After = ? Conclusion: Was mass conserved? How do you know?

Chemical Reactions - Describing Chemical Reactions Conservation of Mass The principle of conservation of mass states that in a chemical reaction, the total mass of the reactants must equal the total mass of the products. This is why we must balance chemical equations (because mass is conserved)!

Chemical Reactions Conservation of Mass Review For the reaction A + B 1. 2. 3. 4. AB, which masses are equal? Mass of A = Mass of B = Mass of AB Mass of A + Mass of B = Mass of AB

Chemical Reactions - Observing Chemical Change Properties and Changes in Matter Chemical reactions occur when bonds break and new bonds form.

Chemical Reactions - Describing Chemical Reactions Cellular phone messages make use of symbols and abbreviations to express ideas in shorter form. Similarly, chemists often use chemical equations in place of words.

Chemical Reactions - Describing Chemical Reactions What Are Chemical Equations? Chemical equations use chemical formulas and other symbols instead of words to summarize a reaction.

Chemical Reactions - Describing Chemical Reactions Chemical Formulas The formula of a compound identifies the elements in the compound and the ratios in which their atoms are present.

Chemical Reactions - Describing Chemical Reactions Balancing Chemical Equations Magnesium metal (Mg) reacts with oxygen gas (O 2), forming magnesium oxide (Mg. O). To write a balanced equation for this reaction, first write the equation using the formulas of the reactants and products, then count the number of atoms of each element.

Chemical Reactions Balancing Chemical Equations Mg + O 2 Mg. O
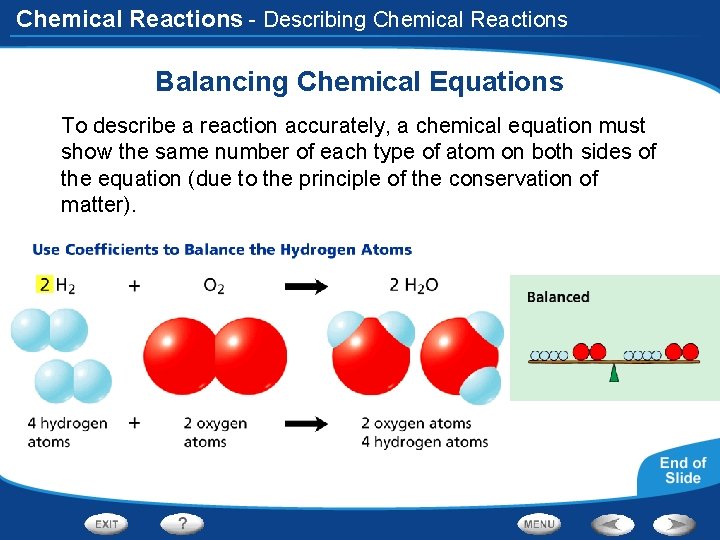
Chemical Reactions - Describing Chemical Reactions Balancing Chemical Equations To describe a reaction accurately, a chemical equation must show the same number of each type of atom on both sides of the equation (due to the principle of the conservation of matter).

Chemical Reactions Balancing Equations Practice K + Na + I 2 KI H 2 O Na. OH + Ag. NO 3 + K 2 S Cu. SO 4 + Na. OH H 2 KNO 3 + Na 2 SO 4 + Ag 2 S Cu(OH)2

Chemical Reactions Noggin Knockers

Chemical Reactions Synthesis Reaction Experiment Goals: Observe and write the balanced chemical equations for 2 synthesis reactions, and identify a synthesis reaction. Procedure: Given verbally. Do NOT touch the penny, tongs, or burner after heating! Only put a pre-1982 penny in the flame! Results: Observations of the penny after heating (but before you place it in the water) and after placing the penny in the water. Conclusion: Write the balanced equations for both reactions and describe the characteristics of a synthesis reaction. (Products: Red = Cu 2 O, Dull black = Cu. O)
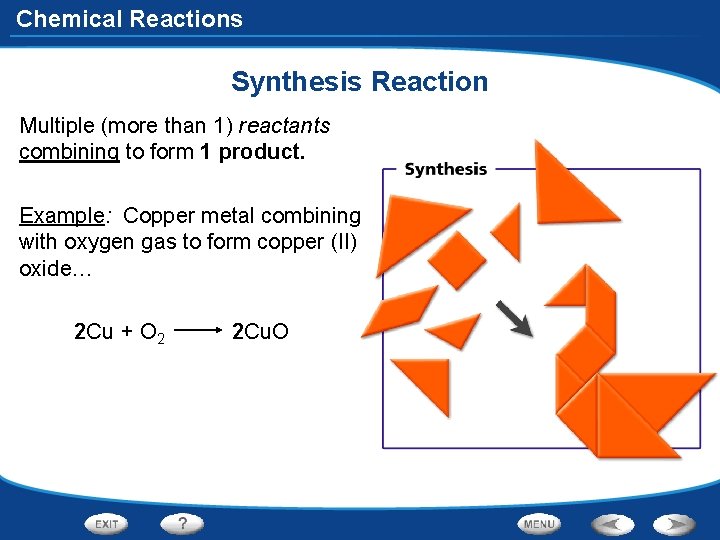
Chemical Reactions Synthesis Reaction Multiple (more than 1) reactants combining to form 1 product. Example: Copper metal combining with oxygen gas to form copper (II) oxide… 2 Cu + O 2 2 Cu. O

Chemical Reactions Synthesis Reaction Review 1. 2. 3. 4. 5. Identify all of the following chemical equations that represent a synthesis reaction: Mg + O 2 2 Mg. O AB A+B AB A + BC AC + B Cu. Cl 2 + Al Al. Cl 3 + Cu

Chemical Reactions Balancing Chemical Equations for Synthesis Reactions Na + Cl 2 Na. Cl Balance the equation for the reaction of sodium metal (Na) with oxygen gas (O 2), forming sodium oxide (Na 2 O). P + Cl 2 N 2 + H 2 PCl 3 NH 3

Chemical Reactions Ratios in Chemical Equations + Energy There’s a 2 to 1 ratio of hydrogen gas (H 2) to oxygen gas (O 2) that produces 2 water molecules (H 2 O). What’s the ratio of oxygen gas (O 2) to water (H 2 O)? 1 to 2

Chemical Reactions Noggin Knockers

Chemical Reactions Decomposition Reactions Goals: Observe and write the balanced chemical equations for 2 decomposition reactions (one from the demo), and identify this type of reaction. Procedure: Heat up a piece of chalk (Ca. CO 3) in the burner flame for at least 5 minutes. Then place the chalk in the cup with water and observe what happens. Results: Observations of the chalk after heating (but before you place it in the water) and after placing the chalk in the water. Conclusions: Write the balanced equations for both reactions (one is from the demo) and describe the characteristics of a decomposition reaction.

Chemical Reactions Balancing Equations (Decomposition Reactions)

Chemical Reactions Decomposition Reactions Only 1 reactant, but multiple products (more than 1). XY X+Y

Chemical Reactions Balancing Chemical Equations (Decomposition): Describe the last one in words & include the number of each molecule- Ion chart on p. 23 1. Hg. O 2. KCl. O 3 3. Na. HCO 3 Hg + O 2 KCl + Na 2 CO 3 + O 2 H 2 O + CO 2

Chemical Reactions Noggin Knockers

Chemical Reactions Single Replacement Reactions Goals: Observe and write the balanced chemical equations for 3 single replacement reactions (3 from the demos), and identify this type of reaction. Procedure: Place the iron nail into the blue solution of copper (II) sulfate. Take it out after a few seconds. Results: Observations of the nail before and after placing it in the copper (II) sulfate solution. Conclusions: Write the balanced equations for BOTH reactions (including the one from the demo) and describe the characteristics of a single replacement reaction.

Chemical Reactions Balancing Equations Demo 1: Demo 2:

Chemical Reactions Balancing Equations Demo 3 (Thermite):

Chemical Reactions Single Replacement Reaction One element (or type of atom) replaces another in a compound. XY + Z XZ + Y Analogy:

Chemical Reactions Noggin Knockers

Chemical Reactions Double Replacement Reactions Goals: Observe and write the balanced chemical equations for 6 double replacement reactions (5 from the lab and 1 from the video), and identify this type of reaction. Procedure: Place 3 -5 drops of the following solutions in their own well (in the chem. plate): Magnesium sulfate (Mg. SO 4), Nickel chloride (Ni. Cl 2), Copper (II) sulfate (Cu. SO 4), Iron (III) chloride (Fe. Cl 3), & Cobalt chloride (Co. Cl 2). Then add 3 -5 drops of the sodium silicate solution (Na 2 Si. O 3) to form metal silicates and stir with the clean end of a toothpick. Disposal- scoop out solids and put them in the trash and wipe the plate clean. WASH hands immediately if you get any chemical on them! Results: Observations of each reaction in terms of what formed. You may want to write down your observations under the chemical formula for the compound that formed in the equation for each reaction. Example: Al 2(Si. O 3)3 white Conclusions: Write the balanced equations for all reactions (including one from the video) and describe the characteristics of a double replacement reaction.

Chemical Reactions Balancing Equations (Silicate ion = Si. O 3 -2) Na 2 Si. O 3 + Mg. SO 4 Mg. Si. O 3 + Na 2 SO 4 Na 2 Si. O 3 + Fe. Cl 3 Fe 2(Si. O 3)3 + Na 2 Si. O 3 + Cu. SO 4 Cu. Si. O 3 + Na 2 Si. O 3 + Ni. Cl 2 Ni. Si. O 3 + Na. Cl Na 2 Si. O 3 + Co. Cl 2 Co. Si. O 3 + Na. Cl Na 2 SO 4
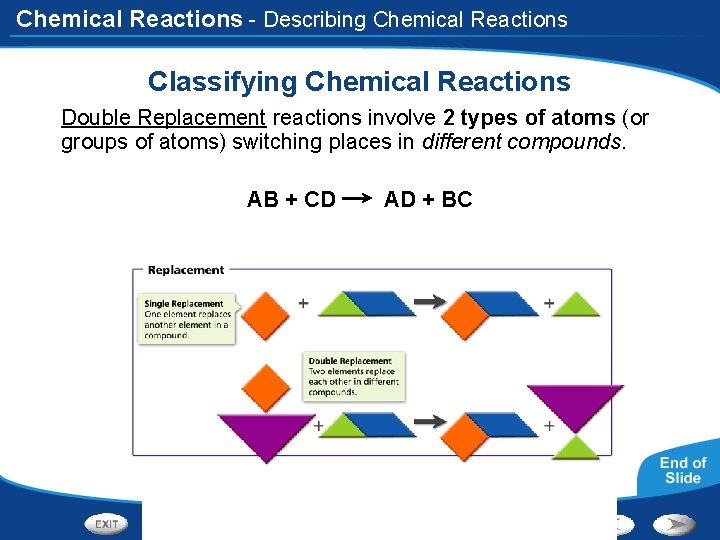
Chemical Reactions - Describing Chemical Reactions Classifying Chemical Reactions Double Replacement reactions involve 2 types of atoms (or groups of atoms) switching places in different compounds. AB + CD AD + BC

Chemical Reactions Double Replacement Reaction (Video)

Chemical Reactions Balancing Equations (Silicate ion = Si. O 3 -2) Na 2 Si. O 3 + Mg. SO 4 Mg. Si. O 3 + Na 2 SO 4 Na 2 Si. O 3 + Fe. Cl 3 Fe 2(Si. O 3)3 + Na 2 Si. O 3 + Cu. SO 4 Cu. Si. O 3 + Na 2 Si. O 3 + Ni. Cl 2 Ni. Si. O 3 + Na. Cl Na 2 Si. O 3 + Co. Cl 2 Co. Si. O 3 + Na. Cl Na 2 SO 4

Chemical Reactions For a single replacement reaction… A. B. C. D. Two types of atoms switch places between compounds. One type of atom replaces another in a compound. There always 2 reactants and 1 product. There is only one reactant.

Chemical Reactions Which of the following represents a single replacement reaction? A. B. C. D. AB A+B AB + CD AD + BC AB + C AC + B A+B AB

Chemical Reactions Which of following only shows copper (Cu) being replaced by silver (Ag)? A. B. C. D. 2 Ag + Cu. SO 4 Ag 2 SO 4 + Cu Ag. Cu Cu. Cl 2 Cu + Cl 2 Cu. SO 4 + 2 Ag. NO 3 Cu(NO 3)2 + Ag 2 SO 4

Chemical Reactions How can you tell if a chemical reaction is a decomposition reaction? A. B. C. D. If there’s 2 or more products. If there’s 2 or more reactants. If there’s only 1 reactant. If there’s only 1 product.

Chemical Reactions Why are synthesis and decomposition reactions considered opposites? A. They both have only one reactant. B. Synthesis reactions have 2 products while decomposition reactions have only 1 product. C. They’re not opposites. D. Synthesis reactions have only 1 product while decomposition reactions have only 1 reactant.

Chemical Reactions Which of the following represents a decomposition reaction? A. B. C. D. A+B AB AB A+B AB + C AC + B AB + CD AD + BC

Chemical Reactions Which of the following shows 2 molecules of hydrogen peroxide (H 2 O 2) breaking down into 2 molecules of water and 1 molecule of oxygen gas (O 2)? A. B. C. D. 2 H 2 O + O 2 2 H 2 O + O 2 H 2 O 2

Chemical Reactions For a double replacement reaction… A. Two types of atoms or groups of atoms switch places between compounds. B. One type of atom replaces another in a compound. C. There always 2 reactants and 1 product. D. There is only one reactant.

Chemical Reactions Which of the following represents a double replacement reaction? A. B. C. D. AB A+B AB + CD AD + BC AB + C AC + B A+B AB

Chemical Reactions Which of the following is a double replacement reaction? A. B. C. D. 2 Ag + Cu. SO 4 Ag 2 SO 4 + Cu Ag. Cu Cu. Cl 2 Cu + Cl 2 Cu. SO 4 + 2 Ag. NO 3 Cu(NO 3)2 + Ag 2 SO 4

Chemical Reactions Math

Chemical Reactions Math

Chemical Reactions Noggin Knockers

Chemical Reactions Combustion Reactions (Balance for Extra Credit on Test) Film Canister Rocket (Combustion of methanol): CH 4 O + O 2 CO 2 + H 2 O Ping Popper (Combustion of ethanol): C 2 H 6 O + O 2 CO 2 + H 2 O Whoosh Bottle (Combustion of 2 -propanol AKA isopropyl alcohol): C 3 H 8 O + O 2 CO 2 + H 2 O

Chemical Reactions Homework- p. 63: 1 a, 2 b, 3 a, 3 b, 3 c, 4, & 5 1 a- Formulas- number of each type of atom, arrow means “yields” or “produces”. Plus sign- what’s reacting together and/or what 2 substances are produced because they separate the substances in the equation. 2 b- 250 g (Mass of reactants = Mass of the products) 3 a- Synthesis, Decomposition, Replacement (Single & Double) 3 b- 2 products minimum 3 c- Synthesis (1 product) 4 - 2 Fe 2 O 3 + 3 C 5 - 2 SO 2 + O 2 4 Fe + 3 CO 2 2 SO 3

Chemical Reactions Practice Problems

Chemical Reactions - Describing Chemical Reactions Building Vocabulary Using a word in a sentence helps you think about how best to explain the word. After you read the section, reread the paragraphs that contain definitions of Key Terms. Use the information you have learned to write a meaningful sentence using each Key Terms: closed system chemical equation replacement reactant coefficient product Examples: In chemical A a closed system, equationmatter is a short, is noteasy allowed way to to enter show or a Replacement is the process in which one element chemical reaction. leave. replaces another in a compound or two elements in you at the a reaction is A substance coefficient is ahave number in abeginning chemicalof equation different compounds trade places. a reactant. telling you how many atoms or molecules of a reactant or product take part in the reaction. A new substance produced in a reaction is a product. synthesis conservation of mass Synthesis means combining two or during more elements or Conservation of mass means that a chemical compounds to make more complex substance. reaction, matter is nota created or destroyed. open system decomposition In an open system, matter can enter from or escape Decomposition means breaking down compounds to the surroundings. into simpler products.

Chemical Reactions End of Section: Describing Chemical Reactions

Chemical Reactions Learning Objective (Pg. 19 of note packet) Describe how the following affect the rate of a chemical reaction… • Temperature • Using a catalyst • Surface Area • Using an inhibitor • Concentration
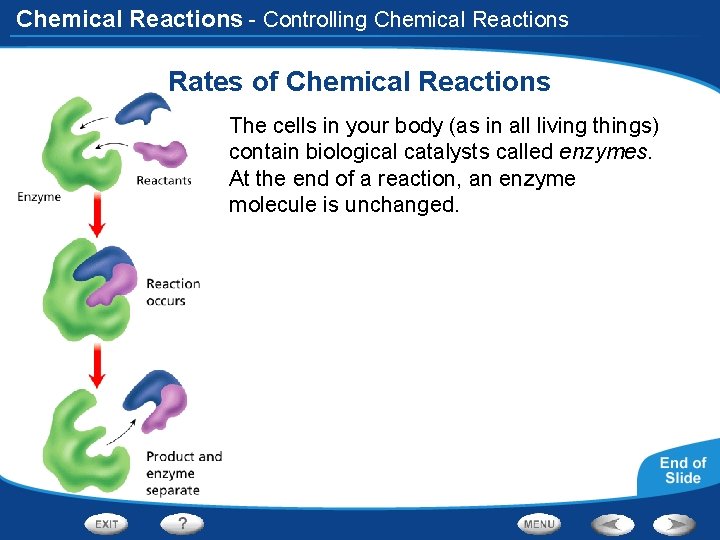
Chemical Reactions - Controlling Chemical Reactions Rates of Chemical Reactions The cells in your body (as in all living things) contain biological catalysts called enzymes. At the end of a reaction, an enzyme molecule is unchanged.
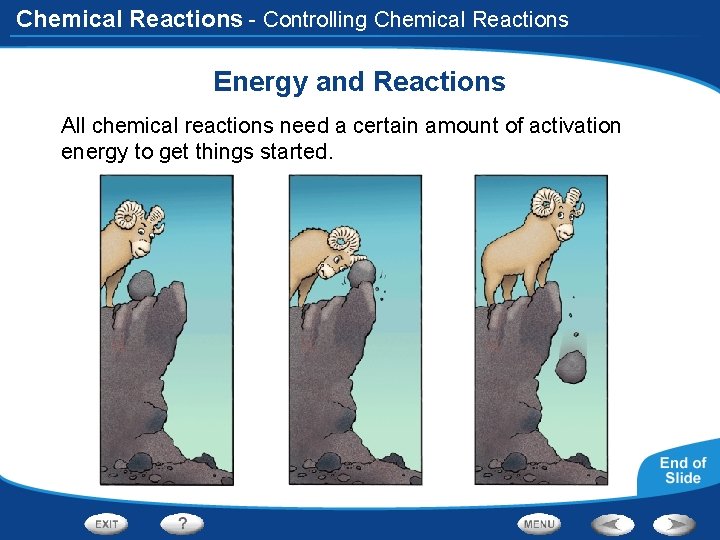
Chemical Reactions - Controlling Chemical Reactions Energy and Reactions All chemical reactions need a certain amount of activation energy to get things started.
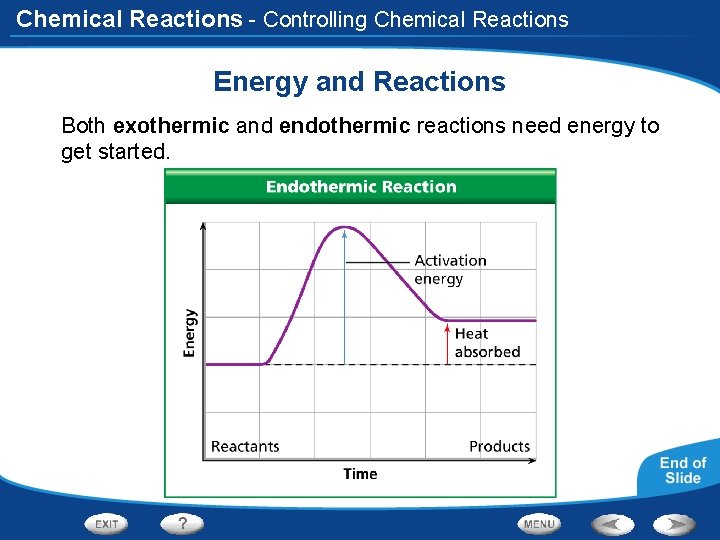
Chemical Reactions - Controlling Chemical Reactions Energy and Reactions Both exothermic and endothermic reactions need energy to get started.

Chemical Reactions - Controlling Chemical Reactions Relating Cause and Effect As you read, identify the factors that can cause the rate of a chemical reaction to increase. Write the information in a graphic organizer like the one below. Causes Increase in surface area Increase in temperature Increase in concentration of reactants Use of a catalyst Effect Increased rate of reaction

Chemical Reactions Homework- p. 63: 1 a, 2 b, 3 a, 3 b, 3 c, 4, & 5 1 a- Formulas- number of each type of atom, arrow means “yields” or “produces”. Plus sign- what’s reacting together and/or what 2 substances are produced b/c they separate the substances in the equation. 2 b- 250 g (Mass of reactants = Mass of the products) 3 a- Synthesis, Decomposition, Replacement (Single & Double) 3 b- 2 products minimum 3 c- Synthesis (1 product) 4 - 2 Fe 2 O 3 + 3 C 5 - 2 SO 2 + O 2 4 Fe + 3 CO 2 2 SO 3

Chemical Reactions Homework- p. 71: 2 a 2 a- Speed up a reaction- increase temperature, increase surface area, increase concentration of the reactants, or add a catalyst; slow down a reaction- decrease temperature, decrease surface area, decrease concentration of the reactants; add an inhibitor.

Chemical Reactions Data Sharing Lab Click the PHSchool. com button for an activity about sharing data for the Skills Lab Temperature and Enzyme Activity.

Chemical Reactions - Controlling Chemical Reactions Exothermic and Endothermic Reactions Click the Video button to watch a movie about exothermic and endothermic reactions.

Chemical Reactions End of Section: Controlling Chemical Reactions
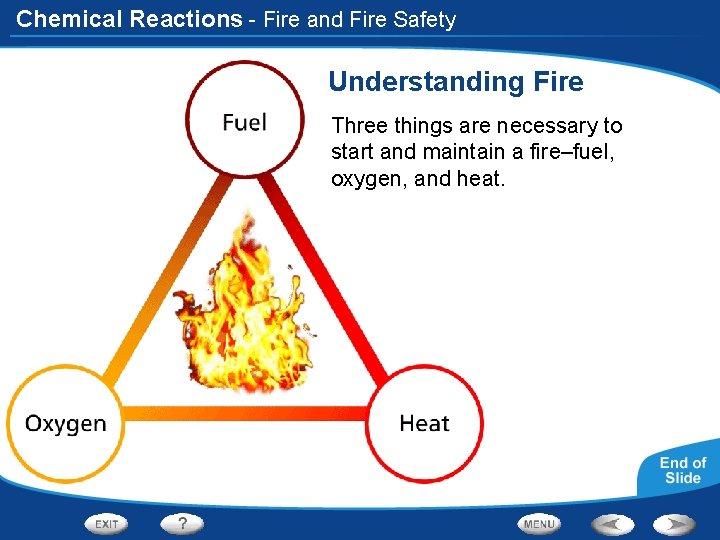
Chemical Reactions - Fire and Fire Safety Understanding Fire Three things are necessary to start and maintain a fire–fuel, oxygen, and heat.

Chemical Reactions - Fire and Fire Safety Understanding Fire The fire triangle can be controlled in the grill. If any part of the fire triangle is missing, the fire will not continue.
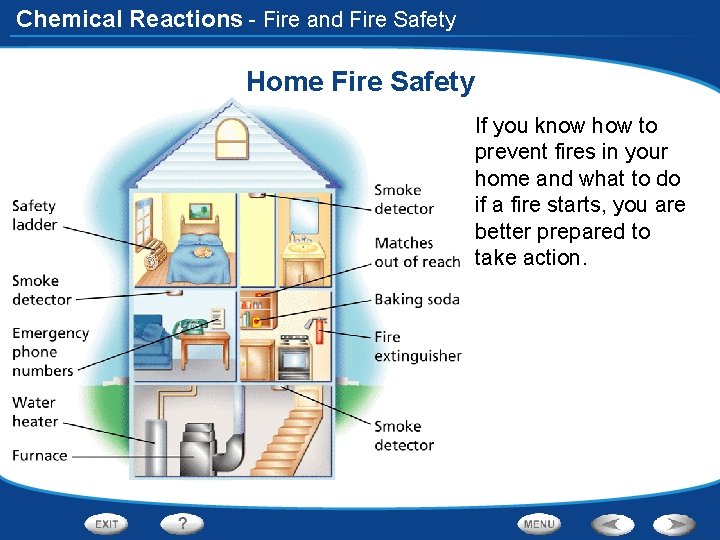
Chemical Reactions - Fire and Fire Safety Home Fire Safety If you know how to prevent fires in your home and what to do if a fire starts, you are better prepared to take action.

Chemical Reactions - Fire and Fire Safety Using Prior Knowledge Before you read, write what you know about fire safety in a graphic organizer like the one below. As you read, write what you learn. What You Know 1. A fire needs fuel to burn. 2. A fire need oxygen to burn. 3. All homes should have smoke detectors. What You Learned 1. Fire is a result of a combustion reaction. 2. The most deadly fires start with cigarettes. 3. Baking soda can be used to put out small fires.

Chemical Reactions - Fire and Fire Safety Links on Fire Safety Click the Sci. Links button for links on fire safety.

Chemical Reactions End of Section: Fire and Fire Safety

Chemical Reactions Graphic Organizer Chemical reactions can be Endothermic reactions are written as Exothermic reactions Equations balanced with which require Activation energy written using Chemical symbols Coefficients Arrows and plus signs

Chemical Reactions End of Section: Graphic Organizer
- Slides: 81